Abstract
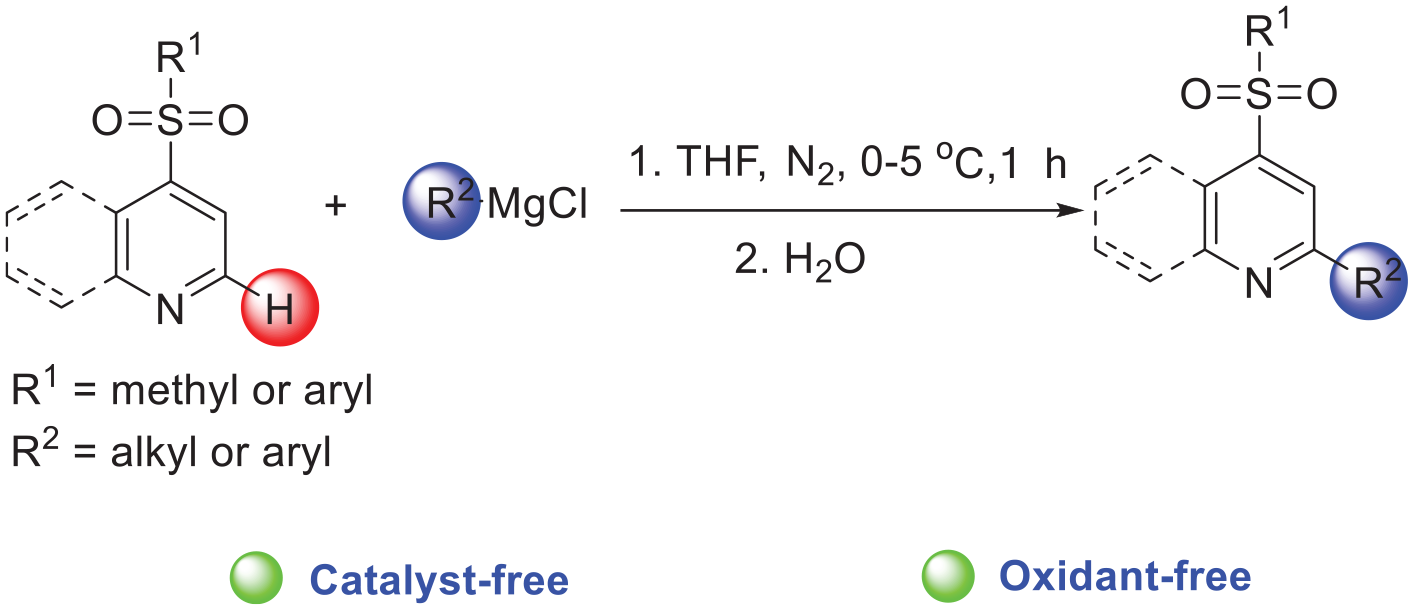
A straightforward synthesis of alkyl-sulfonylpyridines and aryl-sulfonylpyridines is developed by coupling of sulfonylpyridines with the Grignard reagents. The protocol proceeds through a catalyst- and oxidant-free coupling of sulfonylpyridines as substrates via a Chichibabin-type reaction mechanism.
Introduction
Aza-arenes and their derivatives are important naturally occurring substances and building blocks for the synthesis of bioactive compounds with a broad spectrum of activity.1–3 Transition-metal-catalyzed coupling reactions of 2-halogen-substituted aza-arenes with organometallic reagents provide effective protocols for the construction of 2-alkyl-aza-arenes and 2-aryl-aza-arenes (Scheme 1(a)).4–7 However, the use of transition metals usually leads to metal residues and environmental pollution. Recent efforts have been directed toward coupling reactions of pyridinium salts with organometallic reagents via a Chichibabin-type reaction mechanism (addition-oxidation), which avoids the use of transition metals (Scheme 1(b)). For example, Knochel and co-workers reported the addition of BF3-pyridinium salts with magnesium and lithium reagents and subsequent oxidation with chloranil.8,9 Rappenglück et al. have also described a coupling reaction of N-silylpyridinium salts with organomagnesium reagents, 10 in which an oxidant is necessary in order to obtain aza-arenes. In 1986, Furukawa et al. reported the ipso-substitution reaction of 2- and 4-sulfonylpyridines with several aryl and long-chain alkyl Grignard reagents (Scheme 1(c)). 11

Strategies relevant to the coupling of aza-arenes: (a) transition-metal-catalyzed coupling, (b) Chichibabin-type coupling, (c) Furukawa’s work, and (d) this work.
