Abstract
The highly practical acetylation of free nucleosides is achieved using acetic anhydride/acetic acid as a reusable solvent and acetylating regent. A series of nucleosides, including ribosyl, deoxyribosyl, arabinosyl, acyclic and pyranosyl, and many clinical drugs were acetylated efficiently, even on large scale (200 g).

Introduction
In recent years, modified nucleosides have become of significant interest due to their intriguing biological and pharmacological properties.1,2 In the context of the outbreak of COVID-19, nucleoside drugs have once again attracted the attention of scientists, and remdesivir,
3
molnupiravir,
3
azvudine
4
have been used clinically for the treatment of COVID-19. The late-stage modification of nucleosides is an important method for obtaining a variety of nucleoside drugs.
5
However, the direct modification of free nucleosides is often inhibited or there is a lack of selectivity due to the presence of hydroxy groups on the sugar rings.6,7 As a result, the development of methods for the protection of hydroxy groups has been the subject of intense research.
8
In addition, the presence of an acetoxy group in pharmaceuticals often modulates their solubility and pharmacokinetics, while optimizing their pharmacological and physiochemical properties.
9
The acetylation of hydroxy groups is often a powerful and low-cost approach, which is routinely carried out by reacting free nucleosides with acetic anhydride in the presence of a catalyst, for example, DMAP (4-dimethylaminopyridine), which is the most widely used catalyst for this purpose.
10
In factories, the most common way to produce 1,2,3,5-tetraacetyl-β-d-ribose (
The synthesis of 1,2,3,5-tetraacetyl-β-d-ribose (
This finding prompted us to improve the acetylation of the hydroxy groups of nucleosides, avoiding the existing deficiencies. Following on from our previous research on nucleosides,12–15 we herein report the practical acetylation of the hydroxy groups of nucleosides without an additional catalyst by using acetic anhydride/acetic acid as a reusable solvent.
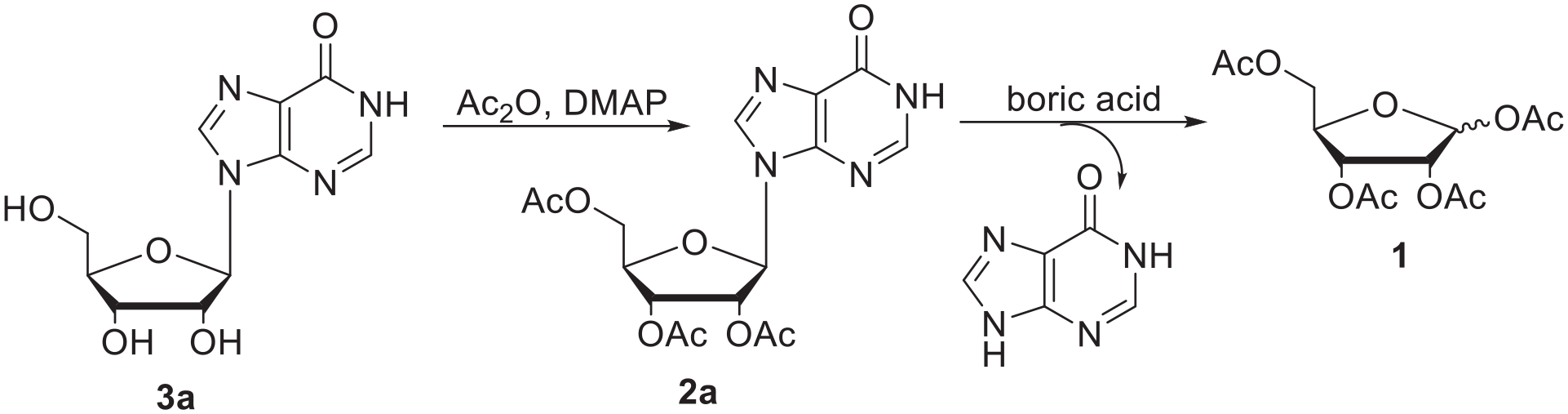
We began our studies by using inosine (

