Abstract
A series of heterogeneous catalysts with Cr as an active component is prepared by an innovative in situ coprecipitation method. The prepared Cr/SiO2 catalyst has high catalytic activity, selectivity, and stability in the conversion of cyclohexane into ketone-alcohol oil using H2O2 as the oxidant under mild conditions, resulting in 54.59% cyclohexane conversion and 90.01% ketone-alcohol oil selectivity. The physical and chemical properties of the prepared materials are studied by X-ray diffraction, Fourier transform infrared, X-ray photoelectron spectroscopy, scanning electron microscopy, transmission electron microscopy, and nitrogen adsorption-desorption analysis (BET). The excellent performance of the Cr/SiO2-400 catalyst is due to the increase of low-valence chromium species and the intimate interaction between SiO2 and CrOx. In addition, the catalyst was easy to separate and maintained good catalytic activity even after four runs.

Introduction
The selective oxidation of cyclohexane to cyclohexanol and cyclohexanone (the mixture is called “KA oil,” where “K” and “A” represent the ketone and the alcohol, respectively) has important academic value and application prospects, with the main investigation being centered on the sp 3 activation of the C–H bond.1–4 Currently, KA oil is mainly prepared by a noncatalytic process, which suffers from a low conversion rate, poor selectivity, high energy consumption, high environmental treatment costs, and so on. 5 Therefore, the development of a mild and efficient catalytic oxidation system for this process is highly promising. Cobalt or manganese salts are widely used as homogeneous catalysts in industry for the selective oxidation of cyclohexane. However, the oxidation process often requires a high temperature and raised air/O2 pressure.6,7 Moreover, although porphyrin and other metallic complexes can be used for the selective oxidation of cyclohexane at moderate temperatures and pressures, they require complicated preparation procedures and are difficult to be industrialized.8–10 Besides, these homogeneous catalyst systems inevitably encounter the problems of catalyst recovery and product separation. To overcome these limitations, it is essential to develop heterogeneous catalysts with good stability from the perspective of green chemistry.
So far, numerous efforts have been made to explore heterogeneous catalysts for the aerobic oxidation of cyclohexane using O2 or air as oxidants, such as Mg-Cu/SBA-15, 11 AgPd/MgO, 12 CoFe2O4, 13 FeMn/Al2O3, 14 Co-Co3O4/CN, 15 and MoO3-AgPd. 16 On one hand, O2 is a non-polluting and inexpensive oxidant, although the reaction often requires a high temperature and a high pressure to achieve activation. On the other hand, the oxidation processes are relatively complex, and the conversion of cyclohexane is low. In addition, other reports using tert-butyl hydrogen peroxide, and H2O2 as oxidants for the liquid-phase oxidation of cyclohexane have been described.17–20 In particular, H2O2, a green oxidant since it produces H2O as the sole byproduct, has received considerable attention in the field of catalytic oxidation. Consequently, several heterogeneous catalysts with H2O2 as the oxidant have been applied to the selective oxidation of cyclohexane, including Fe/H-MSN, 21 CuNi/Al2O3, 22 PdX/TiO2, 23 VPO-TUD-1, 24 CuFeAl2O4, 25 Cu-Cr2O3, 26 CuCr2O4, 27 Cu/ZSM-5, 28 and VPO/KIT-6. 29 Given that some heterogeneous catalysts exhibit problems with poor selectivity and thermal stability, it is necessary to develop highly active and robust catalysts for the conversion of cyclohexane into KA oil.
Furthermore, it is significant for heterogeneous catalysts to select a suitable carrier. SiO2 materials exhibit ordered porosity, high specific surface areas, and good chemical stability among various catalyst carriers.30,31 These properties ensure the excellent mass transfer performance of SiO2-based materials, which is closely related to catalytic activity. At present, Cr catalysts have been widely used in the chemical industry. 32 The sol-gel method is still the most common route to obtain SiO2 carriers33,34; therefore, single-step or in situ methods need to be further developed for the synthesis of supported SiO2-based catalysts. Based on the reasons mentioned above, it is highly desirable to develop experimentally an efficient heterogeneous catalytic system for the conversion of cyclohexane into KA oil under mild conditions using a sustainable method.
