Abstract
The aim of this study is to investigate the effects of the components of the medium on nitrilase expression in recombinant Escherichia coli (E. coli), which is applied in the hydrolysis of racemic mandelonitrile (MN) to R-mandelic acid (MA). In addition, the separation of R-MA from the reaction mixture is studied. The glycerol medium is screened, and compositions are optimized in single-factor experiments. The nitrilase activity is 1.96-fold higher than before using the optimal medium containing peptone 15 g/L, yeast extract 12 g/L, NaCl 10 g/L, glycerol 15 g/L, (NH4)2SO4 5 g/L, KH2PO4 10 mM, K2HPO4·3H2O 10 mM, and MgSO4·7H2O 15 mM. During the acid-base extraction of R-MA, the optimal alkaline pH, the alkaline extractant ratio, the optimal acidic pH, the acidic extractant ratio and the amount of activated carbon are 10.0, 1:0.5 (once), 2.50, 1:2 (twice) and 1%, respectively. This study provides a basis for the industrial production of R-MA.

Reaction scheme.
Introduction
Enantiomerically pure α-hydroxy acids are important chiral building blocks for the synthesis of various pharmaceuticals and fine chemicals, and R-mandelic acid (MA) is a key precursor for the synthesis of semi-synthetic penicillins, cephalosporins, antiobesity drugs, and antitumor agents, and serves as a general resolving agent.1,2 Biocatalysts are inexpensive, environmentally responsible and highly efficient alternatives to chemical catalysts, and have attracted increasing attention in industrial settings. 3 Due to the importance of R-MA as a chiral building block, numerous biosynthetic routes for R-MA production have been reported, including examples based on lipase,4,5 esterase, 6 a biocatalyst for the asymmetric reduction of phenyl glyoxylic acid (PGA) to R-mandelic acid, 7 the production of R-MA from styrene, L-phenylalanine, glycerol, or glucose via cascade biotransformations, 8 the synthesis of R-MA and R-MA amide by utilizing recombinant E. coli strains expressing an R-specific oxynitrilase and an arylacetonitrilase, 9 and biotransformation of mandelonitrile with nitrilase.2,10
Among the enzyme sources, nitrilases have been employed frequently as a powerful tool for the enantioselective hydrolysis of racemic mandelonitrile, and the nitrilase-mediated pathway is superior because of excellent enantioselectivity, low-cost starting materials, and the possibility of performing a dynamic kinetic resolution of the racemic substrate, which offers a theoretical yield of 100%. 11 Although examples of biocatalytic synthesis/resolution with nitrilases have been documented,11–14 few studies have optimized the medium in spite of its importance for large-scale preparation.15–18 To improve the industrialization level of R-MA, it is necessary to improve nitrilase production by optimizing the culture medium. At the same time, there is relatively little information on the extraction of R-MA, and it is difficult to treat the waste water that results from separation.19,20
The aim of this study was therefore to investigate the effects of the components of the medium on nitrilase production in recombinant E. coli, and optimization of the acid-base method of R-MA extraction from the reaction mixture, see Scheme 1.
Results and discussion
Selection of the basal fermentation media
In order to evaluate various basal fermentation media, Luria-Bertani (LB), Terrific-Broth (TB), 2×Trytone-Yeast Extract (2×YT), and a glycerol medium were tested. As shown in Figure 1, the highest biomass and nitrilase activity of E. coli obtained in a glycerol medium after induction with isopropyl-β-D-thiogalactoside (IPTG) 0.1 mM for 12 h were 6.25 g/L and 38.87 U/g, respectively. Compared to other media, glycerol increased the biomass of E. coli, and was therefore selected for further optimization.

Effects of four types of culture medium on the cell growth and expression of nitrilase from recombinant E. coli.
