Abstract
The development of a simple, inexpensive, sensitive, and selective spectrofluorometric assay method for taurine in energy drinks utilizing 4-fluoro-7-nitro-2,1,3-benzoxadiazole as a fluorogenic agent is described. Conditions affecting the derivatization reaction are investigated and optimized. The optimal conditions are found to be as follows: buffer, pH 10; 4-fluoro-7-nitro-2,1,3-benzoxadiazoleconcentration, 0.05%; reaction time, 30 min; temperature, 50 °C. Fluorescence measurements are carried out at a wavelength of 542 nm, with excitation at a wavelength of 485 nm. The method is validated under the optimum conditions. The method is found to be linear in the range of 2.0–12.5 µg/mL, with a correlation coefficient of 0.9993. The limits of detection and quantification (µg/mL) are 0.6 and 2.02, respectively, which are appropriate for taurine assays in commercial energy drinks. In addition, the proposed method recorded excellent accuracy since the recovery values caused by presence of excipients are found to be in the range of 99.3%–102.87%. The values of relative standard deviation for intra- and inter-day precision were found to be in the ranges of 0.236%–0.659% and 2.12%–2.63%, respectively.

Introduction
Energy drinks have recently become popular beverages among young adults, 1 who consume energy drinks to maintain physical comfort, improve performance and awareness, and stimulate thought processes. 2 In addition to other active ingredients, taurine is a major constituent of energy drinks as well as infant formulations and dietary supplements. Taurine stimulates brain activity, attention, and memory. Taurine is also called 2-aminoethanesulfonic acid, is a free nonessential amino acid. It plays a significant role in fat absorption in the human body and heart protection; besides it is considered an antioxidant. 3 On the other hand, overdoses of taurine result in adverse effects. In extreme cases, death was documented by overdoses of taurine–caffeine combinations. 4 In addition, another recent study reported that concomitant ingestion of taurine and caffeine can cause a transient choroidal thinning, which is observed in eyes with thick choroids. 5 A relationship between energy drinks, incorporating taurine, and an acute significant impairment in endothelial function along with considerable hemodynamic changes, in healthy teenagers was recently reported. 6 Furthermore, it was observed that, of considerable relevance to transfusion medicine, taurine modulates red blood Cell antioxidant metabolism in vivo and ex vivo. 7 Therefore, the quantification of taurine in energy drinks is very important in the drinks industry for quality control as well as in food centers for monitoring food safety.
Due to the increase in the consumption and quality of energy drinks, a wide range of analytical techniques, including spectroscopic, separation-based, and electrochemical methods, are utilized for developing assay procedures for taurine. In this context, spectrophotometry, 8 spectrofluorometry, 9 Fourier transform infrared (FTIR) spectrometry,10,11 and Fourier-transform mid-infrared spectrometry 12 have all been exploited for assaying taurine in energy drinks. High- performance liquid chromatography coupled with ultraviolet (UV),13–16 fluorescence, 13 mass spectrometric 17 and evaporative light scattering 16 detection methods are utilized. In addition, capillary electrophoresis with UV,3,18 fluorescence 19 and contactless capacitive coupled conductivity detection18,20,21 methods have been reported.
Spectrofluorometric methods feature the advantage of simplicity over separation-based methods, as well as the benefits of high selectivity and sensitivity over spectrophotometric techniques. Nevertheless, in the case that the targeted analyte does not include a fluorophore group, as in taurine, derivatization is required for developing spectrofluorometric methods to yield a fluorescent product. In this context, several fluorogenic reagents have been reported for fluorescence derivatization reactions, including 4-fluoro-7-nitro-2,1,3-benzoxadiazole (NBD-F), 4-chloro-7-nitrobenzo-2-oxa-1,3-diazole (NBD-Cl), 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate (AQC), o-phthaldialdehyde (OPA), and 1,3,5,7-tetramethyl-8-(N-hydroxysuccinimidyl butyric ester) difluoroboradiaza-s-indacene (TMBB-Su).22,23 Because of its high interactivity with both secondary and primary amines, NBD-F is an efficient and promising fluorogenic reagent. 24 Nucleophilic substitution reactions of NBD-F with amines yield stable and strongly fluorescent products with suitable excitation/emission wavelengths.25–27 Notably, the use of NBD-F as a labeling reagent for taurine determination using spectrofluorometry has not yet been reported. Therefore, this research is directed toward the optimization and validation of a cheap and sensitive spectrofluorometric procedure for assaying taurine in various energy drinks based on the coupling reaction of NBD-F with taurine.
Results and discussion
Optimization of the reaction conditions
The parameters affecting the reaction of taurine with NBD-F, that is, pH, NBD-F concentration, reaction time, and temperature were investigated and adjusted. Each factor was changed individually while others remained unchanged.
Effect of pH
The influence of the pH on the reaction of taurine with NBD-F was studied by changing the pH from 9.0 to 11.5 (Figure 1). At pH 10.0, the fluorescence intensity attained its highest value. In another context, this is the point at which the degree of the nucleophilic substitution reaction was the greatest. At pH >10.0, the fluorescence intensity of the solution declined sharply, which can be attributed to the increase in OH- ions restricting the nucleophilic replacement reaction between taurine and the fluorogenic reagent. Therefore, the fluorescence intensity of the solution decreased. Thus, a pH of 10.0 was considered as optimum.