In this context, Cr/SiO2 was successfully prepared by an in situ homogeneous coprecipitation method. This method, which has been not mentioned in previous papers, involves the direct doping of Cr components into SiO2 by a one-step process. In subsequent studies, the catalytic properties of the prepared samples were evaluated in the conversion of cyclohexane into KA oil by using H2O2 as the oxidant. By investigating the influences of the calcination temperature on the catalyst activity, the Cr/SiO2-400 sample obtained at a lower calcination temperature showed better catalytic performance. Next, different characterization methods were used to further explore the correlation between the catalytic structure and reaction performance. As determined, the excellent performance of the catalyst Cr/SiO2-400 was due to an increase in the low-valence chromium species and the intimate interactions between SiO2 and CrOx. Overall, the developed heterogeneous catalyst can potentially serve as a practical catalyst for the selective oxidation of cyclohexane into KA oil.
Experimental sections
Materials
Cyclohexane, cyclohexanol, cyclohexanone, acetonitrile, hydrogen peroxide (30% wt%), Cr(NO3)3·9H2O, TEOS (tetraethyl orthosilicate), CTAB (hexadecyl trimethyl ammonium bromide), and NaOH were all of analytical reagent grade (AR) and were supplied by Shanghai Maclin Biochemistry Technology Co. Ltd., China. Ethanol was purchased from Sinopharm Chemical Reagent Co. Ltd., China. All the chemicals were used as received, without further purification.
Catalyst preparation
The Cr/SiO2 catalyst was prepared by an in situ homogeneous coprecipitation method. Briefly, CTAB (1.82 g, 5 mmol), TEOS (4.17 g, 20 mmol), and Cr(NO3)3·9H2O (0.8 g, 2 mmol) were added to a mixture of H2O (45 mL) and ethanol (10 mL), followed by stirring the mixture for 2 h to dissolve all the feedstocks. Subsequently, 10 wt% NaOH (5 mL) solution was added to the above mixture, followed by stirring for 2 h and then standing for 12 h. The resulting solid sample was collected by centrifugation and washed sequentially with deionized water and ethanol. After drying at 80 °C under vacuum overnight, the solid sample was transferred to a crucible with a lid and calcined at a given temperature (300, 400, 500, 600, or 700 °C) for 4 h. Finally, the catalyst ground into a fine powder. For convenience, the prepared catalyst is labeled Cr/SiO2-x, where x indicates the calcination temperature (x = 300, 400, 500, 600, or 700 °C). In addition, single SiO2 and Cr oxides were prepared via the same synthetic route (calcined at 400 or 600 °C for 4 h). Furthermore, to compare the effect of the preparative methods, the catalyst denoted as Cr/SiO2* was prepared by a common post-synthesis method. Catalysts with Cr oxide supported on other traditional carriers (e.g. MCM-41, ZSM-5, Al2O3, and HSCN) were prepared and were denoted as Cr/MCM-41, Cr/ZSM-5, Cr/Al2O3, and Cr/HSCN, respectively.
Catalyst characterization
X-ray diffraction (XRD) patterns were collected on a Rigaku D/MAX 2550 diffractometer with CuKα radiation (λ = 1.5418 Å). Fourier transform infrared (FTIR) spectra were recorded on an IRAffinity-1S (Shimadzu) spectrometer using KBr pellets in the range of 400–4000 cm–1. Field emission scanning electron microscopy (FESEM) was performed on a Hitachi SU-8010 microscope, and an energy dispersive X-ray spectrometer (EDS) was applied for elemental distribution mappings. Transmission electron microscopy (TEM) images were recorded on an FEI Talos F200S instrument operating at 200 kV. X-ray photoelectron spectroscopy (XPS) measurements were recorded on a Thermo Scientific K-Alpha spectrometer using AlKα radiation at 1486.6 eV, and all the peaks were calibrated to the C1s peak at 284.8 eV. H2-temperature-programmed reduction (H2-TPR) measurements were carried out on a Micromeritics AutoChem II 2920 instrument with a thermal conductivity detector (TCD). N2 adsorption/desorption isotherms were obtained from a Micromeritics ASAP 2460 analyzer at 77.3 K.