Optimization of the media components
To evaluate the effects of media components, various concentrations of tryptone, yeast extract, glycerol, KH2PO4/K2HPO4·3H2O, and MgSO4·7H2O were investigated. As shown in Figure 2(a) and (b), the maximum nitrilase activity of E. coli was observed when the concentrations of tryptone and yeast extract were 15 g/L and 12 g/L, respectively, while the enzyme activity decreased at higher concentrations. Tryptone and yeast extract in the culture medium can promote the growth of bacteria, and with an increase of organic nitrogen source concentration, the weight of bacteria tends to rise. 21 As shown in Figure 2(c), the optimal growth and nitrilase activity of the bacteria was observed at a glycerol concentration of 15 g/L. Phosphate is necessary to sustain the osmotic pressure and growth of microbial cells, while also affecting plasmid stability, biomass and activity of recombinant proteins produced by bacteria. 22 As shown in Figure 2(d), when the concentration of K2HPO4/KH2PO4 was 10 mM, the biomass and nitrilase activity were the highest. It is possible that the concentration of phosphate affected the pH of the medium, which affected the growth and nitrilase activity. Mg2+ is an activator of microbial metabolic processes, and is used as a cofactor in the enzymatic catalytic reaction, thus affecting enzyme activity. Therefore, Mg2+ not only promotes the growth of bacteria, but also the exogenous protein expression when added to the fermentation medium.23,24 As shown in Figure 2(e), the maximum nitrilase activity was observed at a Mg2+ concentration of 15 mM, and there was no obvious variation ranging from 0 to 30 mM.
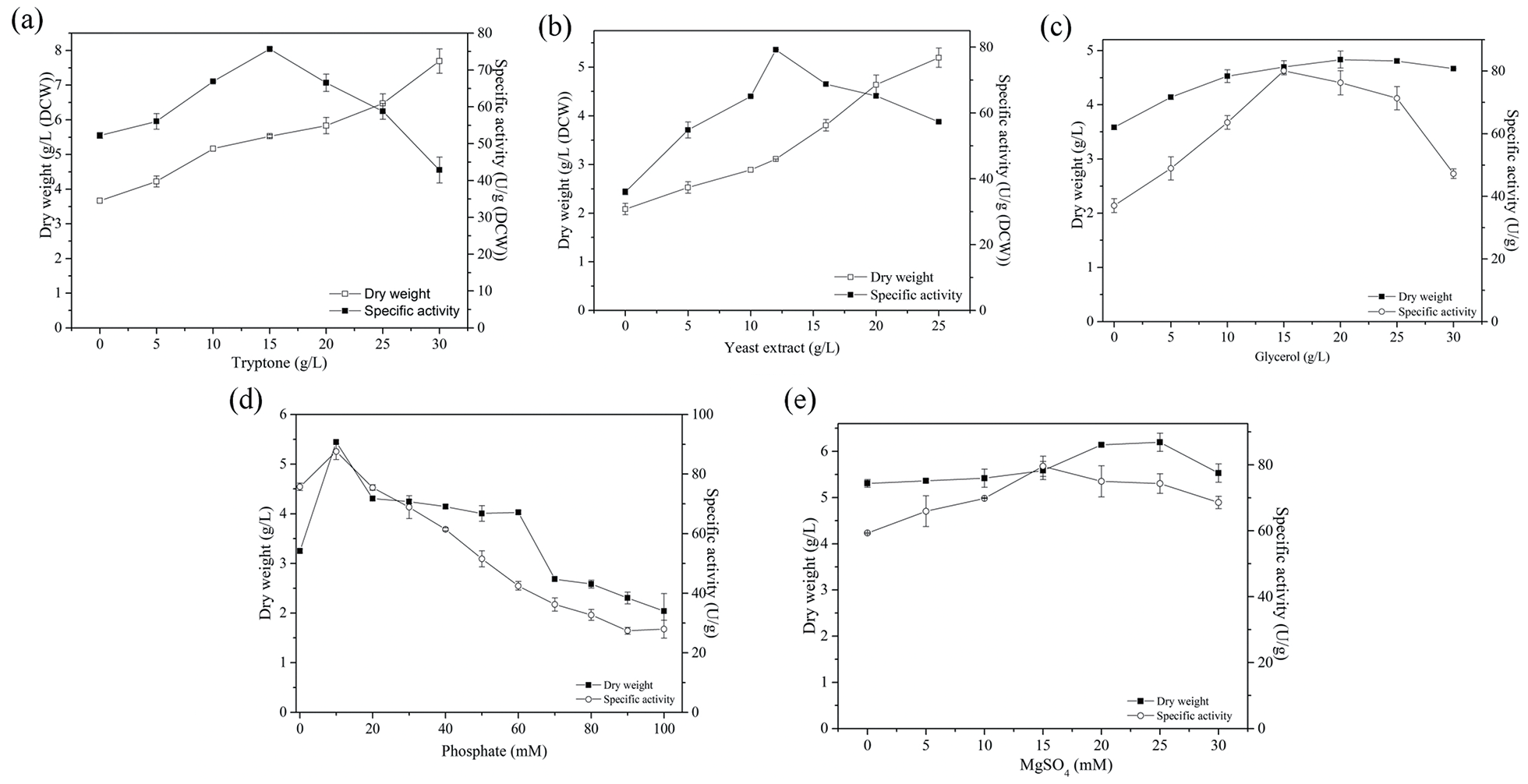
Effects of various concentrations of culture materials on cell growth and expression of nitrilase from recombinant E. coli. (a) Effects of the concentration of tryptone on cell growth and expression of nitrilase. (b) Effects of the concentration of yeast extract on cell growth and expression of nitrilase. (c) Effects of the concentration of glycerol on cell growth and expression of nitrilase. (d) Effects of the concentration of phosphate on cell growth and expression of nitrilase. (e) Effects of the concentration of MgSO4 on cell growth and expression of nitrilase.