Nonetheless, the Chichibabin-type reaction is still not well-developed. Herein, we demonstrate that sulfonylpyridines react with phenyl and short-chain alkyl Grignard reagents to generate aryl-sulfonylpyridines and alkyl-sulfonylpyridines via a Chichibabin-type reaction mechanism. The reaction requires neither activation of the pyridine by generating the corresponding pyridinium species nor addition of a catalyst or oxidant (Scheme 1(d)).
Results and discussion
Initially, to a mixture of 4-sulfonylpyridine (
Optimization of the reaction conditions. a

Conditions: anhydrous solvent (2 mL), N2.
Yield of isolated product.
No starting material
With optimized reaction conditions in hands, we next set out to investigate the substrate scope. The results are summarized in Scheme 2, which show that the method can be applied to various sulfonylpyridines and Grignard reagents to generate the corresponding products
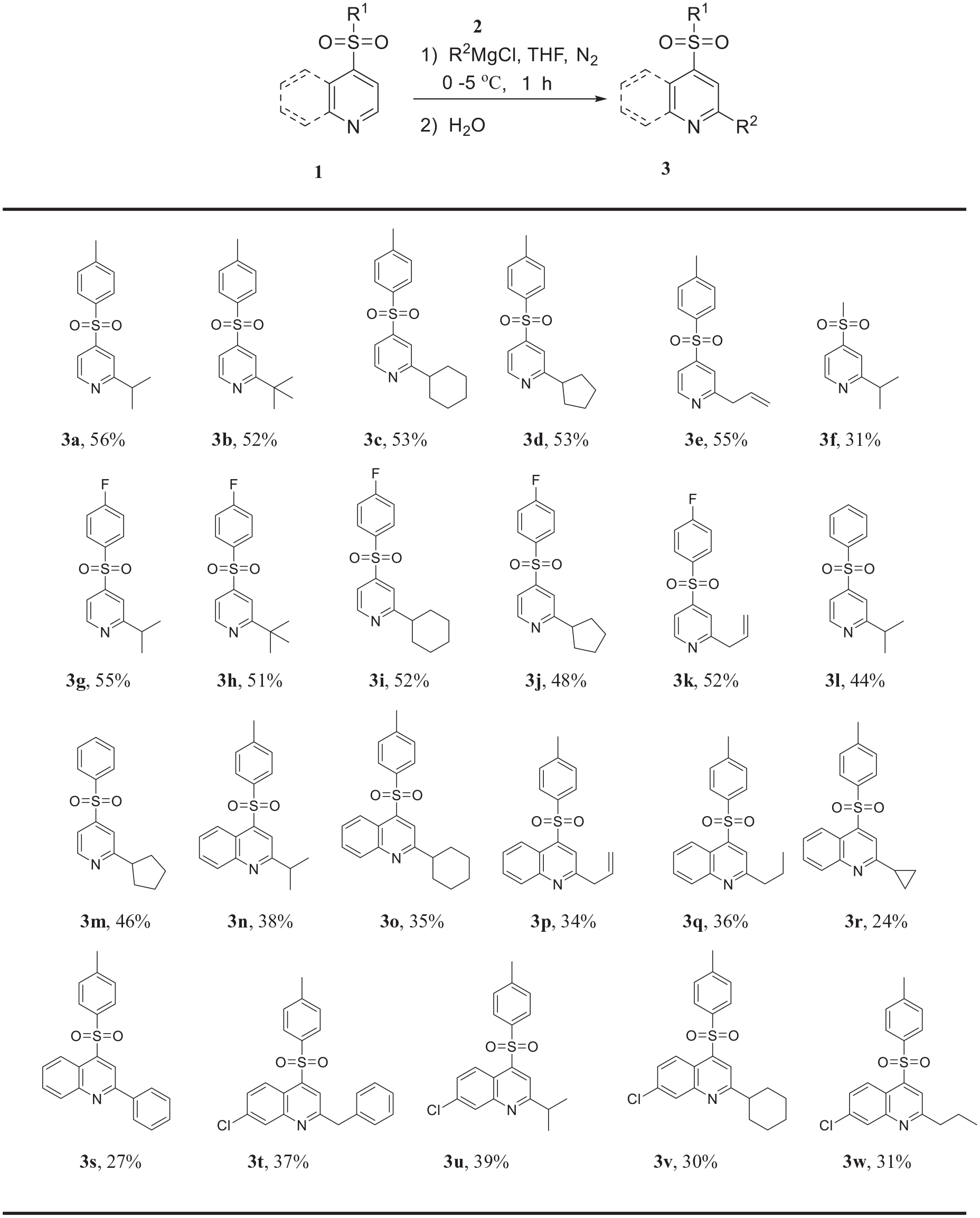
Coupling reactions of sulfonylpyridines with Grignard reagentsa,b.
