Abstract
The solubility data of sodium theophylline salt in the water and aqueous sodium hydroxide systems with molarities of 0.00, 0.01, 0.10, 0.20, 0.49, and 0.96 mol kg−1 at temperatures ranging from 283.15 to 368.15 K are determined via the equilibrium method. The experimental results show that the solubility of sodium theophylline increases on increasing the temperature at the same concentration of NaOH solution and decreases with an increase in the NaOH concentration at the same temperature. These results correlate well with the polynomial empirical equation, the modified Apelblat equation, and the λh equation. At the same time, it is found that the modified Apelblat equation gives better correlation results than the polynomial empirical and the λh equations. The results show that the three models correlate well with the solubility data of sodium theophylline. The modified Van’t Hoff equation is used to estimate the thermodynamic properties including the dissolution enthalpy, the dissolution entropy, and the Gibbs free energy change of sodium theophylline when dissolved in aqueous NaOH solution at different concentrations. The solubility data and correlation results obtained from the experiments can provide basic data for the cooling crystallization, separation, and purification of sodium theophylline and are expected to be of great significance for the research and development of sodium theophylline and for caffeine production.
Keywords

Introduction
Sodium theophylline salt, referred to as sodium theophylline (the chemical name is 1,3-dimethylxanthine sodium salt, Figure 1), is a strong alkali salt of the weak organic acid theophylline (pKa = 8.43 at 298.15 K, 1 LogP value is −0.02 2 ). It is a white crystalline powder with a melting point of 664.15 K 3 and is soluble in water, but is insoluble in ethanol. It can react with dimethyl sulfate to produce caffeine and is therefore an important intermediate in the production of caffeine. 4 Meanwhile, sodium theophylline can dilate coronary arteries, cause diuresis, and stimulate the central nervous system.5,6 It can also be used to treat bronchial asthma, cardiogenic dyspnea, and other symptoms.7–11 Sodium theophylline is usually synthesized from 6-amino-5-formamide, 1,3-dimethyluracil, and sodium hydroxide in a closed loop in a strongly alkaline environment. 12
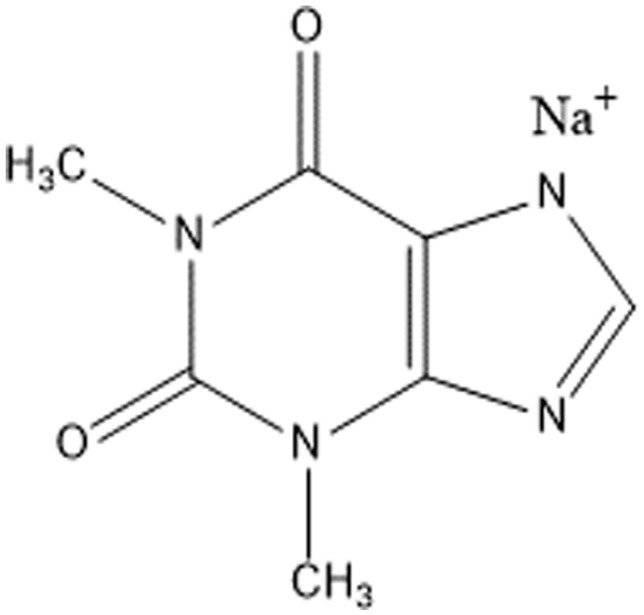
Structure of sodium theophylline (C7H7N4O2Na).
The industrial production of sodium theophylline includes two processes: reactions and solid–liquid separation and purification. 6-amino-5-formamide, 1,3-dimethyluracil, and sodium hydroxide generate sodium theophylline under alkaline conditions. Too low an alkalinity will make the reaction incomplete and reduce the yield of sodium theophylline. However, too high an alkalinity may not only destroy the generated sodium theophylline and reduce the yield but also leads to the waste of excess sodium hydroxide. 4 In addition, in the subsequent solid–liquid separation process, the alkaline strength can also result in the loss of the product solid sodium theophylline in the mother liquor. Therefore, to find an economical and feasible solution to effectively reduce the content of sodium theophylline in the mother liquor, and to take into account the reaction and solid–liquid separation process conditions, investigations on the solubility of sodium theophylline at different sodium hydroxide concentrations and different temperatures were carried out. An understanding of the solubility of sodium theophylline is of great significance for improving the yield of the crystallization process, reducing product loss and improving industrial production efficiency. Unfortunately, the solubility data of sodium theophylline in lye are rarely reported. 13