The acetylation of inosine (
Optimization of the reaction conditions for the acetylation of inosine (
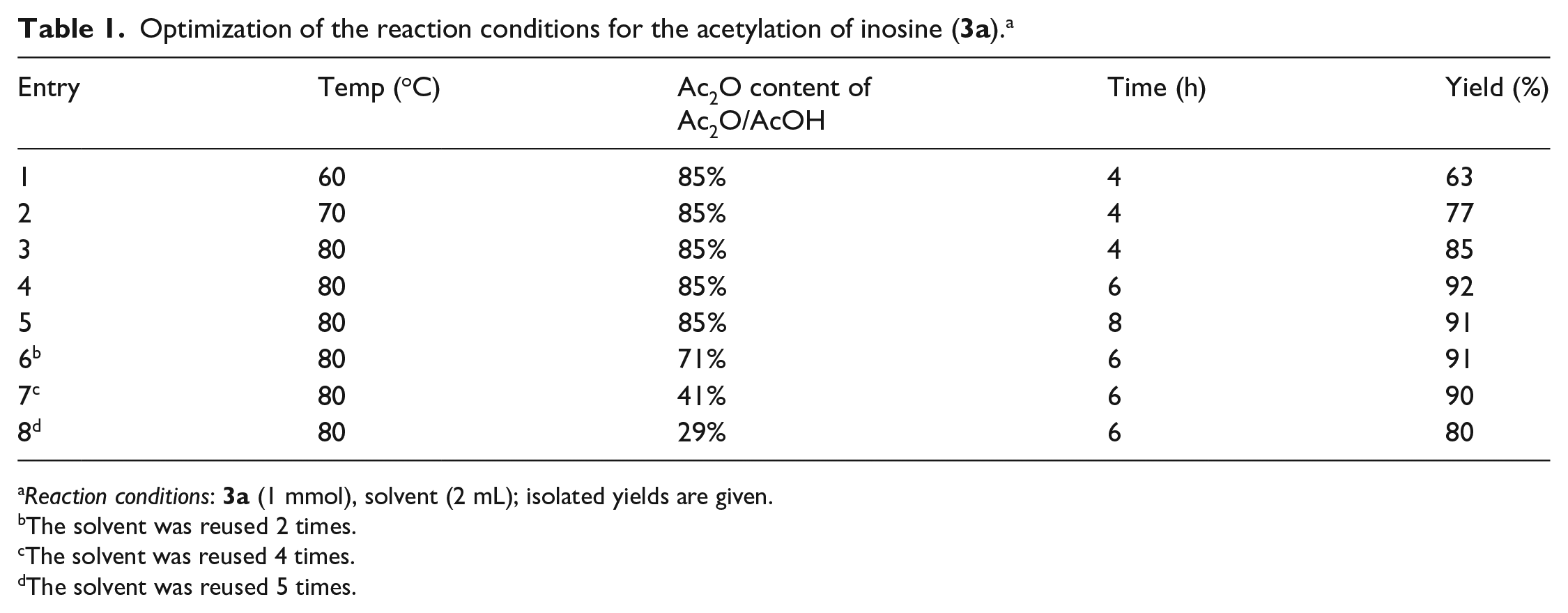
Reaction conditions:
The solvent was reused 2 times.
The solvent was reused 4 times.
The solvent was reused 5 times.
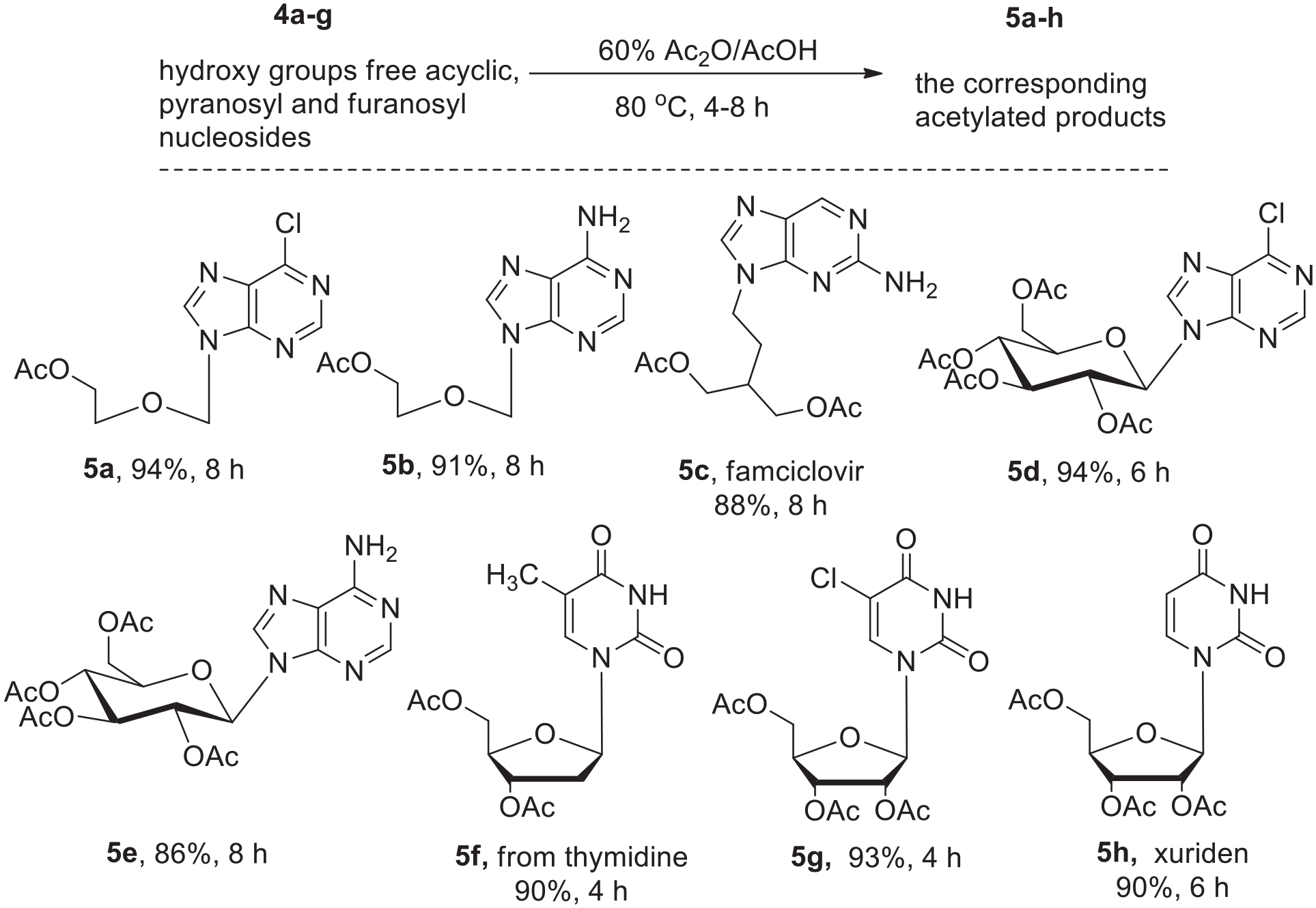
Under the optimized reaction conditions and with 60% acetic anhydride/acetic acid as the solvent, a series of free nucleosides was subjected to the acetylation reaction under the optimized conditions (Scheme 3). To our delight, all the hydroxy groups were acetylated to afford satisfactory yields of products (

