Abstract
Compounds containing a hydantoin moiety are found in several medicines in clinical use. In this research, ethyl- and methyl-[2-(5-benzylidene)-2,4-dioxoimidazolidin-3-yl]acetyl esters are successfully synthesized over four reaction steps using conventional methods. The synthesis begins by subjecting hydantoin to a Knoevenagel condensation reaction with three different benzaldehydes to afford the penultimate products, which are further reacted with ethyl or methyl (bromoacetyl)alaninates, butanoates, valinates, and norvalinates to give the desired products as esters in low to moderate yields.

Introduction
In 2020, Jonnalagadda and co-workers reported that more than 75% of drugs approved by the Food and Drug Administration (FDA) are nitrogen-containing heterocycles.
1
Among the top 25 nitrogen heterocycles in FDA-approved drugs, imidazolidine ranks as number 19 and is found in 11 approved drugs.
2
Imidazolidine-2,4-dione- or hydantoin-containing compounds are known to exhibit a wide range of pharmacological and biological activities and are used to treat various diseases. Both naturally occurring (Figure 1) and synthetic (Figure 2) hydantoin-containing compounds have been reported to possess biological activity. Among naturally occurring hydantoin-containing compounds, hydantocidin (
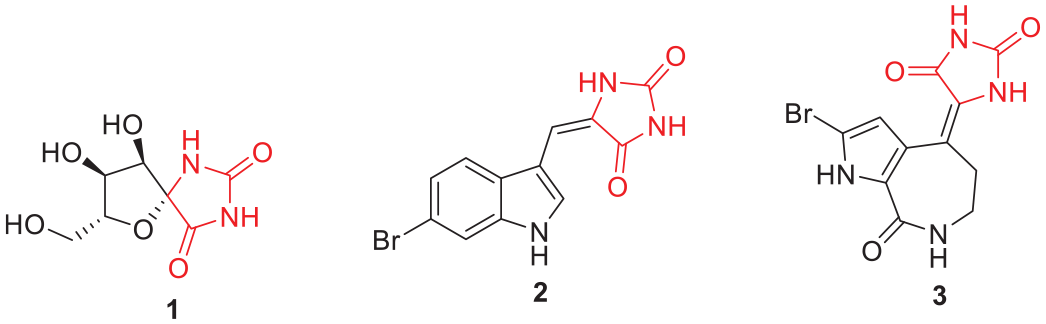
Naturally occurring hydantoin-containing compounds.

Synthetic compounds containing the hydantoin moiety.
Synthetic compounds containing the hydantoin moiety have also been reported to possess different biological activities. Among these are anticonvulsive agents such as phenytoin (
Encouraged by the results obtained in our previous study with 2,4-thiazolidinedione (Figure 3) in the synthesis of 5-benzylidene-2,4-thiazolidinedione esters, 12 we sought to expand this study by replacing a 2,4-thiazolidinedione moiety with a hydantoin moiety for the synthesis of new 5-benzylidene hydantoin esters (Scheme 1).

A schematic diagram of reported and newly synthesized compounds.

Synthesis of ethyl or methyl [2-(5-benzylidene)-2,4-dioxoimidazolidin-3-yl]acetyl esters.
The 5-benzylidene-2,4-thiazolidinedione esters synthesized in our previous work had been tested for their antidiabetic activity on the alpha-glucosidase (unpublished results). Our results indicated that compounds synthesized from para-substituted benzaldehyde had better activity than any other compounds; hence, this research also focused on the usage of 4-methoxybenzaldehyde, 4-methylbenzaldehyde, and the piperonal in the Knoevenagel condensation step.
Unlike the compounds prepared using 2,4-thiazolidinedione, this study starts with the Knoevenagel condensation of three different benzaldehydes with hydantoin as reported by Chung and co-workers.
13
The resulting benzylidene hydantoins
Reagents and conditions include (1) hydantoin, piperidine, reflux, 7 h, (2) SOCl2, EtOH, or MeOH, reflux, (3) bromoacetyl chloride, H2O/DCM (1:1), K2CO3, −10 °C to r.t. 16 h, and (4) KOH, MeOH, reflux, 4 h.