After examining the substrate scope of the reaction with 4-sulfonylpyridines with different Grignard reagents, we next tried replaced the Grignard reagent with a lithium metal reagent. To our surprise, the obtained result was different from the reactions with Grignard reagents. 2,4-Dibutylpyridine was isolated as the main product when 3 equiv. or more of n-butyllithium were employed (Scheme 3).

Coupling reactions of 4-sulfonylpyridines with a lithium reagent.
Conclusion
In summary, we have developed a straightforward synthesis of 2-alkyl-4-sulfonylpyridines and 2-aryl-4-sulfonylpyridines by coupling 4-sulfonylpyridines with various Grignard reagents in the absence of a catalyst or oxidant.
Experimental
General
All sulfonylpyridines were prepared from chloropyridines and sodium sulfite. 12 Other reagents and solvents were purchased from commercial suppliers. Anhydrous solvents were purified and dried following standard procedures. Purification was generally achieved by flash column chromatography on silica gel (200–300 mesh size). Nuclear magnetic resonance (NMR) spectra were recorded on a 500-MHz Bruker spectrometer. Chemical shifts (1H and 13C) are given in ppm relative to the residual solvent peak (CDCl3, 7.26, 77.0 ppm, respectively). 1H NMR and 13C NMR were recorded using tetramethylsilane (TMS) as the internal standard. Spectroscopic data of known compounds matched with the data reported in the corresponding references. All new compounds were further characterized by high-resolution mass spectrometry (HRMS) (EI) (for further details, see the supporting information).
Experimental section
General procedure A: synthesis of 4-sulfonylpyridines
A round-bottom flask was charged with the: chloropyridine (2 mmol), sodium sulfite (4 mmol), and sodium persulfate (0.4 mmol). 12 Dichloromethane (2 mL) and water (0.8 mL) were added. The reaction mixture was stirred at room temperature. After completion of the reaction, the mixture was poured into saturated sodium carbonate solution (20 mL) and extracted with ethyl acetate (3 × 20 mL). The combined organic layers were dried over anhydrous Na2SO4. After removal of the solvent in vacuo, the obtained residue was purified by column chromatography on silica gel to give the desired product.
General procedure B: synthesis of 2-alkyl-4-sulfonylpyridines
A three-necked round-bottom flask was charged with the: 4-sulfonylpyridine (1 mmol) and anhydrous THF (2 mL). The air atmosphere was replaced with nitrogen and the flask was placed in an ice bath. When the reaction temperature reached 0 °C, we slowly added the Grignard reagent (2.5 mmol) dropwise at 0–5 °C. The reaction mixture was stirred at 0–5 °C for 1 h. After completion of the reaction, the mixture was poured into saturated ammonium chloride solution (20 mL) and extracted with ethyl acetate (3 × 20 mL). The combined organic layers were dried over anhydrous Na2SO4. After removal of the solvent in vacuo, the obtained residue was purified by column chromatography on silica gel to give the desired product.
2-Isopropyl-4-p-toluenesulfonylpyridine (3a ):
Pale yellow oil; 56% yield; 1H NMR (500 MHz, CDCl3): δ 8.69 (d, J = 5.1 Hz, 1H), 7.84 (d, J = 8.4 Hz, 2H), 7.64 (d, J = 1.0 Hz, 1H), 7.51 (dd, J = 5.1, 1.7 Hz, 1H), 7.34 (d, J = 7.9 Hz, 2H), 3.08-3.18 (m, 1H), 2.42 (s, 3H), 1.30 (d, J = 7.0 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 169.5, 150.5, 150.3, 145.2, 137.0, 130.2, 128.1, 117.9, 117.5, 36.6, 22.3, 21.6. HRMS (EI): m/z [M]+ calcd for C15H17NO2S: 275.0980; found: 275.0969.