The effect of inorganic nitrogen sources (2 g/L) including urea, NH4Cl, (NH4)2SO4, NH4NO3 and NaNO3 on recombinant E. coli cells was evaluated and the results are illustrated in Figure 3. The four tested culture media did not contain inorganic nitrogen, and five different inorganic nitrogen sources were screened. As shown in Figure 3(a), (NH4)2SO4 led to the highest nitrilase activity and promoted the expression of nitrilase when used as the only inorganic nitrogen source. In addition, the concentration of the inorganic nitrogen source that produced the highest nitrilase activity was evaluated in the range from 0 to 20 g/L. As shown in Figure 3(b), different concentrations of (NH4)2SO4 had no significant influence on the nitrilase activity. Considering the effects on the biomass and nitrilase activity, 5 g/L of (NH4)2SO4 was chosen as the optimum concentration. A maximum nitrilase activity of 76 U/g was obtained using the optimum culture medium with a lactose concentration of 10 g/L.
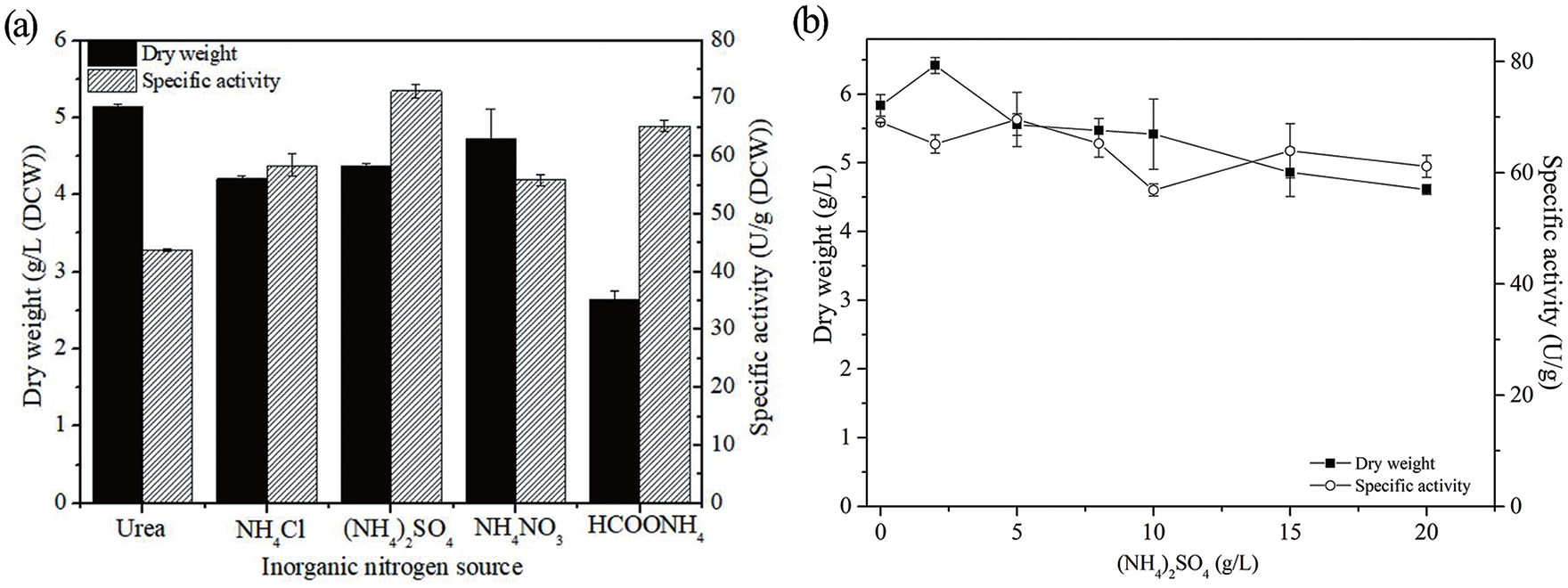
Effects of inorganic nitrogen on cell growth and expression of nitrilase from recombinant E.coli. (a) Effects of different inorganic nitrogen sources on cell growth and expression of nitrilase. (b) Effects of different concentrations of (NH4)2SO4 on cell growth and expression of nitrilase.