In this work, in the temperature range of 283.15–368.15 K, the solubility data of sodium theophylline in different concentrations of aqueous NaOH solution between 0.00 and 0.96 mol kg−1 were measured by the equilibrium method. The polynomial empirical equation, the modified Apelblat equation, and the λh equation were employed to correlate the solubility data. In addition, thermodynamic properties such as the Gibbs free energy change, the dissolution entropy, and the dissolution enthalpy of the dissolution process of sodium theophylline were estimated based on the modified Van’t Hoff equation.
Thermodynamic model
The relationship between the solubility and temperature can be described by a number of thermodynamic methods. The modified Apelblat equation and the λh equation are commonly used in solubility predictions based on non-ideal solutions.14,15 To quantitatively describe the relationship between solubility and temperature in a solid–liquid equilibrium, we used the Apelblat equation and the λh equation to fit the solubility data in this work. At the same time, from the perspective of industrial applications, the simpler the equation of solid–liquid equilibrium is, the more convenient the application is. Therefore, for practical applications, the polynomial empirical equation is also used in this work to correlate the relationship between solubility and temperature. 16
Polynomial empirical equation
After the factors such as solute, solvent, and pressure are determined, the solubility is determined by temperature. If the solubility varies continuously with temperature, it can be expressed as a polynomial temperature equation. 17 The expression of the empirical equation of the polynomial thus obtained is
where T is the absolute temperature (unit is K), x1 is the mole fraction of the solute, and A, B, and C are the fitting parameters of the polynomial empirical equation, respectively.
Apelblat equation
The modified Apelblat model is a semi-empirical equation originally proposed by Apelblat and Manzurola. The equation has a simple structure and is convenient for regression calculation. It contains three adjustable parameters, which have a good correlation between solubility and temperature, and can correlate well the equilibrium data of various systems and is more commonly used by researchers. Therefore, we chose the Apelblat equation for thermodynamic calculations and expected to fit the solubility data of sodium theophylline using this equation to obtain better fitting results.18–21 The Apelblat equation is
where T is the absolute temperature (unit is K), x1 is the mole fraction of the solute, and A, B, and C are empirical constants. The constants A and B represent the change in the activity coefficient of the solution and provide an indication of the effect of a non-ideal solution on the solubility of the solute; the parameter C reflects the effect of temperature on the enthalpy of melting.
λh equation
In 1980, Buchowski et al. studied the solubility behavior of phenol and benzoic acid in nonpolar solvents and delineated the relationship between solvent activity, solubility, and temperature. The solubility model (λh equation) in the binary system was thus derived.22–24 The λh equation can well relate most solid–liquid equilibrium systems to two adjustable parameters. At the same time, it directly correlates solubility with temperature; the physical property data only need melting point data, thereby avoiding the activity coefficient. The equation form is simple and the calculation process is simplified. At present, the λh equation well describes many solid–liquid systems. Therefore, we chose the λh equation for thermodynamic calculations and expected to fit the solubility data of sodium theophylline using the λh equation to obtain better fitting results. The λh equation is
