Abstract
The actinomycete strain R1706-8 is isolated from a soil sample collected from the nest of the horned-face bee (Osmia cornifrons) and identified as Streptomyces sp. based upon the results of 16SrRNA sequence analysis. Two concanamycin derivatives obtained from the solid fermentation have been determined by analysis of the infrared, high-resolution electrospray ionization mass spectrometry, 1D and 2D NMR spectra as well as by comparison with literature data. Of the two derivatives, one is a new compound, named concanamycin H, and the other is the known compound, concanamycin G. These compounds are assayed for antibacterial activity, with concanamycins H and G displaying inhibitory activity against Bacillus subtilis (minimum inhibitory concentration = 0.625 µg mL−1).
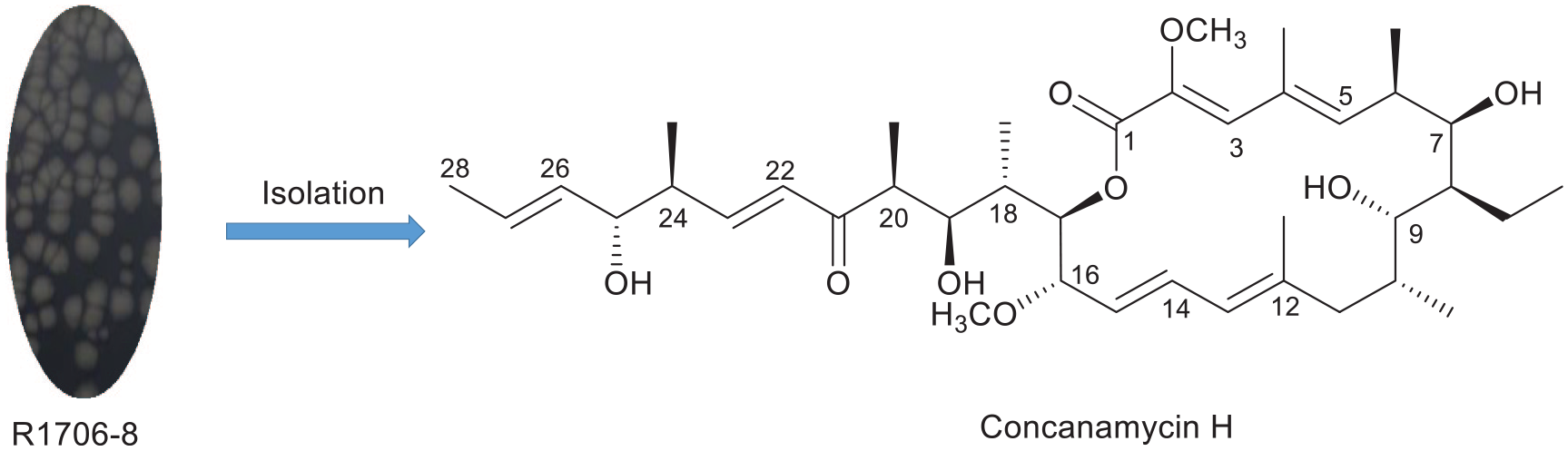
Introduction
To date, seven concanamycin derivatives (A–G) have been isolated from the mycelia of Streptomyces.1–3 These compounds possess antimalarial,3–5 antifungal,6–8 antiviral,9,10 anticancer,11,12 and other biological activities. This family of compounds features an 18-membered macrolide ring 13 and a pyran ring,14,15 but the side-chain structures are rare. Structure–activity relationship studies showed that molecules of the concanamycin family are responsible for potent inhibitory activity. Many researchers have studied the synthesis, 7 semisynthesis, 16 and biosynthesis 17 of these concanamycin derivatives.
