Abstract
A label- and enzyme-free fluorescence assay based on thioflavin T–induced G-quadruplexes is developed to sensitively and specifically detect telomerase activity. Thioflavin T has a dual role as an efficient inducer and fluorescent probe, and the incorporation of thioflavin T into the thioflavin T–induced G-quadruplexes results in an intense fluorescence enhancement. In the presence of thioflavin T and K+, G-quadruplexes are formed by elongation of the telomerase substrate primer that is catalyzed by telomerase extracted from cancer cells. Thus, the telomerase activity in cancer cell extracts can be evaluated by measuring the thioflavin T fluorescence. More importantly, thioflavin T can specifically recognize and bind to G-quadruplexes, whereas it cannot recognize single- and double-stranded DNAs, which leads to the thioflavin T–based fluorescence assay exhibiting a reduced background and improved signal-to-noise ratio. As a result, the proposed assay has the linear range from 5 to 200 HeLa cells and the detection limit is 34 HeLa cells, which holds great potential for use in the detection of telomerase activity and the diagnosis of cancer.

Introduction
Human telomerase is a ribonucleoprotein complex composed of a human telomerase reverse transcriptase (hTERT) and a human telomerase RNA (hTR) template component,1,2 which can bind to the telomere region via the hTR template and catalyze extension of the telomere with a repeat sequence (TTAGGG)n at the end of eukaryotic chromosomes to protect the end of the chromosome from degradation, rearrangements, and end–end fusion.3–5 It is well known that telomerase acts as a promising biomarker for early cancer diagnosis and is a therapeutic target since the telomerase activity is unregulated in more than 85%–90% of human cancer cells compared with normal human cells. 6 Thus, the sensitive and specific detection of telomerase activity is of great significance in biological studies, clinical diagnostics, and treatment.
Since Piatyszek et al. 7 developed the polymerase chain reaction (PCR)-based telomerase repeat amplification protocol (TRAP) to detect telomerase activity in 1995, many PCR-free approaches have been designed for detection of telomerase activity to overcome the limits of expensive equipment and time-consuming PCR-based methods. Among them, methods of enzyme-assisted isothermal amplification and fluorescence-based detection have attracted attention due to their superior sensitivity and specificity.8–12 However, enzyme-assisted methods are usually susceptible to the reaction conditions, such as temperature and pH, which are crucial to the optimum activity of the enzyme.13,14 Besides, the design and manipulation of enzyme-based amplification detection methods are relatively complicated. As for the fluorescence biosensors to detect telomerase activity, many DNA-based biosensors have been developed by using fluorescence-labeled probes, which may result in increased cost and operation.15–17 Thus, there is still an urgent need for the development of a PCR-free, label-free, and enzyme-free method to detect telomerase activity with good sensitivity and specificity.
Thioflavin T [4-(3,6-dimethyl-1,3-benzothiazol-3-ium-2-yl)-N,N-dimethylaniline] (ThT) is a water-soluble fluorescence dye that has been widely used for the identification of amyloid fibrils. Recently, ThT has been demonstrated to induce G-rich oligonucleotide sequences to fold into G-quadruplexes, accordingly, binding of ThT to G-quadruplexes results in a strong fluorescence enhancement. 18 More importantly, ThT can specifically recognize and bind to G-quadruplexes while it cannot recognize single- and double-stranded DNAs, leading to a reduced background and an improved signal-to-noise ratio (SNR) of the ThT-based fluorescence sensor. 10 By utilizing this “light-up” characteristic, ThT has been used as an efficient fluorescence sensor to recognize DNA and RNA G-quadruplexes.19,20 In recent years, Mohanty et al. 19 have developed a selective fluorescent sensor for the human telomeric G-quadruplex DNA based on ThT, and the fluorescence “light-up” of ThT on binding to the telomeric G-quadruplex was shown to be a 250-fold enhancement compared with other single- and double-stranded DNA forms. Besides, Liu et al. 21 have utilized ThT as a novel fluorophore to target the G-quadruplex, and a molecular rotor mechanism was proposed in which the fluorescence enhancement was induced by restriction of the rotation of the benzothiazole (BZT) and dimethylaminobenzene (DMAB) rings in the ThT excited state upon the binding of ThT to the G-quadruplex. As a consequence, and inspired by the excellent fluorescence properties of ThT toward G-quadruplexes, we have designed a label- and enzyme-free fluorescence strategy based on ThT-induced G-quadruplexes for the direct detection of telomerase activity in cancer cell extracts.