where T is the absolute temperature (unit is K), x1 is the mole fraction of the solute, λ and h are the fitting parameters, and Tm is the melting temperature of sodium theophylline (unit is K); here, its value is 666.84 K.
Results and discussion
Melting properties of sodium theophylline
Sodium theophylline exists most of the time in the form of a monohydrate. The first endothermic peak (the detailed DSC spectrum is shown in the Supporting Information in Figure S1) represents the removal of a molecule of water from a sample of sodium theophylline, and the second endothermic peak represents the melting of sodium theophylline itself. 3 The two endothermic peaks observed in this work are basically consistent with those in literature. 3
Figure 2 shows the DSC heat map of sodium theophylline. 25 For clarity, only the peaks of the melting process are intercepted in Figure 2. The melting point of sodium theophylline (the onset temperature Tm of the melting process) is 666.84 K (u(Tm) = 0.5 K). The enthalpy of fusion (ΔfusHm) of sodium theophylline is −279.63 kJ mol−1 (ur(ΔfusHm) = 0.02). In addition, the entropy of fusion of sodium theophylline can be calculated. From the DSC data, the entropy of fusion (ΔfusSm) is found to be −419.34 J mol−1k−1 (ur(ΔfusSm) = 0.02). The melting temperature measured by the experiment is 666.84 K, which is close to that reported in the literature (664.15 K). 3 The discrepancies between this work and previous reports may stem from the differences in the testing apparatus and heating rates.
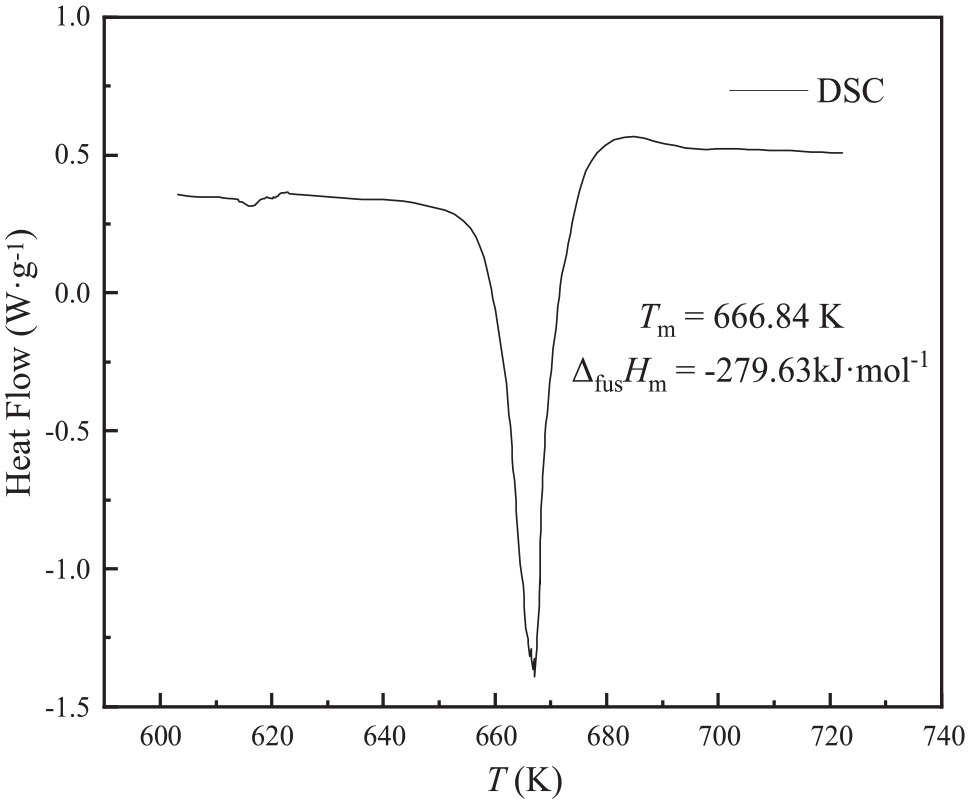
Partial DSC spectrum of sodium theophylline.
Solubility data and model correlation
The solubility data of sodium theophylline in different concentrations of aqueous NaOH solution (between 0.00 and 0.96 mol kg−1; represented by the mole fraction x of the solute sodium theophylline) at temperatures ranging from 283.15 to 368.15 K were measured, and the results are shown in Figures 3–6. It can be seen from these figures that the solubility of sodium theophylline in different solutions increased with an increase in temperature, but the trend was different. In low-concentration (e.g. 0.01 mol kg−1) aqueous NaOH solution, the solubility increased on increasing the temperature, and the growth rate was faster, while the growth rate was relatively slow in high-concentration aqueous NaOH solution. At the same temperature, the solubility of sodium theophylline in water was the highest, and the solubility in 0.96 mol kg−1 aqueous NaOH solution was the lowest. Also at the same temperature, the solubility of sodium theophylline decreases on increasing the NaOH concentration. Sodium theophylline is a weak polar compound with a dielectric constant lower than that of water. Under the action of an electric field, the water molecules with a higher dielectric constant gather around Na+ and OH−, and the sodium theophylline, with a lower dielectric constant, is expelled from the vicinity of Na+ and OH−, resulting in supersaturation. The excess sodium theophylline is precipitated from the water, resulting in a salting-out effect, thereby reducing the solubility of sodium theophylline in aqueous NaOH solution. In addition, NaOH is a strong alkali, and the increase in NaOH content makes the solution more alkaline such that the solubility of sodium theophylline, which is also an alkali, is reduced. Another reason is the co-ion effect of NaOH and sodium theophylline. Adding NaOH with the same Na+ cation as that in sodium theophylline solution leads to the dissociation balance of sodium theophylline being shifted toward generating sodium theophylline molecules, thereby resulting in a decrease in the solubility of sodium theophylline.