Effect of pH on the relative fluorescence intensity (RFI) of the reaction product of taurine with NBD-F under fixed conditions: Time, 30 min; temperature, 50 °C; NBD-F concentration, 0.05%.
Effect of NBD-F concentration
The effect of the NBD-F concentration was studied in the range of 0.0125%–0.075% (weight/volume). As shown in Figure 2, the obtained results show that the reaction was influenced by the NBD-F concentration. The maximum fluorescence intensity was achieved at an NBD-F concentration of 0.05%, while the fluorescence intensity remained almost constant at an NBD-F concentration higher than 0.05% (weight/volume).
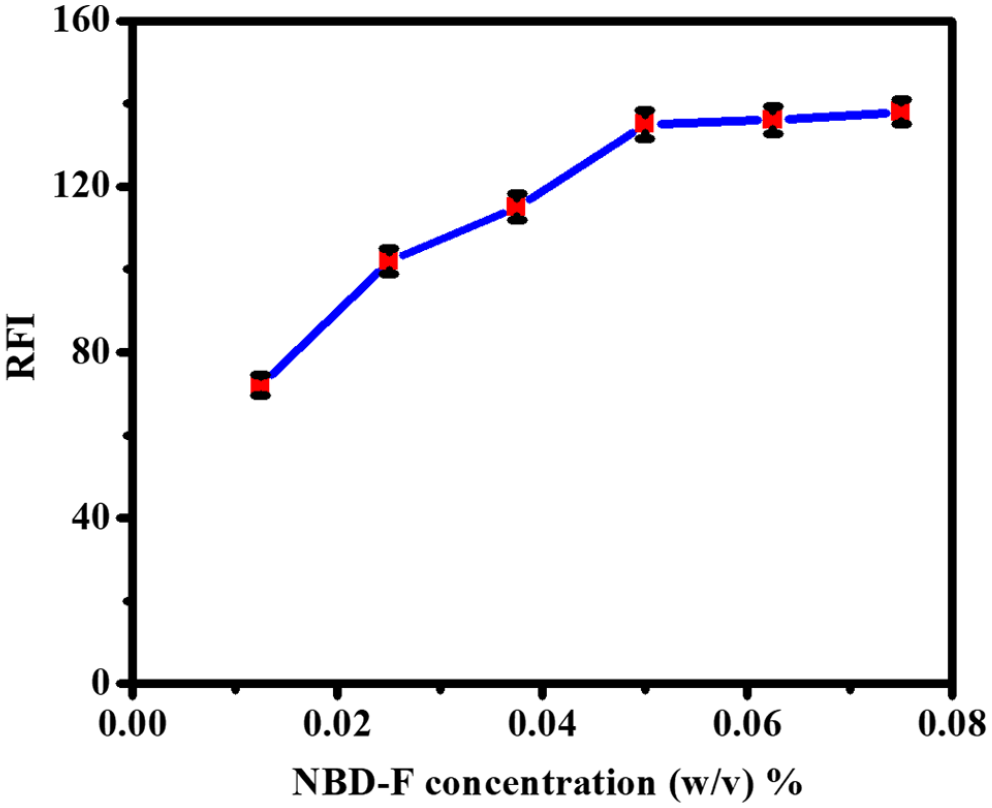
Effect of NBD-F concentration (% (weight/volume)) on the relative fluorescence intensity (RFI) of the reaction product of taurine with NBD-F under fixed conditions: Time, 30 min; temperature, 50 °C; NBD-F concentration, 0.05%; pH, 10.0.
Effect of temperature