It was hypothesized that telomerase elongation products (TEP) could be induced to form G-quadruplexes in the presence of telomerase (cancer cell extracts), a TS primer and ThT. Subsequent incorporation of ThT into the ThT-induced G-quadruplexes could result in an intense fluorescence enhancement. Consequently, the telomerase activity can be evaluated by measuring the ThT fluorescence, where ThT has a dual role as an efficient inducer and fluorescent probe. We anticipated that the proposed label- and enzyme-free fluorescence strategy would provide a new perspective for designing an efficient fluorescence sensor for assaying the telomerase activity of cancer cell extracts.
Results and discussion
Principle of the telomerase activity assay
In this study, we report a label- and enzyme-free fluorescence assay that employs ThT as a fluorescence probe for the detection of the telomerase activity of cell extracts, and the working principle of the proposed fluorescence strategy is depicted in Scheme 1. In the presence of telomerase extracted from cancer cells, the TS primer, and a nucleotide mixture of dNTPs, TEP were generated by telomerase-catalyzed elongation of the TS primer. The resulting TEP existed as a longer single-stranded DNA molecule with tandem repeat units (5′-TTAGGG-3′)n synthesized on the 3′ end. It is well known that G-quadruplexes can be formed by Hoogsteen hydrogen bonding between G-quartets and are stabilized by the presence of K+. 22 Besides, it was demonstrated that ThT can induce G-rich oligonucleotide sequences to fold into G-quadruplexes. 18 Thus, with the addition of ThT solution containing K+, G-quadruplexes could be formed since there was a high-level guanine content in the TEP derived from the repeat units (5′-TTAGGG-3′)n. The telomerase activity was then detected by measuring the ThT fluorescence intensity based on the incorporation of ThT into the ThT-induced G-quadruplexes. The number of G-quadruplexes increased with elongation of the TEP, which resulted in a sharp increase in fluorescence intensity. However, in the absence of telomerase extract, the TS primer could not be elongated and there was no formation of G-quadruplexes, which resulted in no change in the fluorescence intensity upon the addition of ThT and K+ since the specific characteristic of ThT that cannot bind to single-stranded DNA (TS primer). Therefore, the proposed label-free, enzyme-free, and ThT-based fluorescence assay can greatly simplify the operation process for the detection of telomere activity.

Working principle of the fluorescence strategy for the detection of telomerase activity based on the incorporation of ThT into ThT-induced G-quadruplexes.
Feasibility of the proposed fluorescence assay
We first used synthetic telomerase extension products (STEP) (the detailed sequences are listed in Supplemental Table S1) with several TTAGGG repeats to investigate the feasibility of the proposed ThT-based fluorescence assay. As reported, STEP with four TTAGGG repeats could form one G-quadruplex (abbreviated as STEP1). 23 As shown in Figure 1(a), free ThT showed a weak fluorescence (black curve) in K+-containing Tris-HCl buffer. In contrast, ThT exhibited a strong fluorescence upon the addition of STEP1 (red curve), while a similar negligible fluorescence to free ThT was observed by ThT incubated with TS primer (blue curve). The results indicated that STEP1 folded into the G-quadruplex after the induction of ThT, and subsequently formed a ThT/G-quadruplex complex, which resulted in a significantly enhanced fluorescence. Subsequently, we used STEP that included 8 and 16 telomeric repeats to generate 2 and 4 G-quadruplexes (abbreviated as STEP2 and STEP3, respectively) for further study. We found that as the number of ThT-induced G-quadruplexes in the STEP increased, the resulting fluorescence of the ThT enhanced gradually (Supplemental Figure S1), which indicated the specific binding of ThT to the ThT-induced G-quadruplexes.