Molar fraction solubilities of sodium theophylline in selected solutions; the solid lines represent the fit to the experimental data using the polynomial empirical equation.
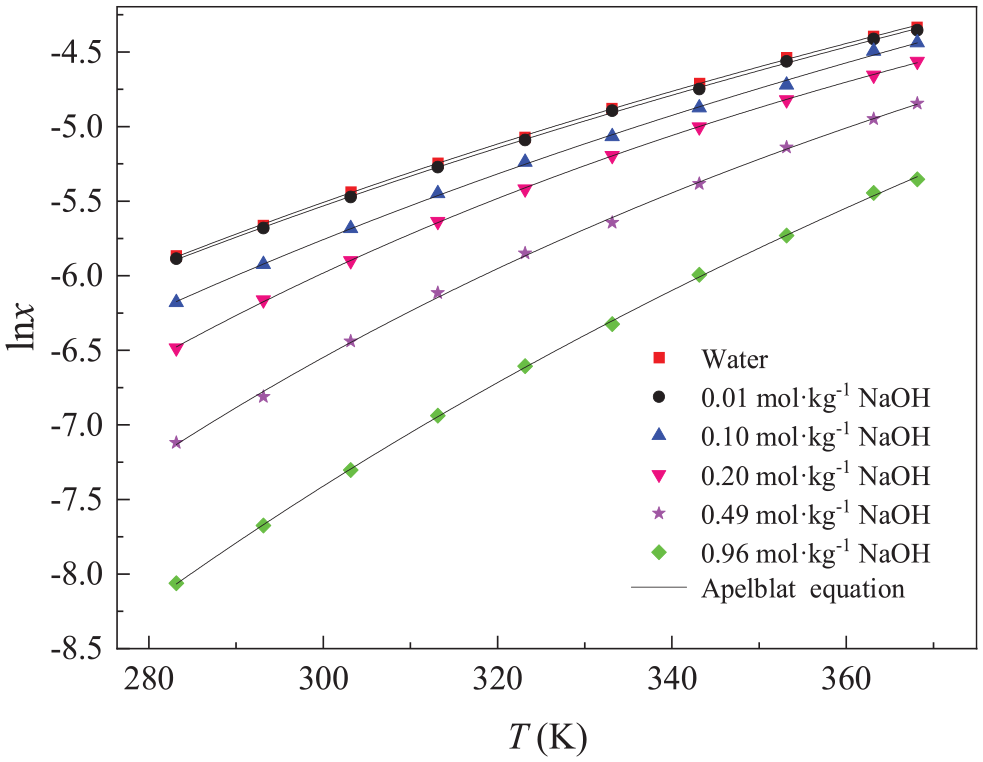
Molar fraction solubilities of sodium theophylline in selected solutions; the solid lines represent the fit to the experimental data using the Apelblat equation.