Results and discussion
The synthesis of the desired ethyl or methyl [2-(5-benzylidene)-2,4-dioxoimidazolidin-3-yl]acetyl esters began by reacting commercial hydantoin with different benzaldehydes to give benzylidene hydantoins
Concurrently, compounds
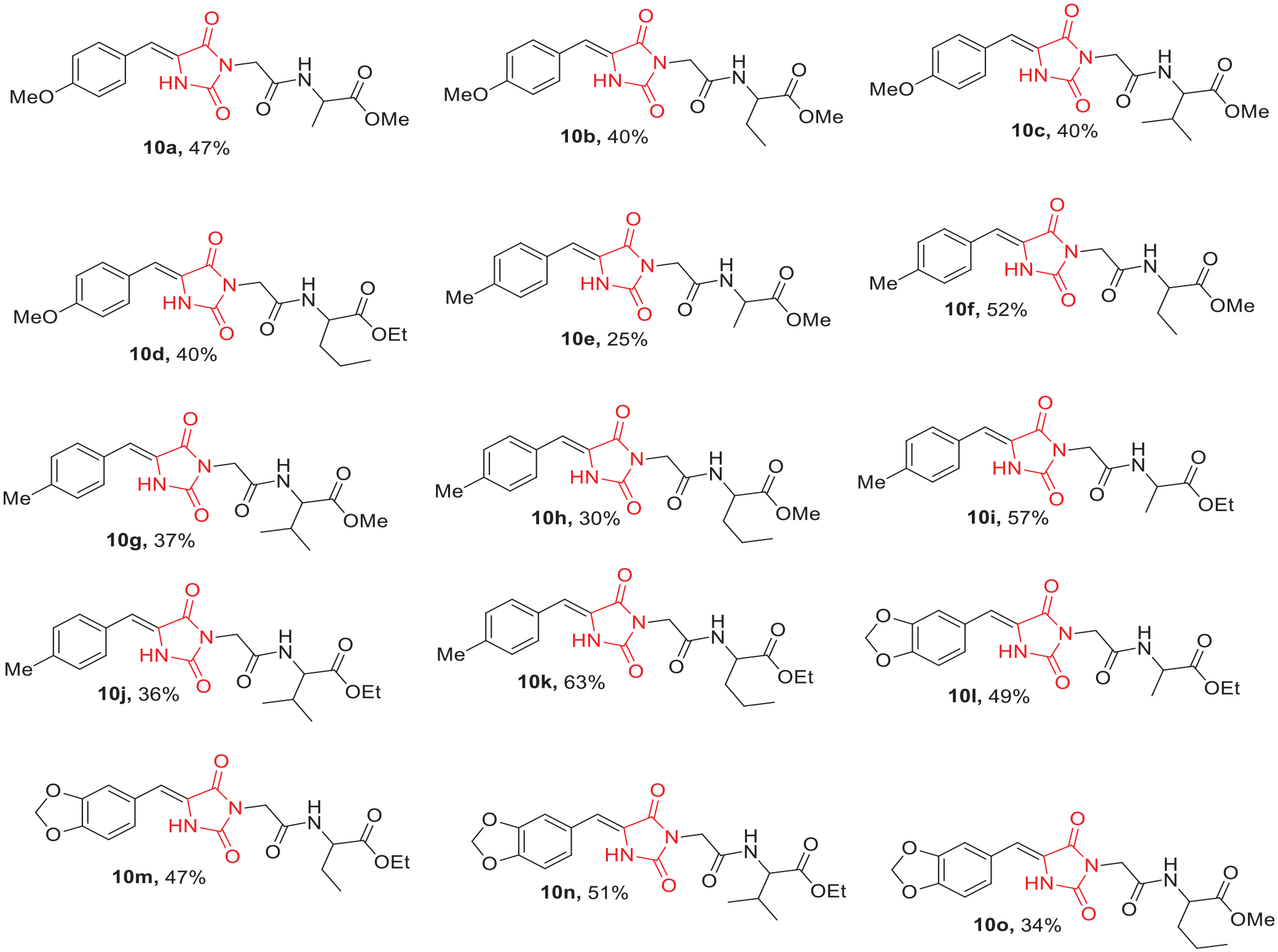
Structures of the synthesized benzylidene-hydantoin esters,
Conclusion
In summary, we have reported the synthesis of 5-benzylidenehydantoin esters. The Knoevenagel condensation of hydantoin and subsequent reactions of protected amino acids with bromoacetyl chloride afforded the desired products. Thus, this protocol provides rapid access to novel benzylidene compounds and could be of some medicinal chemistry value. Therefore, in the future, these compounds will be evaluated for their antidiabetic activities.
Experimental
All reagents used were of analytical grade from Sigma-Aldrich and Fluka. Thin-layer chromatography (TLC) was carried out using Macherey-Nagel Alugram Sil G/UV254 plates, pre-coated with 0.25 mm silica gel 60. Detection was performed under ultraviolet light at 254 nm. 1H NMR (400 MHz) and 13C NMR (100 MHz) spectra were recorded on a Bruker 400 MHz spectrometer using DMSO-d6 or CDCl3 as the solvent and TMS (0.00 ppm) as an internal standard. The values for the chemical shifts are expressed in ppm. The following abbreviations are used: br.s, broad singlet; s, singlet; d, doublets; dd, doublet of doublets; q, quartet; quin, quinet; and m, multiplet; coupling constants (J) are measured in hertz (Hz). Melting points were determined on a Buchi B-540 melting point apparatus using capillary tubes. Infrared spectra were run on a Bruker Platinum 22 Vector Fourier Transform spectrometer (FTIR). Mass spectra (high resolution) were recorded on a Waters GCT mass spectrometer using a Restek Rxi Wintegra Guard column (15 m, 0.25 mm ID, 0.25 µm film thickness). The samples were dissolved in acetonitrile and injected (1 µL) in mode 10:1 at a temperature of 280 °C. The source temperature was 100 °C and the desolvation temperature was set at 300 °C. Helium gas was used as the carrier gas. The software used to control the hyphenated system and to perform all data manipulation was MassLynx 4.1 (SCN 704).