Production of nitrilase by recombinant E. coli in a 5-L fermenter
For fermentation in a 5-L fermenter, 60% of the optimum fermentation medium (pH 7.5), was inoculated with 0.2 L of the seed culture of recombinant E. coli, and an agitation cascade was implemented to adjust the dissolved oxygen (DO) concentration to 100% with the agitation speed set at 500 r/min. The fermentation was performed at 37 °C with an aeration rate of 3 m3/h for 2.5–3 h until the OD600 reached 2.0–3.0. Next, the culture was induced by lowering the temperature to 28 °C and adding 0.03 kg of lactose (10 g/L). As shown in Figure 4(a), the pH remained slightly acidic (6.84–5.72), and was not adjusted during the whole fermentation process. The OD600 value and DO% exhibited an inverse correlation. As the OD600 value increased from 8.79 to 20.84, the DO% decreased from 54.3 to 14.3, and then slightly increased to 23.1, which may be caused by autolysis of cells during the late fermentation period. Figure 4(b) shows that the nitrilase activity and dry cell weight reached 86.17 U/g and 13.75 g/L in the 5-L fermenter after fed-batch fermentation for 9 h.

Production of nitrilase by recombinant E. coli in a 5-L fermenter. (a) Profiles of cell growth in the 5-L fermentor. (b) Profiles of cell growth and nitrilase production in batch fermentation by E. coli in the 5-L fermenter.
Effect of the isolation conditions on the yield of R-MA
Effects of basic pH on the isolation of R-MA
The effects of alkaline pH on the isolation of R-MA were investigated in the pH range of 8 to 12 by adding NaOH 10.5 M before separation. The resulting alkaline supernatant was extracted with 1:0.5 butyl acetate (v/v) for 30 min once at room temperature. As shown in Figure S1 (see the supporting information), the optimum pH for the extraction of R-MA was 10.0, and the extraction ratio reached 92.8%. By contrast, the extraction ratios of R-MA at pH 8.0 and 12.0 were only 89.3% and 85.9%, respectively.
Effect of butyl acetate at pH 10.0 on the isolation of R-MA
In order to optimize the removal of unreacted rac-MN from the final reaction solution, the effects of different ratios of reaction mixture and butyl acetate (v/v) on the isolation of R-MA were investigated at the ratios of 1:0.5, 1:1 and 1:2. As shown in Figure S2 (see the supporting information), the optimum ratio for the separation of R-MA was 1:0.5, and the extraction ratio of R-MA reached 93.99%. In contrast, the extraction ratios of R-MA at extractant ratios of 1:1 and 1:2 were only 84.65% and 75.16%, respectively.
Effect of butyl acetate at pH 1.50 on the isolation of R-MA
In order to liberate the free R-MA from its sodium salt, the aqueous phase containing sodium mandelate was adjusted to pH 1.50 using H2SO4, and the effects of different ratios of the aqueous phase and butyl acetate (v/v) on the isolation of R-MA were investigated at the ratios of 1:1, 1:2, 1:3 and 1:4. As shown in Figure S3 (see the supporting information), the optimum ratio for the separation of R-MA was 1:4, resulting in a product loss of 2.22%, which was the lowest of these tested. However, in consideration of industrial production requirements, a ratio of 1:2 was utilized, which resulted in a product loss of 3.73%, being lower than that at a ratio of 1:1 (7.20%), and slightly higher than that at a ratio of 1:3 (2.77%).