The acetylation of a series of free nucleosides.
Subsequently, a variety of nucleosides bearing other glycals or nucleobases (
The acetylation of free nucleosides bearing other glycals or nucleobases.
Next, reactions on large scale demonstrated the robustness and preparative scale utility of this process. 2’,3’,5’-tri-O-Acetylinosine (
Conclusion
In summary, we have developed a highly practical and improved process for the acetylation of the hydroxy groups of nucleosides. The acetic anhydride could be reused 4–5 times. A series of nucleosides including ribosyl, deoxyribosyl, arabinosyl, acyclic and pyranosyl worked well. What is more, the catalytic system tolerated different functional groups including F, Cl, and NH2. Importantly, the drugs nebularine, clofarabine, fludarabine, vidarabine, famciclovir and xuriden could be successfully acetylated, providing a simple method for the late-stage modification of these drugs. In addition, the reaction scale could be increased to 200 g, which is important for industrial applications. Further investigations on this reaction are underway in our laboratory.
Experimental
General
Melting points were recorded with a X-5 micro melting point apparatus and uncorrected (Beijing Taike Instruments Co., Ltd.). 1H and 13C NMR spectra were recorded on a Bruker AC 400 spectrometer (Bruker, Billerica, MA, USA) as CDCl3 or DMSO-d6 solutions. Chemical shifts are expressed in parts per million (δ) downfield from the internal standard tetramethylsilane. Multiplicities are reported as s (singlet), d (doublet), t (triplet), q (quartet), m (multiplet), and dd (doublet of doublets), and coupling constants (J) are given in hertz (Hz). Mass spectra were obtained on a Waters Q-Tof MicroTM spectrometer (Waters Synapt).
General procedure
Free nucleoside (1 mmol) was added to 60% acetic anhydride-acetic acid (2 mL). The resulting mixture was heated to 80 oC and kept at this temperature for 4–8 h. Upon completion of the reaction, the solvent was removed under vacuum. The product was purified by column chromatography on silica gel (eluent: ethyl acetate/petroleum ether) to give the pure product.
Large-scale procedure (200 g scale) for the synthesis of 2a
Inosine (200 g, 746 mmol) was added to 60% acetic anhydride-acetic acid (800 mL). The resulting mixture was stirred and heated at 80 oC in an oil bath for 10 h. Upon completion of the reaction, the solvent was recovered under vacuum and the residual solvent was co-evaporated with ethanol (50 mL). The oily residue was recrystallized from H2O to give the pure product (90% yield).
(2R,3R,4R,5R)-2-(Acetoxymethyl)-5-(6-oxo-1H-purin-9(6H)-yl)tetrahydrofuran-3,4-Diyl diacetate (