Catalyst activity test
To test the catalytic properties of the prepared materials, the selective oxidation of cyclohexane to KA oil was used as a test reaction. In a typical experiment, cyclohexane (0.168 g, 2 mmol), 30 wt% H2O2 (0.453 g, 4 mmol), and 10 mg of the prepared materials were added into a 25 mL reaction tube and then reacted at 50 °C for 6 h. When the reaction was finished and cooled to the room temperature, the catalyst was separated by centrifugation and the liquid was submitted to gas chromatography (GC) analysis. Specifically, the reaction products were ascertained from the retention times by authentic samples and further identified through gas chromatography–mass spectrometry (GC-MC, Agilent 6890-5973). Gas chromatography (GC, FULI GC9790II) was also used for quantitative analyses of the reactant (cyclohexane) and the principal products (cyclohexanone and cyclohexanol). The experimental data were collected at least three times for each catalyst to ensure accuracy, and the conversion rate of cyclohexane and the selectivity for the two oxidized products were determined by the following formulae:
Result and discussion
Catalyst characterization
Figure 1 shows the results of the XRD characterization of Cr/SiO2: the carrier SiO2 and Cr oxide calcined at 400 and 600 °C. As for SiO2, the broad diffraction peak at 2θ = 22.4° was attributed to the amorphous framework of SiO2. 35 As for the Cr/SiO2 composites, the characteristic diffraction peaks of SiO2 obviously changed when the calcination temperature was increased to 700 °C, and the crystal structure crystallized was mainly quartz. 36 The diffraction peaks of the standard Cr2O3 (PDF #82-1484) were detected in the XRD spectra of Cr-O(600), Cr/SiO2-500, and Cr/SiO2-600, indicating the existence of crystalline Cr2O3 at higher calcination temperatures. However, no crystalline Cr-containing phases were observed in the Cr-O(400), Cr/SiO2-300, and Cr/SiO2-400 samples, indicating that the Cr compound was highly dispersed within the carrier and existed in an amorphous state. The Cr2O3 crystallite structure was also absent in the XRD spectra of Cr/SiO2-700, indicating that the Cr valent state changed when the calcination temperature was increased. Furthermore, it is worth noting that once the calcination temperature reached up to 400 °C, the characteristic peaks of SiO2 shifted appreciably to a smaller angle (see Figure S1 in the supporting information), which was probably due to Cr species embedding in the silica and the intimate interaction between SiO2 and Cr2O3.

X-ray diffraction patterns of the prepared samples.
Figure 2 shows the FTIR spectra of Cr/SiO2 and SiO2. For pure SiO2, a wide characteristic absorption band was observed in the range of 3000–3700 cm–1, corresponding to the stretching vibration of the O–H bond on the surface, while the peak at 1638 cm–1 corresponded to the bending vibration peak of H–O–H due to adsorbed water. 37 As for the broad adsorption bands of SiO2 in the region of 990–1300 cm–1, these could be attributed to the antisymmetric stretching vibration of Si–O–Si which is the basic structural unit of SiO2. 38 The peaks at 798 and 466 cm–1 were assigned to the symmetric stretching and bending vibrations of the Si–O bond. 39 Notably, the absorption peak at 964 cm–1 corresponded to the bending vibration of Si–OH, whereas for Cr-doped catalyst materials, the calcination temperature of less than 500 °C did not appear to be due to the complete contraction of Si–OH to a Si–O–Si bond at a lower calcination temperature. When the calcination temperature did not exceed 500 °C, the FTIR spectra of the Cr/SiO2 catalysts were very similar to that of SiO2, and no significant characteristic absorption peaks associated with Cr metal were observed. However, when the calcination temperature was 500 and 600 °C, respectively, the FTIR spectra presented two characteristic absorption peaks at 565 and 623 cm–1 (both corresponding to contraction of the Cr–O bonds). 40 Compared with pure Cr2O3 and Cr/SiO2, relatively small shifts of the Cr–O bond were observed, indicating that good interactions between the Cr species and the SiO2 carrier had formed. However, the peaks at 565 and 623 cm–1 disappeared when the calcination temperature was increased to 700 °C. Also, a characteristic absorption peak was observed at 885 cm–1, which was assigned to the stretching vibrations of the Cr–O bond in CrO3, 41 indicating a change in the valence state of the Cr species at higher calcination temperatures.

Fourier transform infrared spectra of the prepared samples.