In this paper, we report that a sample of the Streptomyces sp. R1706-8, which was isolated from the soil found in the front wall of the honeycomb chamber of the nest of the horned-face bee (Osmia cornifrons), located between Yantai and Weihai in Shandong Province, China, yielded one new compound, concanamycin H (
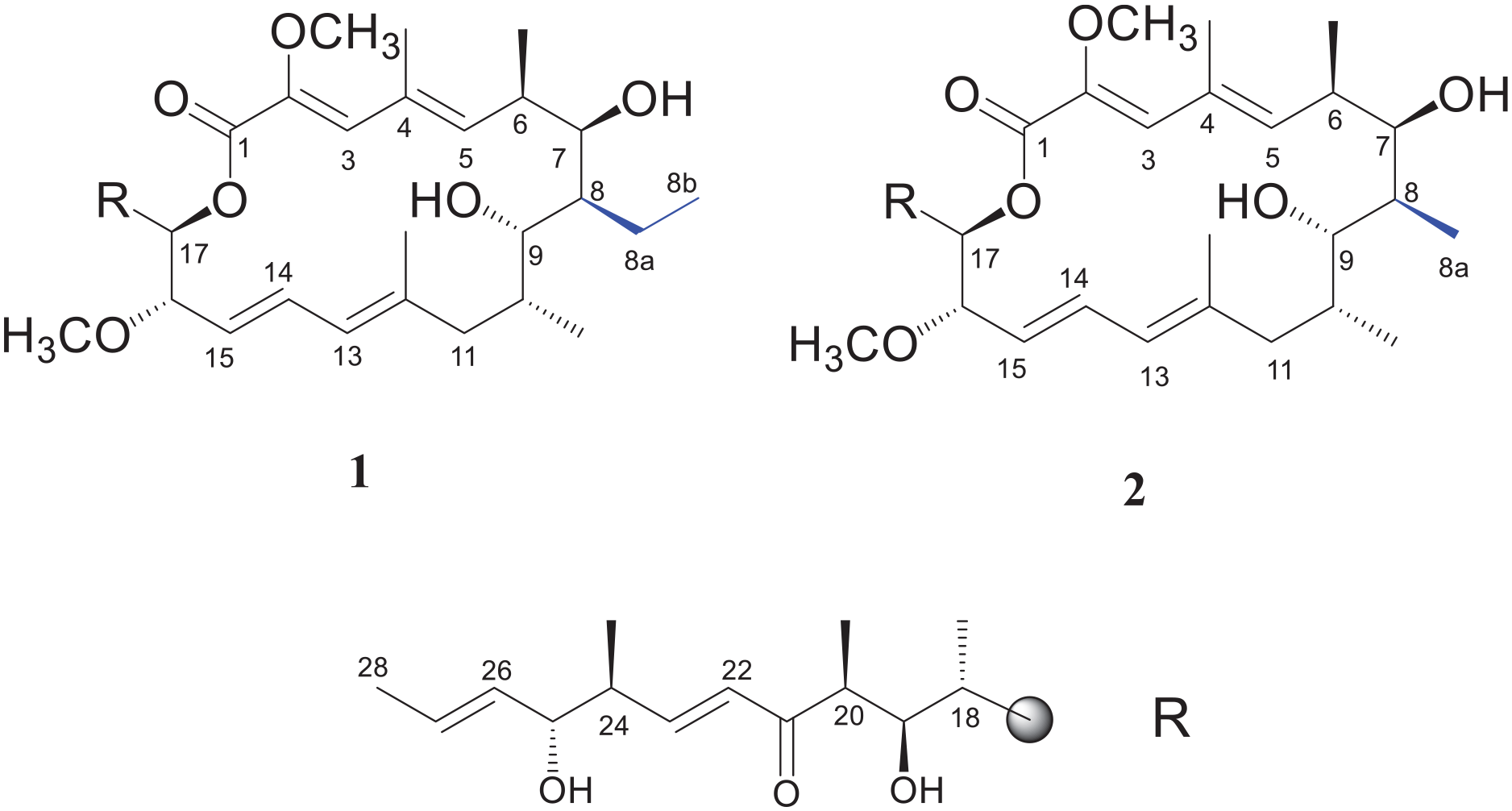
Structures of compounds
Results and discussion
Compound
1H NMR (500 MHz) and 13C NMR (125 MHz) data of compounds

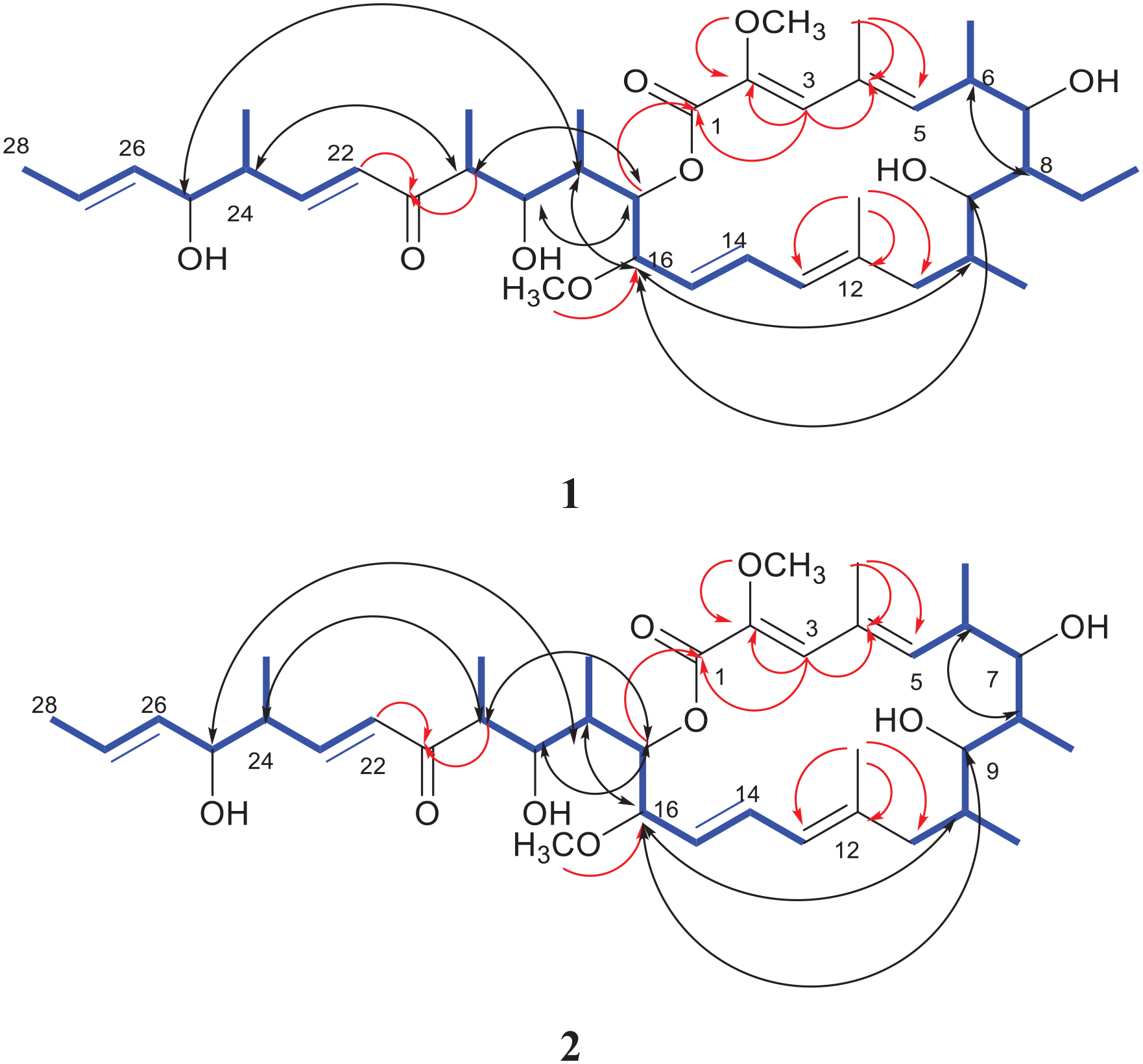
1H−1H COSY (blue bold), key HMBC (red arrows), and NOESY (black arrows) correlations in compounds
Compound
Concanamycin H (
Antibiotic activities of compounds

The diameter of the inhibition zones (mm), including the disk diameter (6 mm), is given as the mean of triplicate experiments.
– denotes none.
The MIC and MBC values of compounds

MIC (μg mL−1): minimum inhibitory concentration.
MBC (μg mL−1): minimal bactericidal concentration.
Conclusion
Two concanamycin derivatives have been isolated from Streptomyces sp. R1706-8, including a new macrocycle named concanamycin H (
Experimental section
General experimental procedures
NMR spectra—including HSQC, HMBC, and COSY—were recorded on a Bruker AVANCE-500 instrument with tetramethylsilane (TMS) as an internal standard (Bruker BioSpin Group, German). NMR spectra recorded on a Bruker Avance III HD spectrometer (500 MHz; Swiss Brook Biersping Co. Let; Switzerland). UV spectra were obtained on a Shimadzu UV-2450 spectrometer (Shimadzu, Japan). HR-ESIMS was conducted on a Bruker APEX II spectrometer (Bruker, Germany). The IR spectra were obtained using a Nicolet IS5 FTIR spectrometer (Thermo Scientific, MA, USA). 1H, 13C, and 2D NMR spectra were recorded at 25 °C on a Bruker ARX 500 NMR spectrometer. GE Sephadex LH-20 (25−100 μm; Pharmacia Biotek, USA) silica gel (200–300 and 300–400 mesh) was used for column chromatography (CC). Silica gel GF254 plates (Qingdao Haiyang Chemical Group Corp., Qingdao, China) were used for TLC.