Molar fraction solubilities of sodium theophylline in selected solutions; the solid lines represent the fit to the experimental data using the λh equation.
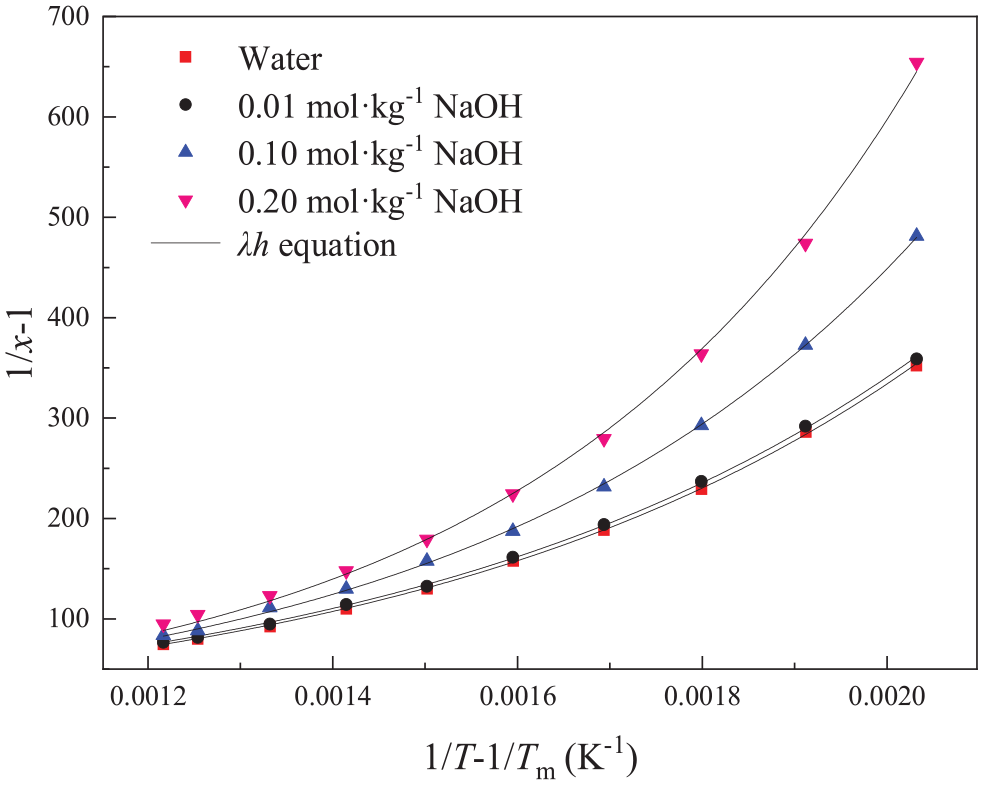
Molar fraction solubilities of sodium theophylline in selected solutions; the solid lines represent the fit to the experimental data using the λh equation (local amplification).
The experimental data were correlated with the polynomial empirical equation, the Apelblat equation, and the λh equation. To evaluate the applicability and accuracy of the three models, the relative deviation (RD), the average relative deviation (ARD), and the root mean square deviation (rmsd) are used, which are defined as follows26,27
where xcal is the calculated value (mole fraction) and xexp is the experimental value (mole fraction); k is the number of experimental points.
The standard uncertainty of temperature is u(T) = 0.03 K, the standard uncertainty of pressure is u(p) = 0.4 kPa, and the relative standard uncertainty of the solute mole fraction is ur(xexp) = 0.03. Figures 3–6 show the fitting results of the three models and Tables 1–3 list the relevant fitting parameters and the rmsd values of the three correlation models. The average ARDs of the polynomial empirical equation, the Apelblat equation, and the λh equation are 1.98%, 0.98%, and 1.70%, respectively (the RD and ARD values are shown in the Supporting Information in Table S1). The results show that the solubility values calculated by these three models are in good agreement with the measured data.
Parameters of the polynomial empirical equation of sodium theophylline in different concentrations of aqueous NaOH solution.