The effect of temperature on the reaction of taurine with NBD-F was investigated by increasing the temperature from 30 to 70 °C. Figure 3 shows that the reaction did not progress at room temperature and that the maximum fluorescence intensity was reached at 50 °C.
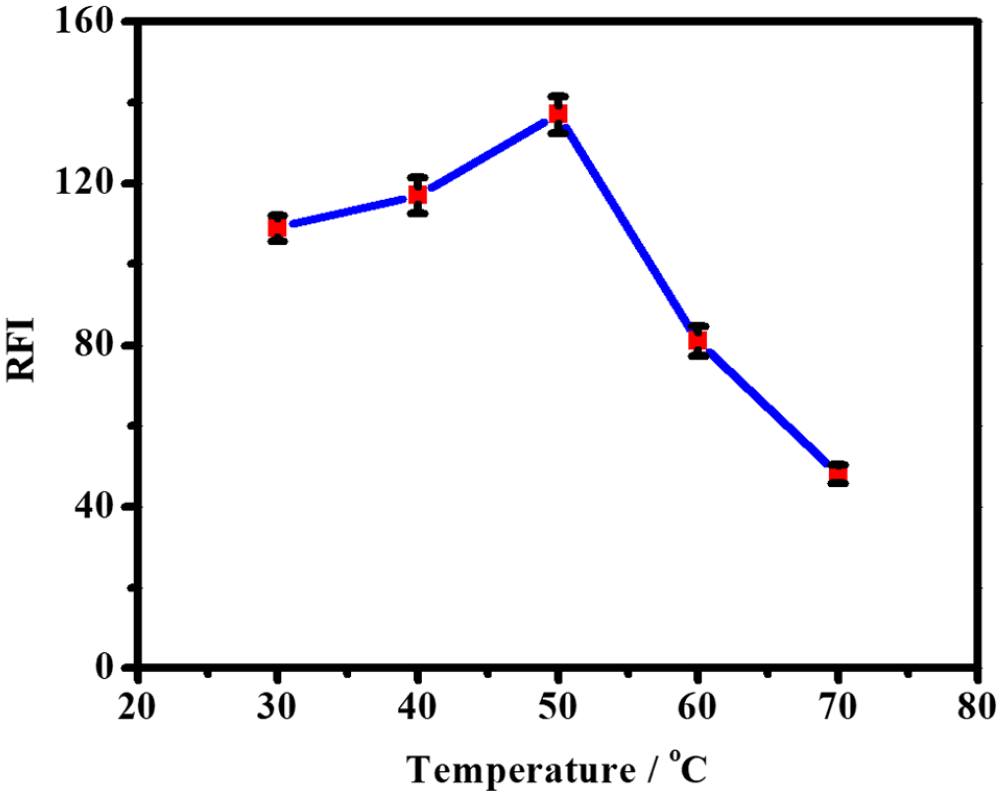
Effect of temperature on the relative fluorescence intensity (RFI) of the reaction product of taurine with NBD-F under fixed conditions: Time, 30 min; NBD-F concentration, 0.05%; pH, 10.0.
Effect of reaction time
The influence of the reaction time on the interaction of taurine with NBD-F was also examined by increasing the reaction time from 10 to 50 min. The results are depicted in Figure 4. The maximum relative fluorescence intensity value was attained at 30 min. In general, the NBD-F reactions recorded reproducible fluorescence intensities when heating at a relatively lower temperature for a longer period.28,29