The morphology and microstructure of the Cr/SiO2-400 catalyst were investigated by FESEM and TEM. The obtained results are shown in Figure 3. Figure 3(a), (b), and (d) revealed that the agglomeration of nano-sized particles occurred. Figure 3(b) exhibits the sulcus-like morphology of Cr/SiO2-400, suggesting that the specific surface of the catalyst may occur mainly in mesoporous form. Figure 3(c) further confirms the analysis results shown in Figure 3(a) and (b) and also suggests that the nano-sized particles are of irregular spherical shape and that the structure of Cr/SiO2 was relatively loose due to the presence of mesoporous and microporous forms. Figure 3(e)–(i) shows the distributions of elements obtained by EDS of the Cr/SiO2-400 sample. The distribution diagrams (Figure 3(f)–(i)) show that Si, O, and Cr are almost uniformly distributed on the surface of the sample and that Cr had been successfully introduced into the SiO2 catalyst carrier. Moreover, Cr oxides of the catalyst Cr/SiO2-400 were not observed by TEM or FESEM, which is in accordance with the results of XRD analysis.

(a) and (b) FESEM images of Cr/SiO2-400, (c) and (d) TEM images of Cr/SiO2-400, and (e–i) the elemental distribution mappings of Cr/SiO2-400 obtained by FESEM-EDS.
XPS was used to further reveal the elemental composition and surface chemical valence of the Cr/SiO2 material and SiO2. In Figure 4(a), the wide range XPS spectrum of the Cr/SiO2-400 catalyst included Si 2p, O 1s, and Cr 2p, indicating the presence of Si, O, and Cr in the catalyst, consistent with the results of EDS analysis. The high-resolution XPS of Si 2p of Cr/SiO2 confirmed the existence of Si–O in SiO2 at 103.2 eV. 42 According to Figure 4(c), the binding energies of 532.5 and 530.9 eV were, respectively, assigned to Si–O and Cr–O. 43 Interestingly, the binding energies of the Si and O peaks in the Cr/SiO2-400 spectrum shifted significantly to lower binding energies compared to those of the carrier SiO2. Such changes confirmed the existence of electron interaction between SiO2 and the Cr oxide, which had a positive effect on the activity and stability of the prepared catalyst.