Effect of acidic pH and repeated extraction on the isolation of R-MA
To investigate the effects of acidic pH on the extraction of R-MA from its sodium salt, the aqueous phase containing sodium mandelate was adjusted to different pH values ranging from 1.50 to 4.50 with H2SO4, and these acidic aqueous phases were then extracted with 1:2 butyl acetate (v/v) for 30 min (once or twice) at room temperature. As shown in Figure S4 (see the supporting information), the optimum pH for the separation of R-MA was 1.50, with an extraction rate of 90.12% during the first acid extraction. However, after taking salt waste into account in terms of the industrial production, pH 2.5 was used, which resulted in an extraction ratio of 85.41% after the first acid extraction and 97.07% after the second acid extraction.
Effect of temperature on the isolation of R-MA
To determine the efficiency of extraction, the effects of temperature on the extraction of R-MA were investigated. The acidic aqueous phases were extracted once with 1:2 butyl acetate (v/v) for 30 min, at temperatures ranging from 0 to 25 °C. As shown in Figure S5 (see the supporting information), the loss ratios at the tested temperatures were 10.65, 10.55, 10.32, 10.36% and 10.38%, respectively. No effects on the extraction could be observed at temperatures ranging from 0 to 25 °C, indicating that the solubility of R-MA and butyl acetate remained proportional at the tested temperatures.
Effect of the amount of activated carbon on the isolation of R-MA
To determine the efficiency of bleaching, the effects of the amount of activated carbon were investigated. The acidic aqueous phases were extracted once with 1:2 butyl acetate (v/v) for 30 min at room temperature, after which deionized water (100 mL) was added to dissolve the crystals of R-MA obtained from the organic phase through vacuum distillation on a rotary evaporator at 75 °C. Next, different amounts of activated carbon, ranging from 0.1% to 1.0%, were added and the samples were incubated at 50 °C and 150 r/min for 30 min. As shown in Figure S6a (see the supporting information), the amount of activated carbon had no effect on the extraction ratio of R-MA, and according to the color of the R-MA crystals shown in Figure S6b (see the supporting information), 1.0% activated carbon was utilized. After dissolving the R-MA crystals in deionized water, HPLC analysis indicated that the purity of R-MA was >99% and the ee was 99.9%. 2
Conclusions
Single-factor experiments were employed to optimize the medium components for nitrilase production in recombinant E. coli and the extraction conditions for the recovery of R-MA have been studied. The nitrilase activity was improved by 1.96-fold compared to the initial activity under the optimized culture conditions. Besides, the acid-base method for extracting R-MA used in this study was superior to a published method based on the addition of resin 20 because it reduces the amount of industrial waste water and costs, and meets the requirements of industrial production.
Experimental
Chemicals
Authentic rac-MN, R-MA and S-MA were purchased from J&K Chemical Co., Ltd. (Shanghai, China). All other reagents and chemicals were commercially available and of analytical grade.
Strains, media and culture conditions
The recombinant E. coli strain expressing Alcaligenes sp. nitrilase was prepared by following a previous study. 11
To select the fermentation medium for high cell density cultivation of recombinant E. coil, four media were chosen as candidates in this study:
LB medium (g/L): tryptone (10), yeast extract (5), NaCl (10).
2×YT medium (g/L): tryptone (16), yeast extract (10), NaCl (5), adjusted to pH 7.5.
Glycerol medium (g/L): tryptone (20), yeast extract (12), NaCl (10), glycerol (10), KH2PO4 (1.36), K2HPO4·3H2O (2.28), MgSO4·7H2O (0.25), adjusted to pH 7.5.
TB medium (g/L): tryptone (12), yeast extract (24), NaCl (10), glycerol (6), KH2PO4 (2.04), K2HPO4·3H2O (11.4), adjusted to pH 7.0.
Where appropriate, kanamycin (Kan) 50 mg/mL was added.