Effect of the reaction time on the relative fluorescence intensity (RFI) of the reaction product of taurine with NBD-F, under fixed conditions: Temperature, 50 °C; NBD-F concentration, 0.05%; pH, 10.0.
Stoichiometric ratio determination
Under the optimized conditions, the stoichiometric ratio between NBD-F and taurine was established to be 1:1 (Figure 5). Accordingly, the reaction is proposed to occur as presented in Scheme 1, suggesting that the taurine used in this investigation reacts with NBD-F to form a fluorescent product.

Determination of the product formation via the continuous variation method, VR: NBD-F, VD: Taurine, VR+VD (NBD-F volume + taurine volume) = 10 mL.

The proposed reaction between taurine and the NBD-F reagent.
Analytical method validation
The proposed spectrofluorometric method was validated in terms of linearity, limit of quantitation (LOQ), limit of detection (LOD), and intra- and inter-day precision, following the ICH recommendations. 30
Linearity, limit of detection (LOD) and limit of quantification (LOQ)
In the current proposed spectrofluorometric method, a linear plot, using six standard solutions, with a good correlation coefficient (0.9993) was obtained in the concentration range of 2.0–12.5 µg/mL. The calibration equation of the linear regression was found to be y = 40.657x + 31.559; where y is the relative fluorescence intensity and x is the taurine concentration in µg/mL. The obtained high value of the slope (40.657) indicates a high sensitivity for the proposed method. Notably, the standard deviation of the slope was 0.720, while the standard deviation of the intercept was 4.994. In addition, the standard error of the estimate (or standard deviation of the regression) was 8.126, indicating closer points to the calibration curve. Furthermore, the F value was 3186.569, while the degree of freedom was 4. Whereas the sum of the squares of the regression was 210437.523, the sum of the squares of the residuals was 264.156.
LOD and LOQ were measured using the formula: LOQ or LOD = κSDa/b; where κ = 10 for LOQ and 3.3 for LOD; SDa is the standard deviation of the intercept, and b is the slope. The LOD and LOQ were found to be 0.41 and 1.23 µg/mL, respectively. The range of taurine in commercial energy drinks available in the Saudi market is from 30 to 400 mg in 100 mL, suggesting that the current spectrofluorometric method is suitable for routine assaying of taurine in commercial energy drinks.
Table 1 shows a comparison of various assay methods for taurine in terms of methods of detection, linear range, and LOQ. As shown, while the LOQ of the current spectrofluorometric method was higher than a previous spectrofluorometric method, 9 the linear range of the current method was wider than that of the previous method. 9 In addition, the current method recorded a lower level of the linear range than that of a previously described spectrophotometric method. 8 Notably, the LOQ values of previous high performance liquid chromatography (HPLC) methods with UV, 13 fluorescence 13 and MS detection were lower than that of the current method. For a previous capillary electrophoresis/UV method, 3 a higher level and a wider linear range was obtained from the previous method compared to that was obtained from the current method. Nevertheless, the previous CE/UV method 3 reported a higher LOQ value than that of the current method.
Previously reported assay methods for taurine.

LOQ: limit of quantitation.
Capillary electrophoresis.
High-performance liquid chromatography.
Contactless capacitive coupled conductivity detection.
Precision and accuracy
The precision of the procedure was assessed by analysis of taurine at three concentrations (1, 5 and 10 µg/mL) (each n = 6) for six successive days. The values of RSD for intra- and inter-day precision were measured to be 0.2362%–0.6589% and 2.12%–2.63%, respectively, indicating the high precision of the method (Table 2). Comparable results for the intra-day precision of the current spectrofluorometric method and a previously reported spectrophotometric method (0.14%–1.59%) were observed. 8
Intra- and inter-day precision for the repeated measurements of different concentrations of taurine.

The standard addition method was also employed to examine the accuracy of the procedure. Sample solutions of 2 µg/mL were added to 2, 4, 8 µg/mL concentrations of standard solutions and analyzed. The obtained results are recorded in Table 3. The average recoveries were measured to be 101.35%.
The recovery levels of the proposed spectrofluorometric method.

Relative standard deviation of three determinations.
Analytical method application
The proposed method was applied for the analysis of taurine in three samples of energy drinks using the calibration equation presented in the section on analytical method validation. The obtained results, presented in Table 4, are compared with those of the labeled amounts to calculate the recovery. Notably, for the energy drink samples including 30 mg/100 mL of taurine (i.e. Code Red® and W1®), the samples were diluted with a dilution factor of 50. For the energy drink sample including 400 mg/100 mL of taurine (i.e. Bison®), the dilution factor was 1000. The dilutions are carried out to assay the analyte in the range of calibration curve to obtain acceptable accuracy and precision. In the event, high recovery levels (101.8%–108.5%) were observed. Advantageously, the reported procedure has the advantage of being practically free from interference by other ingredients in the energy drink.
Content of taurine in energy drinks.