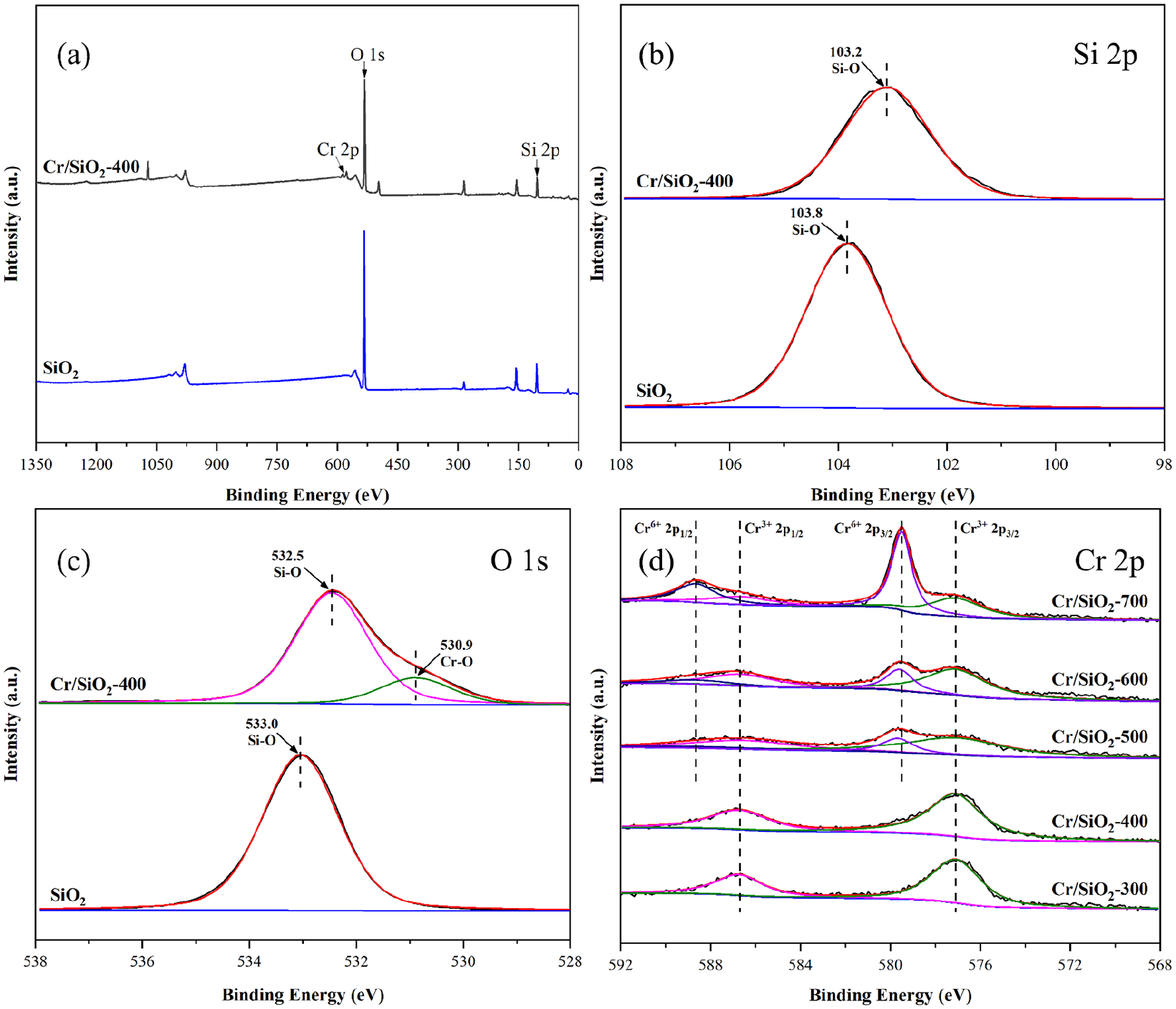
(a) The wide-range XPS spectrum of Cr/SiO2-400. High-resolution XPS spectra of (b) Si 2p and (c) O 1s of Cr/SiO2-400 and SiO2 and (d) Cr 2p spectra of Cr/SiO2-300, Cr/SiO2-400, Cr/SiO2-500, Cr/SiO2-600, and Cr/SiO2-700.
Figure 4(d) summarizes the Cr 2p high-resolution XPS spectra for a series of Cr/SiO2 catalysts. The peaks at 577.1 and 586.7 eV correspond to Cr 2p3/2 and Cr 2p1/2 of Cr3+, respectively, while the other two peaks at 579.5 and 588.7 eV correspond to Cr 2p3/2 and Cr 2p1/2 of Cr6+, respectively.44–46 Furthermore, the ratios of Cr in different valence states on the surfaces of the catalyst samples obtained at continuous calcination temperatures were calculated. Above 400 °C, the content of Cr3+ in the catalyst decreased with increasing calcination temperature, and the catalytic activity of the corresponding catalyst for the cyclohexane oxidation decreased gradually, indicating that Cr3+ played an important role in this reaction. Although Cr in Cr/SiO2-300 mainly existed in the form of Cr3+, the Cr oxides may not be well dispersed, resulting in a lower catalytic activity than that of the Cr/SiO2-400 catalyst.
H2-TPR was used to test the reduction of the catalyst, and the results are shown in Figure 5. For Cr oxide and Cr/SiO2-400 catalysts, a significant hydrogen depletion peak (due to reduction of Cr3+) could be observed, indicating that Cr exists mainly as Cr3+ in the catalysts. 47 In addition, the reduction peak of Cr/SiO2-400 shifted to a higher temperature than that of the single Cr oxide, indicating that the intimate interaction between SiO2 and the Cr oxide in the catalyst can influence the metal reduction temperature.

H2-temperature-programmed reduction profiles of the bare Cr oxide and the Cr/SiO2-400 catalysts.