rmsd is the root mean square deviation, calculated using equation (6).
Parameters of the Apelblat equation of sodium theophylline in different concentrations of aqueous NaOH solution.

rmsd is the root mean square deviation, calculated using equation (6).
Parameters of the λh equation of sodium theophylline in different concentrations of aqueous NaOH solution.

rmsd is the root mean square deviation, calculated using equation (6).
Fitting results of different models
From the experimental data, the average ARD values of the polynomial empirical equation, the Apelblat equation, and the λh equation are 1.98%, 0.98%, and 1.70%, respectively. Tables 1–3 show that all three models can be used for pseudo-binary systems containing electrolytes and are well correlated with the solubility data of sodium theophylline. From Tables 1–3, it can be seen that the maximum ARD value related to the Apelblat equation is 1.70% and the maximum rmsd is 13.609 × 10−5, the maximum ARD value related to the polynomial empirical equation is 5.51% and the maximum rmsd is 14.933 × 10−5, and the maximum ARD value related to the λh equation is 3.20% and the maximum rmsd is 33.941 × 10−5 (the RD and ARD values are shown in the Supporting Information in Table S1). Comparatively speaking, the fitting result of Apelblat equation is better than that of λh equation and polynomial empirical equation.
Dissolution properties of sodium theophylline
According to the Van’t Hoff equation, the logarithm of the mole fraction of solute in a solution is linearly related to the reciprocal of the absolute temperature difference. 28 This work uses the Van’t Hoff model to calculate the thermodynamic parameters of the dissolution process. 29
The Van’t Hoff equation is

where T is the absolute temperature (unit is K), x1 is the mole fraction of the solute, R is the ideal gas constant (8.314 J mol−1 K−1), ΔHdiss is the dissolution enthalpy (kJ mol−1), and ΔSdiss is the dissolution entropy (J mol−1 K−1).
The Van’t Hoff equation can be simplified to equation (8)

The molar Gibbs energy change of dissolution can be calculated as

where ΔGdiss is the dissolution Gibbs free energy change (kJ mol−1), a and b are the semi-empirical constants of the Van’t Hoff equation, and Tmean is calculated from equation (10)

where Ti is the experimental temperature, n is the number of experimental points, and Tmean is the average temperature (the average temperature calculated in this paper is 327.65 K).
According to equation (8), a straight line is obtained by plotting 1/T with lnx1. According to slope a and the intercept b of the straight line, the dissolution enthalpy and dissolution entropy of the solution can be calculated. The dissolution Gibbs free energy change can be calculated from equation (9).
By comparing the contribution of enthalpy and entropy to the Gibbs free energy change in the dissolution process, the dissolution principle of the dissolution process can be determined.


The thermodynamic parameters calculated for the dissolution of sodium theophylline in aqueous NaOH solution at different concentrations are listed in Table 4 and the fitting results are shown in Figure 7. As can be seen from Table 4, ΔHdiss>0, ΔSdiss>0, ΔGdiss>0 for the dissolution process of sodium theophylline in different concentrations of aqueous NaOH solution is an endothermic process and an entropy-increase process. This result is consistent with the solubility increasing with temperature. With increasing NaOH concentration, the dissolution of the enthalpy of sodium theophylline gradually increases. Meanwhile, the value of
Dissolution properties of sodium theophylline in different solvents.

ΔSdiss, ΔHdiss, and ΔGdiss represent the dissolution entropy, the dissolution enthalpy, and the dissolution Gibbs free energy change, respectively, which can be calculated from the model parameters of the Van’t Hoff model and equations (7)–(10).