Expression of recombinant nitrilase
The recombinant E. coli cells were grown at 37 °C in 50 mL of the indicated media with Kan. Production of nitrilase was induced by the addition of IPTG or lactose when the optical density of the culture broth at 600 nm (OD600) reached 0.6, and then the culture was incubated at 28 °C for 20 h and harvested by centrifugation at 9000 r/min for 10 min.
Optimization of media components
Factors that influence the activity of nitrilase, including peptone, yeast extract, glycerol, KH2PO4/K2HPO4·3H2O and MgSO4·7H2O, were investigated. The tested concentrations (w/v, g/L) of peptone, yeast extract, and glycerol were 0–30, 0–25, and 0–30, respectively, and the concentrations (mM) of KH2PO4/K2HPO4·3H2O and MgSO4·7H2O were 0–100, and 0–30, respectively. The inorganic nitrogen source (2 g/L) including urea, NH4Cl, (NH4)2SO4, NH4NO3 and NaNO3 was also investigated, and the tested concentration range (w/v, g/L) of the inorganic nitrogen source that resulted in the highest activity of nitrilase was 0–20.
Fed-batch fermentation of recombinant E. coli in a 5-L fermenter
The recombinant E. coli was cultured at 37 °C and 250 r/min for 14 h in a 500 mL Erlenmeyer flask containing 100 mL of LB medium (pH 7.5). For fermentation in a 5-L fermenter, 60% of the optimal fermentation medium (pH 7.5) was inoculated with 0.2 L of a seed culture of recombinant E.coli, and an agitation cascade was applied to adjust the dissolved oxygen concentration at 100% with an agitation speed of 500 r/min. 25 The fermentation was performed at 37 °C with an aeration rate of 3 m3/h for 2.5–3 h until the OD600 reached 2.0–3.0. Next, the culture was induced by lowering the temperature to 28 °C and adding 0.03 kg of lactose (10 g/L). Samples comprising 10 mL of the culture broth were taken at regular intervals. A 2 mL sample was used for OD600 determination and the remainder was used for enzyme activity assays.
Effect of isolation conditions on the yield of R-MA
Effect of a basic pH on the isolation of R-MA
The reaction supernatant was generated in triplicate, and adjusted to pH 8.0, 10.0 and 12.0 with NaOH 10.5 M, respectively. The supernatant was extracted once with 1:0.5 butyl acetate (v/v) for 30 min at room temperature, after which 100 μL of the aqueous phase was removed to be diluted to 100 times with deionized water and acidified with H2SO4 for HPLC determination. The amount of R-MA without treatment was used as a blank and regarded as 100%.
Effect of butyl acetate on the isolation of R-MA under alkaline condition
The pH of the supernatant was adjusted to 10.0 with NaOH 10.5 M in triplicate, and the parallel samples were extracted once with 1:0.5, 1:1 and 1:2 butyl acetate (v/v) for 30 min at the room temperature, after which 100 μL of the aqueous phase was diluted 100 times with deionized water and acidified with H2SO4 for HPLC determination. The amount of R-MA without treatment was used as a blank and regarded as 100%.
Effect of butyl acetate on the isolation of R-MA under acidic conditions
The pH of the supernatant was adjusted to 10.0 with NaOH 10.5 M, followed by extraction once with 1:0.5 butyl acetate (v/v) for 30 min. The aqueous phase was adjusted to pH 1.50 with H2SO4 in four replicates, which were extracted once with 1:1, 1:2, 1:3 and 1:4 butyl acetate (v/v) for 30 min at the room temperature, after which 200 μL of the aqueous phase was removed and diluted 5 times with deionized water for HPLC determination. The amount of R-MA without treatment was used as a blank and regarded as 100%.
Effects of acidic pH and repeated extraction on the isolation of R-MA
The pH of the supernatant was adjusted to 10.0 with NaOH 10.5 M, followed by extraction once with 1:0.5 butyl acetate (v/v) for 30 min. The aqueous phase was separated into 5 replicates, which were adjusted to pH 1.50, 2.50, 3.00, 3.50 and 4.50 with H2SO4, and these supernatants were extracted once with 1:2 butyl acetate (v/v) for 30 min or twice at room temperature. Next, 300 μL of the aqueous phase was removed and diluted 3 times with deionized water for HPLC determination. The amount of R-MA without treatment was used as a blank and regarded as 100%.