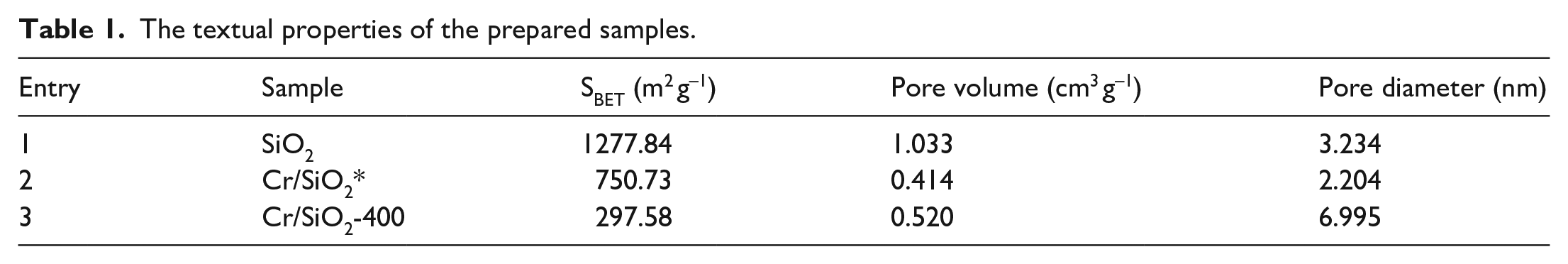
The N2 adsorption-desorption isotherms and the pore size of the corresponding samples were measured, and the results are shown in Figure 6. According to the International Union of Pure and Applied Chemistry (IUPAC) classification, pure SiO2 is represented as a type IV isotherm with H2 hysteresis loops, while the Cr/SiO2-400 catalyst is represented as a type IV isotherm with H4 hysteresis loops, indicating that both were relatively typical of mesoporous materials and the pore size distribution of SiO2 was more regular than that of the catalyst. The Cr/SiO2* catalyst obtained by general impregnation was shown as a type I isotherm, which reflects the micropore filling phenomenon on the microporous adsorbent. Such a phenomenon indicated that the pore channels in the catalyst exist mainly in the form of micropores. Figure 6(b) and (c) shows the pore profiles of Cr/SiO2-400 and SiO2 based on the Barret-Joyner-Halenda (BJH) model, suggesting that their pore size was in the 2–10 nm range, which supported the mesoporous structure of the prepared catalyst. As shown in Table 1, the specific surface area of the Cr/SiO2-400 was smaller than that of the SiO2 carrier, which may be due to the collapse or plugging of the mesopores after doping with the metal. The Cr/SiO2-400 catalyst had a larger pore diameter (6.995 nm), so it is reasonable to deduce that wider channels are more favorable for the enhancement of catalytic activity, which can offer more active sites and promote mass transfer during the reaction process.

(a) N2 adsorption-desorption isotherms of the prepared catalyst and (b) and (c) pore size distribution curves (inset) of Cr/SiO2-400 and SiO2.
The textual properties of the prepared samples.
Catalyst activity
The prepared Cr/SiO2 and other catalyst samples were examined for their catalytic activity in the selective oxidation of cyclohexane. The results are shown in Table 2. Entry 1 represents a blank experiment without any catalyst, and no products were detected after the reaction, indicating that it was difficult to realize the oxidation process with hydrogen peroxide alone. According to entry 2, SiO2 also did not exhibit any catalytic activity for this reaction. As for entries 3–7, on increasing the calcination temperature, the selectivity for KA oil increased. The Cr/SiO2-700 catalyst gave the highest selectivity for KA oil (94.64%), but the yield of KA oil was only 39.96%, indicating that Cr/SiO2-700 had poorer catalytic capacity compared to the other catalysts (Cr/SiO2-400, Cr/SiO2-500, and Cr/SiO2-600). With decreasing calcination temperature, the catalytic activity of the catalyst increased significantly. Notably, the best catalyst Cr/SiO2-400 calcined at 400 °C and can achieve a 54.59% cyclohexane conversion and 90.01% selectivity for KA oil. To sum up, when Cr/SiO2 was used as the catalyst, the yield of KA oil was the highest, which suggested that 400 °C was a more suitable calcination temperature.
Effect of the different prepared catalysts on the catalytic activity for the selective oxidation of cyclohexane to KA oil.

KA: ketone-alcohol.
Reaction conditions: cyclohexane (2 mmol), H2O2 (4 mmol), acetonitrile (1 mL), catalyst (10 mg), 6 h, 50 °C.