Media and culture conditions
The culture medium used Gauze’s Synthetic Medium No. 1 that was prepared from soluble starch (20 g), KNO3 (1 g), K2HPO4 (0.5 g), MgSO4·7H2O (0.5 g), NaCl (0.5 g), FeSO4·7 H2O (0.01 g), agar (20 g), and pH = 7.0–8.0, After sterilization by steaming at 121 °C for 31 min and reducing the temperature to room temperature, actinomycete colonies were purified by streaking on the same agar medium. The solid medium was cultured at a constant temperature of 32 °C for around 10 days. Genomic DNA was extracted using a DNA isolation kit (Shanghai Sangon Biotech Co., China). Polymerase chain reaction (PCR) amplification of the 16S ribosomal RNA (rRNA) was performed using primers Fd2 (5’-GAGTTTGATCATGGCTCAG-3’) and 16Sr (5’-TTGCGGGACTTAACCCAAC-AT-3’) and sequenced by Shanghai Sangon Biotech Co., China. The nucleotide sequence was submitted to GenBank (accession numbers KX588720) and compared with reference 16SrRNA gene sequences in databases by BLAST searching. A phylogenetic tree was constructed by the neighbor-joining method using MEGA 5.0 software (Figure 3). Streptomyces sp. R1706-8 was deposited at the Laboratory of Natural Products Chemistry, Shandong University, Weihai, P.R. of China.

Neighbor-joining tree based on nearly complete 16S rRNA gene sequences showing relationships between strains R1706-8 and related members of the family Streptomyces. The percentage numbers (>50 are given) at the nodes are the levels of bootstrap support based on neighbor-joining analyses of 1000 resampled data sets. The tree is rooted with Streptacidiphilus albus NBRC 100918T (NR024930).
Extraction and isolation
The Streptomyces sp. R1706-8 was conserved as a glycerin spore suspension at −80 °C. Streptomyces sp. R1706-8 was grown in Petri dishes containing 50 L of Gauze’s Synthetic Medium No. 1 (20 mL dish−1), (20 g L−1 amylogen, 1 g L−1 KNO3, 0.5 g L−1 NaCl, 0.5 g L−1 K2HPO4·H2O, 0.5 g L−1 MgSO4·H2O, and 0.01 g L−1 FeSO4·H2O adjusted to pH 7.0–8.0). The plates were cultured for 10 days at 32 °C. After fermentation, the culture medium was extracted four times with a mixture of ethyl acetate and methanol (10:1). The extracting solution was concentrated in vacuo to yield the crude extract (4.2 g), which was separated using silica gel CC (50 g, 200–300 mesh) eluting with n-hexane: acetone (10:1, 5:1, 2:1, 1:1, and 0:1) and 100% methanol to give five fractions (Fr.1–Fr.5). Fr.3 (680.0 mg) was passed through a silica gel column eluting with (Fr.1–Fr.4) n-hexane acetone (5:1, 2:1, 1:1, and 0:1) to obtain four sub-fractions (Fr.3.1–3.4). Fr.3.1 (170.0 mg) was purified on a reverse C-18 column, using 60% MeOH/H2O as the solvent to give four further fractions (Fr.3.1.1–Fr.3.1.4). Fraction 3.1.1 (35.0 mg) was fractionated by silica gel CC eluting with CH2Cl2/MeOH (8:1) to yield
Antibacterial assay
The antibacterial activities of compounds
Compound characterization
Compound 1 :
Colorless oil,
Compound 2 :
Colorless oil,
Supplemental Material
sj-docx-1-chl-10.1177_17475198221109161 – Supplemental material for Concanamycin H from the soil actinomycete Streptomyces sp. R1706-8
Supplemental material, sj-docx-1-chl-10.1177_17475198221109161 for Concanamycin H from the soil actinomycete Streptomyces sp. R1706-8 by Guo-Li Li, Hui-Min Qi, Yi-Lin He, Yu-Kai Shen and Tong Shen in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the Lanzhou Jiao Tong University for providing the necessary facilities for carrying out this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NFSC No. 21967015) and the Natural Science Foundation for Young Scholars of Gansu Province (20JR5RA384).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