General procedure for the synthesis of (Z)-5-benzylidene hydantoins (8a–c)
These compounds were synthesized from the reaction of hydantoin and appropriate benzaldehydes as described by Chung and co-workers.
13
Compounds
Synthesis of ethyl or methyl (2-bromoacetyl)esters (9a–d)
These compounds were synthesized from their respective amino esters as described by Tshiluka et al. 12 or Barman et al. 14
General procedure for the synthesis of ethyl or methyl 2-(2,4-dioxoimidazolidin-3-yl)acetamido esters (10a–o)
A mixture of arylidene hydantoins
Methyl (2-(5-(4-methoxybenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)alaninate (10a):
The reaction of 5-(4-methoxybenzylidene)hydantoin (
Methyl 2-(2-(5-(4-methoxybenzylidene)-2,4-dioxoimidazolidin-3-yl)acetamido)butanoate (10b):
The reaction of 5-(4-methoxybenzylidene)hydantoin (
Methyl (2-(5-(4-methoxybenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)valinate (10c):
The reaction of 5-(4-methoxybenzylidene)hydantoin (
Ethyl (2-(5-(4-methoxybenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)norvalinate (10d ):
A reaction of 5-(4-methoxybenzylidene)hydantoin (
Methyl (2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)alaninate (10e):
The reaction of 5-(4-methylbenzylidene)hydantoin (
Methyl 2-(2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetamido)butanoate (10f):
The reaction of 5-(4-methylbenzylidene)hydantoin (
Methyl (2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)valinate (10g):
A reaction of 5-(4-methylbenzylidene)hydantoin (
Methyl (2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)norvalinate (10h):
The reaction of 5-(4-methylbenzylidene)hydantoin (
Ethyl (2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)alaninate (10i):
The reaction of 5-(4-methylbenzylidene)hydantoin (
Ethyl (2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)valinate (10j):
The reaction of 5-(4-methylbenzylidene)hydantoin (
Ethyl-(2-(5-(4-methylbenzylidene)-2,4-dioxoimidazolidin-3-yl)acetyl)norvalinate (10k ):
The reaction of 5-(4-methylbenzylidene)hydantoin (
Ethyl-(2-(5-(benzo[d][1,3]dioxol-5-ylmethylene)-2,4-dioxoimidazolidin-3-yl)acetyl)alaninate (10l):
The reaction of 5-(benzo[d][1,3]dioxol-5-ylmethylene)hydantoin
Ethyl-2-(2-(5-(benzo[d][1,3]dioxol-5-ylmethylene)-2,4-dioxoimidazolidin-3-yl)acetamido)butanoate (10m):
The reaction of
Ethyl-(2-(5-(benzo[d][1,3]dioxol-5-ylmethylene)-2,4-dioxoimidazolidin-3-yl)acetyl)valinate (10n):
A reaction of 5-(benzo[d][1,3]dioxol-5-ylmethylene)hydantoin
Methyl-(2-(5-(benzo[d][1,3]dioxol-5-ylmethylene)-2,4-dioxoimidazolidin-3-yl)acetyl)norvalinate (10o):
The reaction of
Supplemental Material
sj-docx-1-chl-10.1177_17475198221104183 – Supplemental material for Synthesis of new 5-benzylidene-hydantoin esters
Supplemental material, sj-docx-1-chl-10.1177_17475198221104183 for Synthesis of new 5-benzylidene-hydantoin esters by Ndivhuwo R Tshiluka, Mpelegeng V Bvumbi, Unarine Tshishonga and Simon S Mnyakeni-Moleele in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the National Research Foundation (NRF) of South Africa and the Sasol Inzalo Foundation for the scholarships provided to Mr N.R.T. and Ms U.T., the THUTHUKA research funds, and the University of Venda Research and Publication Committee (RPC) for sponsoring this research.
Author contributions
Ms U.T. and Mr N.R.T. are the students who executed the project and synthesized reported compounds whereas Dr M.V.B. and Dr S.S.M.-M. are the supervisors of the projects who conceived the projects.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Ms U.T. and Mr N.R.T. were funded by the South African National Research Fund (NRF) for their studies. Mr N.R.T. was also funded by Sasol Foundation for his studies.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