For comparison, catalysts loaded with Cr oxides on other supports were used for the oxidation of cyclohexane (entries 8–13). Based on the results of entries 8–13, it was obvious that the catalysts (Cr/SiO2) prepared by in situ homogeneous coprecipitation commonly showed better catalytic activity. Accordingly, the in situ homogeneous coprecipitation method can guarantee the formation of an intimate interaction between SiO2 and Cr oxides, which makes Cr/SiO2 an efficient catalyst for the selective oxidation of cyclohexane in the liquid phase.
To study the effects of different reaction parameters on the oxidation of cyclohexane, the catalyst Cr/SiO2-400 was used for its selective oxidation. It was found that the solvent had an important influence on the reaction process. Therefore, we first investigated the solvent effect, using water, acetonitrile, N,N-dimethylformamide (DMF), acetone, ethyl acetate, and methylene chloride, and the results are shown in Table 3. When using water as the solvent, the oxidation reaction proceed sparingly (entry 1). Moreover, it was difficult to obtain satisfactory conversion and selectivity in a two-phase solvent (entries 5 and 6). A significant increase in the catalytic activity may be due to the greater solubility of hydrogen peroxide in DMF and acetone; however, the best result was obtained in acetonitrile, with 54.59% cyclohexane conversion and 90.01% KA oil selectivity.
Effect of different solvents on the catalytic activity for the selective oxidation of cyclohexane to KA oil.

KA: ketone-alcohol.
Reaction conditions: cyclohexane (2 mmol), H2O2 (4 mmol), Cr/SiO2-400 (10 mg), 6 h, 50 °C.
As shown in Figure 7(a), the conversion of cyclohexane increased on increasing the temperature, but there was a slight downward trend above 50 °C, which may be due to the decomposition of H2O2 at higher reaction temperatures. Furthermore, the selectivity for cyclohexanone (K) increased with increasing temperature, reaching a maximum value at 50 °C. Therefore, considering the conversion of cyclohexane and the selectivity of KA oil, 50 °C was considered the optimal temperature for the next step.

Effects of (a) the reaction temperature and (b) the reaction time on the selective oxidation of cyclohexane over Cr/SiO2-400.
Figure 7(b) displays the effect of the reaction time on the selective oxidation of cyclohexane. The selectivity of cyclohexanol (A) decreased as the reaction progressed, and the cyclohexanol (K) selectivity reached a maximum of 72.92% at 5 h, which successfully verified that cyclohexanol (A) was a reaction intermediate en route to cyclohexanone (K). The conversion of cyclohexane leveled off after 6 h, indicating a balanced catalytic capacity of the catalyst. On further extending the reaction time, a slight decrease in selectivity and an increase in byproducts in the KA oil may be due to excessive oxidation of cyclohexane to undesired byproducts. The highest yield of KA oil (49.14%) was obtained at 6 h, which was thus chosen as the optimum reaction time for the oxidation of cyclohexane. As shown in Figure S2, when the catalyst dosage was increased from 2.5 to 10 mg, the conversion of cyclohexane increased significantly. On increasing the catalyst loading to 25 mg, the conversion of cyclohexane showed no further improvement, so the optimum catalyst dosage was 10 mg. As shown in Figure S3, when the molar ratio of hydrogen peroxide to cyclohexane was less than 2, the conversion of cyclohexane increased obviously.
For heterogeneous catalysts, the stability of the catalyst is also an important aspect. Therefore, the Cr/SiO2-400 catalyst was used in catalyst cycling experiments under the optimized reaction conditions. After each reaction, the catalyst was separated by centrifugation, washed several times with ethanol, and dried at 80 °C for 12 h in an oven. It can be seen from Figure 8 that when using Cr/SiO2-400 as the catalyst for the selective oxidation of cyclohexane, the selectivity for KA oil was still over 90% even after four runs. However, over continued cycling, the conversion of cyclohexane continued to decline, possibly due to slight leaching of Cr species. Besides, the XRD pattern of the catalyst after four cycles was similar to that of the fresh catalyst (Figure S4), thus showing that the structure of silica has excellent stability. In general, Cr/SiO2 demonstrated an excellent catalytic performance, was easy to prepare, and exhibited good stability for the selective oxidation of cyclohexane, thereby highlighting its potential for the preparation of KA oil under mild conditions.