Effect of temperature on the isolation of R-MA
The pH of the supernatant was adjusted to 10.0 with NaOH 10.5 M, followed by extraction once with 1:0.5 butyl acetate (v/v) for 30 min at room temperature. The aqueous phase was adjusted to pH 2.50 with H2SO4 in four replicates, which were extracted once with 1:2 butyl acetate (v/v) for 30 min at the indicated temperatures ranging from 0 to 25 °C. Next, 200 μL of the aqueous phase was removed and diluted 5 times with deionized water for HPLC determination. The amount of R-MA without treatment was used as a blank and regarded as 100%.
Effect of the amount of activated carbon on the isolation of R-MA
The pH of the supernatant was adjusted to 10.0 with NaOH 10.5 M, followed by extraction once with 1:0.5 butyl acetate (v/v) for 30 min at room temperature. The aqueous phase was adjusted to pH 2.50 with H2SO4, and the supernatant was extracted once with 1:2 butyl acetate (v/v) for 30 min at room temperature. Crystals of R-MA were subsequently obtained from the organic phase through vacuum distillation on a rotary evaporator (Adyantag ML, Heidolph, Germany) at 75 °C to remove the butyl acetate which could be recycled. Next, 100 mL of deionized water was added to dissolve the crystals of R-MA and separated into 5 aliquots, to which 0.1%, 0.3%, 0.5%, 0.8%, and 1.0% of activated carbon was added to eliminate the unwanted color at 50 °C for 30 min and 200 r/min. The activated carbon was removed by vacuum filtration (SHB-III, Zhengzhou trade Co., LTD, China), and 100 μL of the supernatant was removed and diluted 2000 times with deionized water for HPLC determination. The amount of R-MA without removal of color was used as a blank and regarded as 100%. Finally, the supernatants were stored at 0 °C to reduce the solubility of R-MA and to precipitate it in the form of crystals.
Analytical methods
Biomass was quantified by measuring the dry cell weight (DCW). A sample comprising 10 mL of the fermentation broth was centrifuged at 8000 r/min for 10 min, after which the pellet was washed twice with distilled water and dried at 100 °C to a constant weight.
The standard enzyme activity assay was carried out using rac-MN 64 mM as the substrate and 0.5 g of wet cells mixed in water. The reaction mixture (total volume 10 mL) was incubated at 35 °C for 0.5 h and the reaction was stopped by adding HCl (final 2 M). Next, 200 μL of the mixture was centrifuged at 12,000 r/min for 3 min and the supernatant was subjected to HPLC analysis on an LC-10AS instrument (Shimadzu, Japan) equipped with a CHIRALCEL OD-RH column (150 mm × 4.60 mm, 5 μm, Diacel Co., Japan) with a solvent system composed of isopropyl alcohol and 0.02% trifluoroacetic acid in a ratio of 1:9 (v/v) and at a flow rate of 0.4 mL/min at 35 °C. The detection wavelength was set to 228 nm. The retention times for S-MA and R-MA were 17.87 and 19.50 min, respectively. 2 One unit of nitrilase activity was defined as the amount of enzyme that produced 1 μmol of R-MA per minute under the conditions described above.
Statistical analysis
If not specifically noted otherwise, all experiments in this study were performed in triplicate. Analysis of variance (ANOVA) was carried out using SAS version 8.1 (SAS Institute Inc., Cary, NC). Least significant differences (LSD) were computed at P < 0.05.
Supplemental Material
sj-doc-1-chl-10.1177_17475198221109155 – Supplemental material for Optimization of the medium composition and product extraction for R-mandelic acid using recombinant Escherichia coli expressing Alcaligenes sp. nitrilase
Supplemental material, sj-doc-1-chl-10.1177_17475198221109155 for Optimization of the medium composition and product extraction for R-mandelic acid using recombinant Escherichia coli expressing Alcaligenes sp. nitrilase by Xinhong Zhang, Fei Chang, Fucheng Zhu, Tao Xu and Yi Zhang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially sponsored by the National Natural Science Foundation of China (No.31800042), the Natural Science Foundation of Anhui Province (No.2008085QB96) and the Natural Science from Education Department of Anhui Province (No. KJ2021A1007).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