The Van’t Hoff plot of the logarithmic molar fraction solubility of sodium theophylline in the selected solutions versus the reciprocal of temperature.
Conclusion
(1) The solubility data of sodium theophylline in different concentrations of aqueous NaOH solution between 0.00 and 0.96 mol kg−1 have been determined by the equilibrium method at T = 283.15–368.15 K. The results showed that the solubility of sodium theophylline increases with increasing temperature, while the solubility of sodium theophylline decreases with an increase in the concentration of aqueous NaOH solution.
(2) The polynomial empirical equation, the Apelblat equation, and the λh equation were used to correlate the solubility data. The experimental and calculated values are in good agreement. The average ARD values of the polynomial empirical equation, the Apelblat equation, and the λh equation are 1.98%, 0.98%, and 1.70%, respectively. They indicate that the three models can well correlate the solubility data of sodium theophylline in aqueous NaOH solutions of different concentrations.
(3) The solubility thermodynamic properties (ΔHdiss, ΔSdiss, and ΔGdiss,
Experimental
Materials and instruments
Sodium theophylline was obtained from Shandong Xinhua Pharmaceutical Co., Ltd.; sodium hydroxide was purchased from Sinopharm Chemical Reagent Co., Ltd.; acetic acid and methanol were obtained from Macklin. Ultrapure water was prepared in our laboratory. The reagents and materials used in the experiments were of analytical grade and were used without further purification. The materials used in the experiment are described in Table 5.
Source and mass fraction purity of the materials.

The purity of the raw materials and mass fractions was provided by the supplier.
The electrical resistivity of ultrapure water is 18.2 MΩ cm at 298.15 K.
A constant temperature oscillator (SHA-C, Changzhou Huapuda Teaching Instrument Co., Ltd., temperature control accuracy ± 0.1 K), an electronic balance (BSA224S, Beijing Sartorius Scientific Instruments Co., Ltd., measurement accuracy ± 0.1 mg), an ultrapure water machine (UPR-II-10T, Xi’an Youpu Instrument Equipment Co., Ltd.), and a high-performance liquid chromatography (HPLC) (U3000, Thermo Fisher Scientific, equipped with a UV detector RS-VWD and a fluorescence detector RS-FLD detector) were employed.
Preparation of aqueous NaOH solutions
During this process, a BSA224S analytical balance with a sensitivity of ± 0.1 mg was used to prepare 0.01, 0.10, 0.20, 0.49, and 0.96 mol kg−1 aqueous NaOH solutions with ultrapure water.
Solubility measurements
Common methods for measuring solubility include the equilibrium method30,31 and the dynamic method. 32 In this work, the equilibrium method was used to determine the solubility.
An appropriate amount of ultrapure water, aqueous NaOH solutions with molar concentrations of 0.01, 0.10, 0.20, 0.49, and 0.96 mol kg−1, and excess sodium theophylline were added to a conical flask. The conical flasks were placed in a constant temperature oscillator and oscillated (oscillation rate: 150 r·min−1) for 2 h and then allowed to stand still until the upper solution had clarified. Then at intervals, about 1 mL of liquid was taken out with a syringe with a 0.45 μm filter and weighed to determine the weight of the saturated solution. The saturated liquid was diluted 2000 times with ultrapure water and then analyzed by HPLC. Finally, the mass fraction of sodium theophylline in the saturated solution was determined by HPLC. This process was repeated at regular intervals until the relative error of the analyzed data of the two adjacent samples was within 2%. In this way, the solubility data of sodium theophylline in solution were obtained, and the temperature of the system was maintained constant throughout the process. Next, the solubility data of sodium theophylline in different concentrations of aqueous NaOH solution at different temperatures were obtained by changing the system temperature and repeating the above steps.
The solubility data were determined using an HPLC system. Chromatographic analysis was performed on a chromatographic column (Hypersil GOLD C18, 150 mm × 4.6 nm, 2.6 μm), and the detector wavelength was set to 270 nm. The two mobile phases were 0.5% aqueous acetic acid solution and methanol, respectively. The mobile phase flow rate was 1 mL·min−1. A reference standard solution containing approximately 0.3 mg·mL−1 of sodium theophylline was prepared in water. The injection volumes of the sample and reference standard solution were 20 μL.
Under the above chromatographic conditions, the chromatogram of the sodium theophylline sample is shown in Figure 8, the retention time of sodium theophylline is 7.843 min, and the calibration curve is shown in Figure 9. Equation (13) is used to generate the calibration curve. In this work, the mass fraction ws of sodium theophylline salt in aqueous NaOH solution was determined by HPLC. The formula used for the calculation is shown in equation (14) (unit is μg g−1). The mass fraction ws was converted into mole fraction xs using equations (15) and (16).

Chromatogram of the sodium theophylline standard sample.