Fluorescence spectra (a) and absorption spectra (b) of 50 mM Tris-HCl (pH = 7.2, 63 mM K+) containing 1 μM ThT (black curve), 1 μM ThT with 1 μM TS primer (blue curve), and 1 μM ThT with 1 μM STEP1 (red curve).
In addition, interaction of the ThT with the G-quadruplex structure was further verified by the absorption spectra. From Figure 1(b), free ThT showed its characteristic absorption profile with an absorption maximum at about 412 nm. There was no absorption spectral change for ThT with the addition of TS primer; however, the addition of STEP1 to ThT induced a red shift from 412 nm to 422 nm, which further confirmed the specific interaction of ThT with the G-quadruplex structure in STEP1, and similar results had also been demonstrated previously.18,20
To directly demonstrate the feasibility of the proposed ThT-based fluorescence assay, we further used telomerase extract from cancer cells to catalyze the telomerization process of the TS primer. In this study, human cervical carcinoma cells (HeLa, telomerase-positive) were used as the model cancer cells, 24 where the cell extracts from HeLa cells were used as the source of telomerase. As shown in Figure 2(a), the ThT showed a weak fluorescence in both the absence and presence of the TS primer, whereas there was an intense fluorescence enhancement when ThT was incubated with the TS primer and 1000 HeLa cell extracts. In order to demonstrate the variation of the ThT fluorescence response to the telomerase activity, ThT was then incubated with the TS primer and inactive telomerase from heated HeLa cells, and as a result, the fluorescence intensity was similar to that of free ThT. In addition, the ThT fluorescence intensity in the presence of the TS primer and HeLa cell extracts at 490 nm was found to be almost as large as 20 times the fluorescence enhancement of free ThT (Figure 2(b)). These results indicated the specific binding interaction of ThT with the ThT-induced G-quadruplex, and the change of ThT fluorescence was attributed to the telomerase activity from HeLa cells, which suggests that the proposed method for the detection of telomerase activity was feasible.

Fluorescence emission spectra (a) and corresponding fluorescence intensity at 490 nm (b) of telomerase extension reaction buffer containing 1 μM ThT, 1 μM ThT with 1 μM TS primer, 1 μM ThT with 1 μM TS primer and 1000 inactive HeLa cell extracts, and 1 μM ThT with 1 μM TS primer and 1000 HeLa cell extracts. Error bars indicate the standard deviation of measurements (n = 3).
Optimization of the assay conditions
In order to obtain a high sensitivity for the proposed assay, several detection parameters, including the telomerase extension reaction buffer and the extension time, were optimized. Despite the fact that ThT can induce the formation of a G-quadruplex, cations play a key role in stabilizing the formation of the G-quadruplex structure. It had been reported that the G-quadruplex was more stable in the presence of K+ than that formed in the presence of Na+. 25 Therefore, we studied the ThT fluorescence change after the incubation of ThT with the TS primer and telomerase extract in telomerase extension reaction buffer in the presence of different concentrations of K+. As shown in Supplemental Figures S2A and S2B, the ThT fluorescence intensity increased on increasing the concentration of K+ due to the fact that formation of the G-quadruplex structure was induced and stabilized by potassium, while ThT showed a relatively weak fluorescence emission in the absence of K+. The results indicated the stable conformation of the G-quadruplex was formed at a higher concentration of potassium (more than 50 mM), which was consistent with a previous study. 26 Hence, the telomerase extension reaction buffer with 63 mM K+ was selected for the proposed assay according to another previous study. 27
In addition, the extension time of the telomerase extension reaction had a significant influence on the length of the TEP, which would affect the number of folded G-quadruplexes and the enhancement of the ThT fluorescence intensity. In Supplemental Figure S3, the ThT fluorescence increased on increasing the extension time for the telomerase extension reaction, with the fluorescence reaching a maximum at 60 min and remained stable. Thus, 60 min was selected as the optimal telomerase extension time.