2-tert-Butyl-4-p-toluenesulfonylpyridine (3b ):
Pale yellow oil; 52% yield; 1H NMR (500 MHz, CDCl3): δ 8.69 (d, J = 5.1 Hz, 1H), 7.83 (d, J = 8.4 Hz, 2H), 7.80 (dd, J = 1.7, 0.8 Hz, 1H), 7.48 (dd, J = 5.1, 1.7 Hz, 1H), 7.32 (d, J = 7.9 Hz, 2H), 2.40 (s, 3H), 1.34 (s, 9H). 13C NMR (126 MHz, CDCl3): δ 171.7, 150.4, 149.9, 144.2, 137.1, 130.2, 128.1, 117.6, 115.7, 38.1, 29.9, 21.6. HRMS (EI): m/z [M]+ calcd for C16H19NO2S: 289.1137; found: 289.1150.
2-Cyclohexyl-4-p-toluenesulfonylpyridine (3c ):
Pale yellow oil; 53% yield; 1H NMR (500 MHz, CDCl3): δ 8.64 (d, J = 5.9 Hz, 1H), 7.80 (d, J = 8.4 Hz, 2H), 7.61 (d, J = 1.1 Hz, 1H), 7.48 (dd, J = 5.1, 1.8 Hz, 1H), 7.29 (d, J = 8.1 Hz, 2H), 2.72 (tt, J = 11.9, 3.4 Hz, 1H), 2.36 (s, 3H), 1.87 (d, J = 11.3 Hz, 2H), 1.80 (d, J = 13.0 Hz, 2H), 1.69 (d, J = 15.9 Hz, 1H), 1.51-1.43 (m, 2H), 1.39-1.30 (m, 2H), 1.24-1.20 (m, 1H). 13C NMR (126 MHz, CDCl3): δ 169.2, 150.4, 150.3, 145.1, 137.0, 130.2, 128.1, 117.9, 117.8, 45.3, 32.6, 26.3, 25.8, 21.6. HRMS (EI): m/z [M]+ calcd for C18H21NO2S: 315.1293; found: 315.1285.
2-Cyclopentyl-4-p-toluenesulfonylpyridine (3d ):
Pale yellow oil; 53% yield; 1H NMR (500 MHz, CDCl3): δ 8.66 (d, J = 5.9 Hz, 1H), 7.82 (d, J = 8.4 Hz, 2H), 7.62 (d, J = 1.6 Hz, 1H), 7.48 (dd, J = 5.1, 1.8 Hz, 1H), 7.32 (d, J = 8.2 Hz, 2H), 3.26-3.16 (m, 1H), 2.39 (s, 3H), 2.09-2.01 (m, 2H), 1.82-1.66 (m, 6H). 13C NMR (126 MHz, CDCl3): δ 168.1, 150.4, 150.2, 145.2, 137.0, 130.2, 128.1, 118.4, 117.7, 48.0, 33.5, 25.8, 21.6. HRMS (EI): m/z [M]+ calcd for C17H19NO2S: 301.1137; found: 301.1128.
2-Allyl-4-p-toluenesulfonylpyridine (3e ):
Pale yellow oil; 55% yield; 1H NMR (500 MHz, CDCl3): δ 8.70 (d, J = 5.1 Hz, 1H), 7.83 (d, J = 8.4 Hz, 2H), 7.64 (s, 1H), 7.54 (dd, J = 5.2, 1.7 Hz, 1H), 7.33 (d, J = 8.0 Hz, 2H), 6.08-5.92 (m, 1H), 5.17 (d, J = 11.2 Hz, 2H), 3.64 (d, J = 6.9 Hz, 2H), 2.41 (s, 3H). 13C NMR (126 MHz, CDCl3): δ 162.4, 150.7, 150.6, 145.3, 136.9, 134.3, 130.2, 128.2, 119.4, 118.2, 118.0, 42.7, 21.6. HRMS (EI): m/z [M]+ calcd for C15H15NO2S: 273.0824; found: 273.0811.
2-Isopropyl-4-methylsulfonylpyridine (3f ):
Pale yellow oil; 31% yield; 1H NMR (500 MHz, CDCl3): δ 8.78 (d, J = 5.0 Hz, 1H), 7.65 (d, J = 1.0 Hz, 1H), 7.57 (dd, J = 5.0, 1.7 Hz, 1H), 3.16 (hept, J = 6.9 Hz, 1H), 3.06 (s, 3H), 1.31 (d, J = 6.9 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 169.9, 150.6,148.7, 117.7, 117.4, 43.8, 36.6, 22.3. HRMS (EI): m/z [M]+ calcd for C9H13NO2S: 199.0667; found: 199.0672.