Calibration curve of the sodium theophylline standard sample.
The equation for the calibration curve is

where y is the peak area of sodium theophylline and x is the concentration of sodium theophylline.
The equation for calculating the mass fraction ws in HPLC is

where C0 is the concentration of sodium theophylline (μg·mL−1), which can be calculated according to equation (13) (equivalent to x), N is the constant volume (mL), and m is the total mass of liquid in the syringe (g).
The molar fraction of sodium theophylline in water is calculated using equation (15)

The molar fraction of sodium theophylline in aqueous NaOH solution is calculated using equation (16)

where ws is the mass fraction of sodium theophylline in the syringe, which can be analyzed by the HPLC, w is the initial mass fraction of sodium hydroxide solution, m is the total mass of liquid in the syringe (g), MN, MW, and MS are the relative molecular masses of NaOH, water, and sodium theophylline (g mol−1), respectively. 33
Equilibrium time determination and deviation analysis
To determine the equilibrium time of the system, taking an aqueous NaOH solution with a molar concentration of 0.10 mol kg−1 as an example, the samples were analyzed every 30 min at 333.15 K, and the mass fractions of sodium theophylline measured at different times were 4.01%, 6.26%, 6.99%, 7.04%, and 7.04%, respectively. It can be seen from the data that the system had reached equilibrium within 120 min. In the experiment, to ensure sufficient time for the establishment of the dissolution equilibrium, samples were analyzed after standing for 3 h.
To ensure the accuracy of sample analysis, the same sample was analyzed three times, and the average value was taken as the experimental value. Taking a sample of aqueous NaOH solution with a molar concentration of 0.20 mol kg−1 and a temperature of 368.13 K as an example, the mass fractions of sodium theophylline analyzed three times were 11.22%, 11.22%, and 11.25% respectively, with the average value being 11.23%; the maximum RD was 0.2%.
Melting properties measurements
Under nitrogen protection, using a differential scanning calorimeter (DSC 3, Mettler Toledo), thermal analysis experiments were performed with a temperature accuracy of ± 0.1 K, in an attempt to determine the melting temperature (Tm) and enthalpy of fusion (ΔfusHm) of sodium theophylline. The DSC apparatus was calibrated using melting temperature standards and enthalpy of fusion standards before taking measurements. The standards were indium: ΔfusHm = 3266.58 J mol−1 and Tm = 429.75 K, and zinc: ΔfusHm = 7028.36 J mol−1 and Tm = 692.65 K. In this analysis, the nitrogen purge rate was 25 mL·min−1, the experimental temperature increased from 298.15 to 723.15 K, and the heating rate was 4 K·min−1. In addition, the weight of the sample was in the 5–10 mg range.
The value of ΔfusSm can be obtained from equation (17) 34

where Tm is the melting temperature (K), ΔfusHm is the enthalpy of fusion (J mol−1), and ΔfusSm is the entropy of fusion (J mol−1 K−1).
Reliability verification of the experimental device
To verify the reliability of the solubility data measured by the device, the solubility of potassium chloride in water was measured, and the data values were compared with the literature values. 35 As shown in Figure 10 and Table 6, the ARD between the experimental value and the literature value is 0.106%, and the maximum RD is 0.21%. This shows that the device is reliable and can be used to determine the solubility of sodium theophylline.

Comparison of the experimental and literature data 35 of KCl solubility in water.
Reliability experimental data and RD.

Supplemental Material
sj-docx-1-chl-10.1177_17475198231166563 – Supplemental material for Solubility and thermodynamic properties of sodium theophylline salt in aqueous sodium hydroxide solution at different temperatures
Supplemental material, sj-docx-1-chl-10.1177_17475198231166563 for Solubility and thermodynamic properties of sodium theophylline salt in aqueous sodium hydroxide solution at different temperatures by Zhenhuan Dong, Lingqi Kong, Zhen Liu and Anming Yang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from the National Natural Science Foundation of China (Grant No. 21775081) and Shandong Province Natural Science Foundation (Grant No. ZR2020MB145) is gratefully acknowledged.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.