Sensitivity of the fluorescence assay
To examine the sensitivity of the developed fluorescence assay, a series of telomerase extracts from different numbers of HeLa cells were incubated with TS primer, dNTPs, and ThT under the optimized detection conditions. As shown in Figure 3(a), as the number of HeLa cells increased in the range of 5–1000 cells, the ThT fluorescence was enhanced, which is consistent with the higher content of telomerase in the cell extracts. Moreover, it was found that the ThT fluorescence intensity at 490 nm had a good linear correlation to the number of HeLa cells in the range of 5–200 cells with a correlation coefficient of 0.9958 (Figure 3(b)), and a limit of detection (LOD) of 34 HeLa cells was determined according to 3σ/K (σ is the standard deviation and K is the slope of the curve of linear regression). Compared with some recent telomerase activity detection approaches (Supplemental Table S2), the proposed ThT-based method shows good sensitivity, which derives from special property of the ThT fluorophore.

(a) Fluorescence spectra for detecting telomerase activity in telomerase extracts equivalent to 5, 20, 50, 100, 200, 500, and 1000 HeLa cells, respectively. (b) The relationship between the fluorescence intensity at 490 nm and different number of HeLa cells in the range of 5−1000 cells. Inset: the linear plot of the fluorescence intensity versus the number of the HeLa cells (5–200 cells). F is the resulting ThT fluorescence response to the corresponding HeLa cells, and F0 is the fluorescence of the ThT without the addition of the HeLa cell extract. [ThT] = 0.5 µM. Error bars indicate the standard deviation of measurements (n = 3).
Although the proposed method showed enough sensitivity, there is still great potential to improve the detection sensitivity. It is well known that the enhancement of SNR is crucial to improve the detection sensitivity. Therefore, optimization of the SNR by decreasing the background fluorescence signal with improved fluorescence quenching, and simultaneously increasing the response fluorescence after binding to the target can effectively enhance the detection sensitivity. Recently, graphene oxide (GO) has been widely used for the design of biosensors due to its distinctive physicochemical properties, such as large surface area, small size, amphiphilicity, and fluorescence-quenching ability. 28 More importantly, G-quadruplexes and i-motif structures are demonstrated to have weak affinity for GO due to the nucleobases in the structures being efficiently shielded within the negatively charged phosphate backbone, and the binding affinity difference between GO and diverse DNA structures has been utilized for DNA-based biosensors.29,30 Considering the ability of GO to sensitively discriminate between single-stranded DNA and G-quadruplexes, as well as its super and universal fluorescence-quenching ability, it can be utilized as a ThT quencher in this study to further decrease the background fluorescence and to increase the SNR and sensitivity.
Generality and selectivity of the fluorescence assay
To demonstrate the generality and selectivity of this assay, the telomerase activity in human normal liver cells (L02) and in other cancer cell lines (MCF-7 (human breast cancer cell) and A549 (human lung carcinoma cell)) was tested, where HeLa cells were used as a positive control and heated HeLa cells were used as a negative control. As shown in Figure 4, the fluorescence intensity in HeLa, A549, and MCF-7 cells was stronger than that in normal L02 cells and heated HeLa cells. The results confirm that all of the cancer cell lines showed positive telomerase activity, while the normal cells had a low level of telomerase activity, which has also been confirmed in other reports.31,32 Besides, it was also found that the telomerase activity of HeLa and A549 cells was higher than that of MCF-7 cells, which was attributed to a relatively low expression of telomerase activity in MCF-7 cells. 33 All of the results demonstrate the generality and selectivity of the proposed assay for the detection of telomerase activity, which implies the proposed assay has great potential to detect telomerase activity among different cell lines.
