Abstract
A ligand-promoted, palladium-catalyzed transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor is developed. The chemical selectivity control is achieved based on ligand regulation. The use of triethanolamine and tetrahydrofuran at 80 °C under N2 is found to be critical for the transfer hydrogenation of terminal alkynes. The general applicability of this procedure is highlighted by the synthesis of 27 terminal alkenes in moderate to good yields.

Introduction
Alkenes are a useful and important structural motif in many organic molecules including pharmaceuticals, natural products, and fragrances, such as combretastatin-A4, pinosylvin, resveratrol, and eugenol.1–3 Hence, novel methods for the synthesis of alkenes are very desirable. The semi-hydrogenation of alkynes to alkenes is one of the most important reactions in organic synthesis, and it has been extremely used for the synthesis of pharmaceuticals, natural products, agrochemical products, and industrial materials.4–7 Several alternative catalytic systems have been reported for the transformation of alkynes into alkenes, for example, Lindlar’s catalyst. 8 Among all the reported transformation systems, metal-catalyzed transfer hydrogenation has attracted attention.9–11 However, to achieve the transfer hydrogenation reaction and to obtain good product yields, these methods often require the use of explosive, flammable, expensive, or corrosive hydrogenating agents, such as ammonia borane, hydrogen, triethylsilane, and formic acid.12–18 Therefore, it is necessary to find an economical and safe hydrogen donor for the semi-hydrogenation of alkynes to alkenes. In recent years, ethanol has attracted attention as a safer and more economical hydrogen donor, and more reactions have been reported for the semi-hydrogenation of alkynes to alkenes using ethanol as the hydrogen source.19–24 However, the semi-hydrogenation of terminal alkynes with ethanol as the hydrogen donor is rarely reported. Very recently, Wang et al. 25 reported the iridium-catalyzed transfer hydrogenation of terminal alkynes with ethanol. Herein, we report a ligand-promoted, palladium-catalyzed semi-hydrogenation of terminal alkynes with ethanol as the hydrogen donor (Scheme 1).
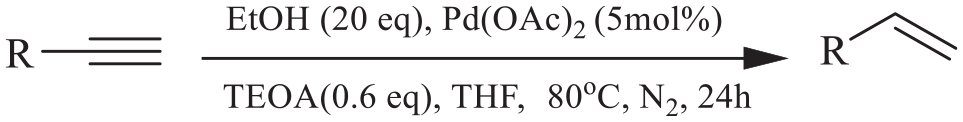
Palladium-catalyzed semi-hydrogenation of terminal alkynes with ethanol.
Results and discussion
We commenced our studies by treating 1-chloro-4-ethynylbenzene and ethanol with 1,2-bis(diphenylphosphi-no)ethane (DPPE) as the ligand. First, various catalysts were screened to identify a suitable candidate. The use of FeI2, CuI, MnCl2, BrMn(CO)5, and Co(OAc)2 gave no catalytic activity to the reaction. However, to our delight, Pd(OAc)2 gave good catalytic activity for the semi-hydrogenation of terminal alkynes to alkenes in the presence of ethanol (Table 1, entries 1–7). To improve the reaction efficiency, we further screened different ligands to identify a suitable ligand (Table 1, entries 8–12). GC analysis showed that 79% yield of the desired alkene was obtained when triethanolamine (TEOA) was used, and a 10% yield of the corresponding alkane was also obtained, which we speculate may be related to the reaction temperature being too high. Next, we screened various reaction temperatures (Table 1, entries 13–18), and 80 °C was found to be the optimum temperature, furnishing the corresponding product in 89% yield (Table 1, entry 16). Furthermore, we screened different reaction solvents (Table 1, entries 19–21), with tetrahydrofuran (THF) proving to be the best.
Optimization of the reaction conditions.

Yields were determined by GC analysis.
DIPAMP = ethylenebis(2-methoxyphenylphenylphosphine).
TEOA = triethanolamine (0.6 eq).
With the optimized reaction conditions in hand, a series of terminal alkynes was investigated to examine the substrate scope of the semi-hydrogenation reaction. As summarized in Scheme 2, this ligand-promoted, palladium-catalyzed transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor exhibited good functional group tolerance to give products 2a–x. Arylethynes with electron-withdrawing groups such as chloro, fluoro, nitro, cyano, and trifluoromethyl on the aryl ring, all gave the corresponding terminal alkenes in good yields (2a–k). In addition, the reaction of an arylacetylene containing two trifluoromethyl substituents at the meta positions of the aromatic ring gave the corresponding terminal alkene(2l) in good yield. Arylacetylenes with electron-neutral or electron-donating groups on the aryl ring, such as alky, methoxy, pyridyl, naphthyl, phenyl, and anthracyl gave the corresponding arylethylenes (2m–w, 2y–z, 2aa) in moderate to good yields. To our delight, the unactivated terminal alkyne(1x) also show good compatibility.