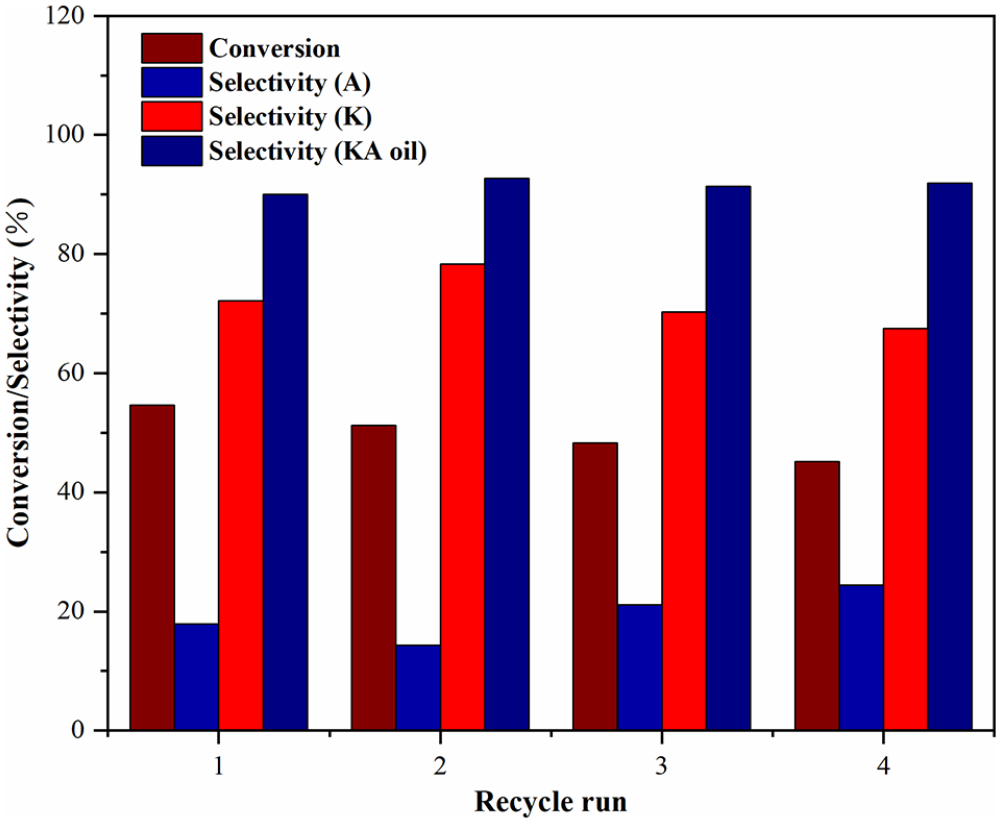
Recycling of the Cr/SiO2-400 catalyst for the selective oxidation of cyclohexane to ketone-alcohol oil with H2O2.
Conclusion
In summary, a series of Cr-doped SiO2 composites were successfully prepared by a facile in situ coprecipitation method. A maximum KA oil yield of 49.14% was obtained within 6 h, which was better than the yields obtained from most published studies based on heterogeneous catalysts. Based on various characterization methods, the catalytic performance of the catalysts can be correlated with the content of low valence chromium. Moreover, the intimate interaction between CrOx and SiO2 guaranteed good catalytic activity and excellent catalyst reusability making the Cr/SiO2-400 catalyst efficient over four cycles. This work provides a promising approach for the design of catalysts for KA oil production with highly selective oxidation of cyclohexane under mild conditions in the presence of H2O2.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221131289 – Supplemental material for A one-step method for preparing Cr-doped SiO2: An efficient catalyst for the direct oxidation of cyclohexane to ketone-alcohol oil
Supplemental material, sj-docx-1-chl-10.1177_17475198221131289 for A one-step method for preparing Cr-doped SiO2: An efficient catalyst for the direct oxidation of cyclohexane to ketone-alcohol oil by Xiaozhong Wang, Kaicheng Xue, Cai Xu, Yingqi Chen and Liyan Dai in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Provincial Key Laboratory of Advanced Chemical Engineering Manufacture Technology (No. 2017E10001), Zhejiang Province (China).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