(2R,3R,4R,5R)-2-(Acetoxymethyl)-5-(6-amino-9H-purin-9-yl)tetrahydrofuran-3,4-diyl Diacetate (
(2R,3R,4R,5R)-2-(Acetoxymethyl)-5-(9H-purin-9-yl)tetrahydrofuran-3,4-diyl Diacetate (
(2R,3R,4R,5R)-2-(Acetoxymethyl)-5-(6-amino-2-chloro-9H-purin-9-yl)tetrahydrofuran-3,4-diyl Diacetate (
(2R,3R,4S,5R)-2-(Acetoxymethyl)-5-(6-amino-9H-purin-9-yl)tetrahydrofuran-3,4-diyl Diacetate (
((2R,3R,4S,5R)-3-Acetoxy-5-(6-amino-2-chloro-9H-purin-9-yl)-4-fluorotetrahydrofuran-2-yl)methyl Acetate (
(2R,3R,4S,5R)-2-(Acetoxymethyl)-5-(6-amino-2-fluoro-9H-purin-9-yl)tetrahydrofuran-3,4-diyl Diacetate (
((2R,3S,5R)-3-Acetoxy-5-(6-amino-9H-purin-9-yl)tetrahydrofuran-2-yl)methyl Acetate (
2-((6-Chloro-9H-purin-9-yl)methoxy)ethyl Acetate (
2-((6-Amino-9H-purin-9-yl)methoxy)ethyl Acetate (
2-(2-(2-Amino-9H-purin-9-yl)ethyl)propane-1,3-diyl Diacetate (Famciclovir) (

(2R,3R,4R,5R,6R)-2-(Acetoxymethyl)-6-(6-amino-9H-purin-9-yl)tetrahydro-2H-pyran-3,4,5-triyl Triacetate (

(2R,3R,4R,5R,6R)-2-(Acetoxymethyl)-6-(6-amino-9H-purin-9-yl)tetrahydro-2H-pyran-3,4,5-triyl Triacetate (
((2R,3S,5R)-3-Acetoxy-5-(5-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)tetrahydrofuran-2-yl)methyl Acetate (

(2R,3R,4R,5R)-2-(Acetoxymethyl)-5-(5-chloro-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)tetrahydrofuran-3,4-diyl Diacetate
Xuriden (
Supplemental Material
sj-pdf-1-chl-10.1177_17475198221115020 – Supplemental material for The practical acetylation of nucleosides using acetic anhydride/acetic acid as a reusable solvent
Supplemental material, sj-pdf-1-chl-10.1177_17475198221115020 for The practical acetylation of nucleosides using acetic anhydride/acetic acid as a reusable solvent by Ran Xia, Chao Xia, Ying-Xing Yang, Li-Jie Liu, Lei-Shan Chen and Li-Ping Sun in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Key Scientific and Technical Projects of Henan Province (212102310333, 222102110403), the Training Plan for Young Key Teachers in Universities of Henan Province (202GGJS248), the Key Scientific and Technical Projects of Xinxiang City (GG2021016) and the Major Scientific and Technical Projects of Xinxiang City (21ZD008) for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