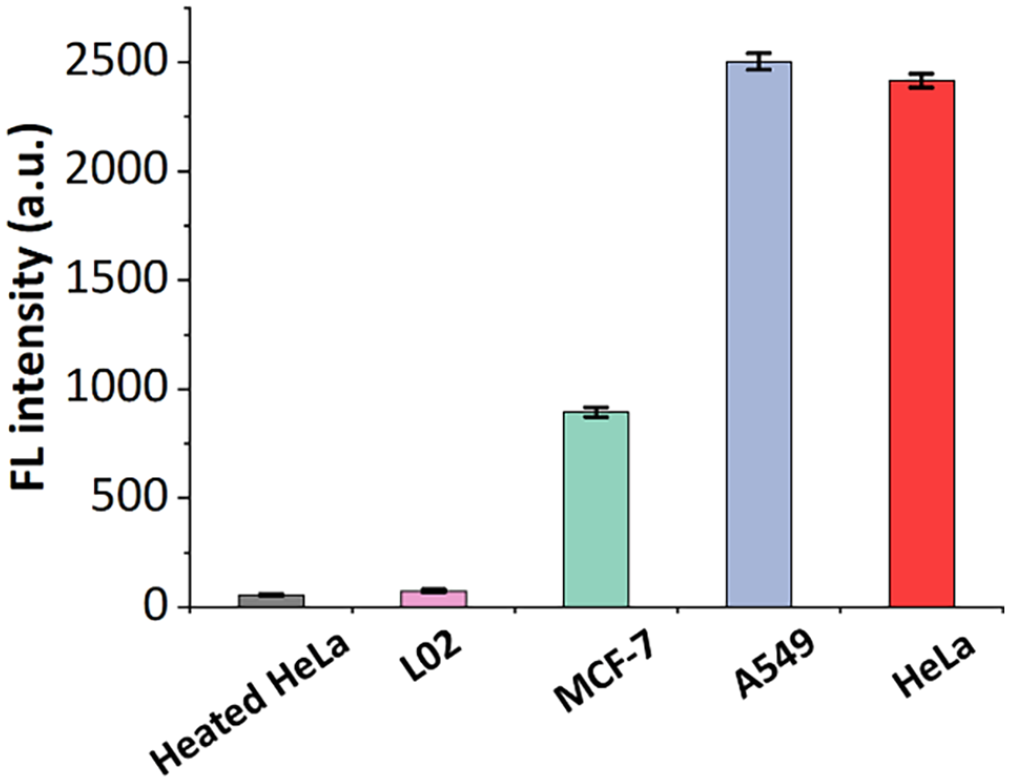
The detection of telomerase activity in different cell lines including normal L02 cells, HeLa cells, MCF-7 cells, and A549 cells. Heated HeLa cells acted as a negative control (telomerase activity of cell extracts equivalent to 1000 cells). [ThT] = 1 µM. Error bars indicate the standard deviation of measurements (n = 3).
Conclusions
In summary, we have developed a label- and enzyme-free fluorescence assay by using ThT as a DNA G-quadruplex fluorescent inducer. The proposed fluorescence assay was based on the intercalation of ThT into the ThT-induced G-quadruplexes, which were formed by elongation of the TS primer catalyzed by telomerase extracted from cancer cells, and an LOD of 34 HeLa cells was determined. Furthermore, the proposed assay was PCR-free and simple in operation, showing great potential for the highly sensitive and selective detection of various biomolecules.
Experimental section
Materials
All the DNA sequences (including the TS primer and STEP) were purified by high-performance liquid chromatography (HPLC) and obtained from Sangon Biotech Co., Ltd (Shanghai, China). The oligonucleotide sequences are listed in Supplemental Table S1. ThT, 3-[(3-cholamidopropyl)dimethylamino]-1-propanesulfonic acid (CHAPS), β-mercaptoethanol, glycerol, phenyl-methane sulfonyl fluoride (PMSF), ethylene glycol bis(2-aminoethyl ether)-N,N,N′,N′-tetraacetic acid (EGTA), Tween 20, and tris-(hydroxymethyl)aminomethane (Tris) were obtained from Aladdin Co., Ltd (Shanghai, China). Deoxyribonucleoside 5′-triphosphate mixture (dNTPs) was purchased from Tiangen Biotech Co., Ltd (Beijing, China). All cells were purchased from the Cell Center of the Institute of Basic Medical Sciences, Chinese Academy of Medical Science. Solvents of analytical grade were employed without additional purification.