Palladium-catalyzed transfer hydrogenation of terminal alkynes.a
To check the synthetic utility of the current method, it was tested by performing a gram-scale (1g) palladium-catalyzed semi-hydrogenation of a terminal alkyne with ethanol under the optimized conditions. In this case, the target 2aa was obtained in 51% yield.
On the basis of previous work on the semi-hydrogenation of terminal alkynes with ethanol as the hydrogen donor,
25
we propose a possible catalytic cycle reaction mechanism (Scheme 3). First, TEOA coordinates with Pd(Ⅱ) to form active catalytic species

A possible reaction mechanism.
Conclusion
In conclusion, palladium catalysts have good catalytic activity and selectivity, and have important application value in petrochemicals, pharmaceuticals, fine chemicals, organic synthesis, and other fields. We have developed a ligand-promoted palladium catalysts transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor. This process is safer and more convenient than traditional strategies, and is compatible with a range of functional groups. Further investigations of the use of cheaper metals, green hydrogenation agents, and other reaction types are underway in our laboratory.
Experimental section
General information
Unless stated otherwise, all reactions were carried out in pressure tubes under N2. Commercially available reagents were used as received. Non-commercially available substrates were synthesized following reported protocols. Thin-layer chromatography performed on brand and samples were visualized by UV fluorescence. Silica gel (particle size 40–63 µm) was used for flash column chromatography. Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AV 400 spectrometer at 400 MHz (1H NMR) and 100 MHz (13C NMR). Proton and carbon chemical shifts are reported relative to the solvent used as an internal reference.
Typical procedure for the synthesis of terminal ethylenes
To a 15 mL pressure tube were added 1-chloro-4-ethynylbenzene
Gram-scale synthesis
According to the typical procedure: To a 250 mL pressure tube was charged with added 9-ethynylanthracene
1-chloro-4-vinylbenzene (
1-chloro-3-vinylbenzene (
1-chloro-2-vinylbenzene (
1-fluoro-4-vinylbenzene (
1-fluoro-3-vinylbenzene (
1-fluoro-2-vinylbenzene (
1-nitro-4-vinylbenzene (
1-nitro-3-vinylbenzene (
4-vinylbenzonitrile (
1-(trifluoromethyl)-4-vinylbenzene (
1-(trifluoromethyl)-3-vinylbenzene (
1,3-Bis(trifluoromethyl)-5-vinylbenzene (
styrene (
1-methyl-4-vinylbenzene (
1-methyl-3-vinylbenzene (
1-methyl-2-vinylbenzene (
1-tert-butyl-4-vinylbenzene (
1-methoxy-2-vinylbenzene (
1-methoxy-3-vinylbenzene (
1-methoxy-4-vinylbenzene (
3-vinylpyridine (
2-vinylnaphthalene (
4-vinyl-1,1'-biphenyl (
Allylbenzene (
2-vinylpyridine (
3-vinyl-1,1'-biphenyl (
9-vinylanthracene (
Supplemental Material
sj-rar-1-chl-10.1177_17475198221145838 – Supplemental material for Palladium-catalyzed transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor
Supplemental material, sj-rar-1-chl-10.1177_17475198221145838 for Palladium-catalyzed transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor by Jie Hui and Feng Wang in Journal of Chemical Research
Supplemental Material
sj-rar-2-chl-10.1177_17475198221145838 – Supplemental material for Palladium-catalyzed transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor
Supplemental material, sj-rar-2-chl-10.1177_17475198221145838 for Palladium-catalyzed transfer hydrogenation of terminal alkynes using ethanol as the hydrogen donor by Jie Hui and Feng Wang in Journal of Chemical Research
Footnotes
Acknowledgements
I would like to express my gratitude to my family and friends who helped me during the writing of this thesis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by project of Jiangsu United Vocational and Technical College (NO. B/2022/11/147).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