Experimental
Chemicals
NBD-F (purity 98.0%), taurine (purity ⩾ 99.0 %) and hydrochloric acid (purity 37%) were purchased from Sigma-Aldrich (Steinheim, Germany).
Instrumentation
All spectrofluorometric measurements were carried out using a spectrofluorometer with a xenon lamp (Shimadzu, model RF-1501, Japan). A pH meter (pH-25 Benchtop pH meter) was used for pH measurements.
Preparation of standard solutions and reagents
A taurine stock standard solution (1.0 mg/mL concentration) was prepared by dissolving taurine (0.050 g) in 50 mL of deionized water. Thereafter, a series of taurine standard solutions (concentration range of 2.00–12.5 µg/mL) was prepared by dilution.
NBD-F solution (0.5% w/v) was prepared by transferring 0.125 g into a 25 mL volumetric flask and dissolving in methanol. The solution was stored in an amber glass bottle in a refrigerator, and was stable for 1 week.
The borate buffer solution (10 mM) was prepared by dissolving 0.29 g of sodium chloride and 0.31 g of boric acid in deionized water in a 100 mL volumetric flask. The pH was then adjusted to 10 with 1.0 M KOH.
Energy drink samples
Three samples of energy drinks containing taurine were collected from a local market in Al-Ahssa, Saudi Arabia, including Code Red® (Al-ELASI Beverage Co. Ltd., Jeddah, Saudi Arabia), Bison® (Abu Jedayel Refreshment, Jeddah, Saudi Arabia), and W1® (M.S. Beverage Industry, Jeddah, Saudi Arabia). The samples were diluted with water to give test solutions with a concentration of 6.0 μg/mL of taurine according to the labeled amount.
General procedure
Taurine (1 mL, 2.0–12.5 µg/mL) was transferred into a 10 mL volumetric flask followed by adding 1.0 mL of buffer solution at pH 10.0 and 1.0 mL of 0.5% NBD-F. The mixture was warmed using a thermostat at 50 °C for 30 min. The fluorescence intensities of the resulting solutions were measured at a wavelength of 542 nm after excitation at a wavelength of 485 nm against a reagent blank that was prepared in the same manner (see Figures S1-S4 in the supporting information).
Stoichiometric ratio determination of the reaction by the Job method
The Job method of continuous variation was performed to control the stoichiometric ratio of the reaction of taurine with NBD-F. 26 Equimolar (5.0 × 10-4 M) aqueous solutions of taurine and NBD-F were prepared. A sequence of 5 mL rations of stock solutions of taurine and NBD-F were made up from different corresponding proportions. The solutions were then utilized as described in the general procedure.
Conclusion
This work demonstrates the utilization of NBD-F as a fluorogenic agent for the development of a simple, sensitive, and selective spectrofluorometric technique for the analysis of taurine in beverages. The parameters presumed to be controlling the method (pH, NBD-F concentration, temperature and time) were optimized. The optimization process revealed that pH 10 resulted in the highest fluorescence intensity while the fluorescence intensity declined sharply at pH >10.0. In addition, the fluorescence intensity remains almost constant at the NBD-F concentration higher than 0.05% (weight/volume). In terms of the temperature, the maximum fluorescence intensity was reached at 50 °C, whereas a sharp decrease was observed at a temperature higher than 50 °C. Furthermore, a bell shape was noticed for the effect of the reaction time, with the highest fluorescence intensity level at 30 min. On the other hand, the proposed stoichiometric ratio between NBD-F and taurine was established to be 1:1 under the optimized conditions. The new procedure is superior to chromatographic-based methods in terms of simplicity, instrumentation cost, and running costs, while it is beneficial over spectrophotometric approaches in terms of sensitivity and selectivity. This process could potentially be employed for the routine analysis of taurine in the beverage industry.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221114760 – Supplemental material for Utilization of 4-fluoro-7-nitro-2,1,3-benzoxadiazole (NBD-F) as a fluorogenic reagent for the development of a spectrofluorometric assay method for taurine in energy drinks
Supplemental material, sj-docx-1-chl-10.1177_17475198221114760 for Utilization of 4-fluoro-7-nitro-2,1,3-benzoxadiazole (NBD-F) as a fluorogenic reagent for the development of a spectrofluorometric assay method for taurine in energy drinks by Ahmed O Alnajjar, Abdallah Ahmed Elbashir, Rafea Elgack Elgorashe, Ammar M Ebrahim, Abubakr M Idris and Hany M Abd El-Lateef in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Deanship of Scientific Research and the Vice Presidency for Graduate Studies and Scientific Research at King Faisal University in Saudi Arabia for financial support through the annual funding track [GRANT162].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