Measurements of fluorescence emission and UV-Vis absorbance based on the STEP
ThT and STEP were mixed in 50 mM Tris-HCl (pH = 7.2, 63 mM K+), and the final concentration of each sample was 1 μM. After incubation of 30 min, the fluorescence emission spectra of the mixture was measured with a Fluoromax-4 spectrofluorometer (HORIBA, Irvine, CA, USA) and a quartz fluorescence cuvette (2 mm × 10 mm), with the following settings: λex = 425 nm, λem = 490 nm. For UV-Vis absorbance spectra, the mixture was measured using a UV-1800 spectrophotometer (SHIMADZU, Japan) and a quartz cell with a 1 cm optical pathway.
Cell cultures
Human cervical cancer cells (HeLa), human lung cancer cells (A549), human breast cancer cells (MCF-7), and normal human liver cells (L02) were cultured in RPMI-1640 medium containing 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin in a humidity incubator (5% CO2, 37 °C).
Telomerase extract preparation
Telomerase extracts were prepared according to previous procedures. 10 Cells were digested by trypsin and re-dispensed in a 1.5 mL RNase-free EP tube. The cells were then washed 3 times with cooled 1× PBS (phosphate-buffered saline) buffer by centrifugation at 1800 r/min for 5 min. Subsequently, the cells were resuspended in 200 μL of ice cold 1× CHAPS lysis buffer (0.5% CHAPS, 10 mM Tris-HCl pH 7.5, 1 mM MgCl2, 1 mM EGTA, 5 mM β-mercaptoethanol, 0.1 mM PMSF, 10% glycerol) and incubated for 30 min on ice, and then centrifuged at 15,000 r/min for 30 min at 4 °C. Finally, the supernatant was collected and stored at –80 °C for further use.
Telomerase extension reaction
First, the telomerase extract was diluted in 1× CHAPS lysis buffer with the required number of cells. Then, 10 μL of telomerase extract was added to 100 μL of the telomerase extension reaction buffer (20 mM Tris-HCl pH 8.3, 1.5 mM MgCl2, 1 mM EGTA, 63 mM KCl, 0.05% Tween 20, 1 mM dNTPs, and 200 nM TS primer) and then incubated at 37 °C for 2 h. For the inactive telomerase activity, the telomerase extract was incubated at 95 °C for 10 min.
Fluorescence assay for the detection of telomerase activity
After the telomerization reaction, 100 µL of ThT (the final concentration of ThT was 1 μM) was mixed with a solution of the telomerase extension reaction products. After incubation for 30 min, the fluorescence intensity of the solution was detected using an Agilent Cary Eclipse Fluorescence Spectrophotometer (Agilent Co., Santa Clara, CA, USA) and a quartz fluorescence cuvette (the emission spectra were recorded in the wavelength of 460–600 nm upon excitation at 425 nm).
For the detection of the sensitivity of the fluorescence assay, the telomerase extract from HeLa cells was diluted in lysis buffer with the respective numbers of cells (from 5 to 1000 cells). Then, after the telomerase extension reaction and the incubation of ThT as described above, all ThT fluorescence spectra were recorded using a Tecan plate reader (Tecan, Switzerland) with a Costar black bottom 96-well plate (425 nm for excitation and 460–600 nm for emission).
As for the generality and selectivity of the fluorescence assay, telomerase extract from human normal liver cells (L02), human breast cancer cells (MCF-7), human lung carcinoma cells (A549), HeLa cells, and the control group with heat-inactivated HeLa cells extracts were used. All fluorescence spectra were recorded using the Tecan plate reader with the same detection parameters as described above.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221139085 – Supplemental material for A label- and enzyme-free fluorescence assay based on thioflavin T–induced G-quadruplexes for the detection of telomerase activity
Supplemental material, sj-docx-1-chl-10.1177_17475198221139085 for A label- and enzyme-free fluorescence assay based on thioflavin T–induced G-quadruplexes for the detection of telomerase activity by Zhe Chen and Yunxia Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support in the form of grants from the Fundamental Research Program of Shanxi Province (No. 20210302124181), the Fund Program for the Scientific Activities of Selected Returned Overseas Professionals in Shanxi Province (No. 20220021), the Startup Foundation for Doctors of Shanxi Medical University (No. XD2037), and the Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi (No. 2021L239).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