2-Isopropyl-4-p-fluorobenzenesulfonylpyridine (3g ):
Pale yellow oil; 55% yield; 1H NMR (500 MHz, CDCl3): δ 8.71 (d, J = 5.1 Hz, 1H), 8.01-7.96 (m, 2H), 7.63 (d, J = 1.0 Hz, 1H), 7.51 (dd, J = 5.1, 1.8 Hz, 1H), 7.25-7.16 (m, 2H), 3.12 (sept, J = 6.9 Hz, 1H), 1.29 (d, J = 7.0 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 169.8, 166.0 (d, J = 257.6 Hz), 150.5, 150.0, 136.1 (d, J = 3.2 Hz), 131.0 (d, J = 9.8 Hz), 117.9, 117.5, 117.0 (d, J = 22.9 Hz), 36.6, 22.3. HRMS (EI): m/z [M]+ calcd for C14H14NO2FS: 279.0729; found: 279.0717.
2-tert-Butyl-4-p-fluorobenzenesulfonylpyridine (3h ):
Pale yellow oil; 51% yield; 1H NMR (500 MHz, CDCl3): δ 8.73 (d, J = 5.0 Hz, 1H), 8.01-7.97 (m, 2H), 7.80 (dd, J = 1.7, 0.8 Hz, 1H), 7.50 (dd, J = 5.1, 1.7 Hz, 1H), 7.25-7.19 (m, 2H), 1.35 (s, 9H). 13C NMR (126 MHz, CDCl3): δ 171.9, 165.9 (d, J = 257.3 Hz), 150.0, 149.8, 136.1, 131.0 (d, J = 9.7 Hz), 117.6, 116.9 (d, J = 22.6 Hz), 115.7, 38.1, 29.9. HRMS (EI): m/z [M]+ calcd for C15H16NO2FS: 293.0886; found: 293.0879.
2-Cyclohexyl-4-p-fluorobenzenesulfonylpyridine (3i ):
Pale yellow oil; 52% yield; 1H NMR (500 MHz, CDCl3): δ 8.69 (d, J = 5.1 Hz, 1H), 7.99-7.94 (m, 2H), 7.60 (s, 1H), 7.50 (dd, J = 5.1, 1.8 Hz, 1H), 7.23-7.18 (m, 2H), 2.76 (tt, J = 11.9, 3.4 Hz, 1H), 1.91 (d, J = 13.4 Hz, 2H), 1.84 (d, J = 12.9 Hz, 2H), 1.73 (d, J = 14.5 Hz, 1H), 1.50 (qd, J = 12.4, 3.0 Hz, 2H), 1.38 (qt, J = 12.7, 3.2 Hz, 2H), 1.30-1.24 (m, 1H). 13C NMR (126 MHz, CDCl3): δ 168.9, 165.9 (d, J = 257.3 Hz), 150.5, 149.9, 136.1 (d, J = 3.1 Hz), 131.0 (d, J = 9.8 Hz), 117.8, 117.8, 116.9 (d, J = 22.6 Hz), 46.6, 32.6, 26.3, 25.8. HRMS (EI): m/z [M]+ calcd for C17H18NO2FS: 319.1042; found: 319.1031.
2-Cyclopentyl-4-p-fluorobenzenesulfonylpyridine (3j ):
Pale yellow oil; 48% yield; 1H NMR (500 MHz, CDCl3): δ 8.68 (d, J = 5.1 Hz, 1H), 7.99-7.94 (m, 2H), 7.62 (d, J = 1.2 Hz, 1H), 7.48 (dd, J = 5.1, 1.8 Hz, 1H),7.24-7.16 (m, 2H), 3.21 (p, J = 8.3 Hz, 1H), 2.09-2.01 (m, 2H), 1.82-1.64 (m, 6H). 13C NMR (126 MHz, CDCl3): δ 168.3, 165.9 (d, J = 257.3 Hz), 150.6, 149.7, 136.1, 131.0 (d, J = 9.8 Hz), 118.4, 117.7, 116.9 (d, J = 22.6 Hz), 47.4, 33.5, 25.8. HRMS (EI): m/z [M]+ calcd for C16H16NO2FS: 305.0886; found: 305.0879.
2-Allyl-4-p-fluorobenzenesulfonylpyridine (3k ):
Pale yellow oil; 52% yield; 1H NMR (500 MHz, CDCl3): δ 8.70 (d, J = 5.1 Hz, 1H), 7.98-7.93 (m, 2H), 7.62 (s, 1H), 7.54 (dd, J = 5.2, 1.7 Hz, 1H), 7.23-7.14 (m, 2H), 6.06-5.90 (m, 1H), 5.16 (d, J = 14.2 Hz, 2H), 3.63 (d, J = 6.9 Hz, 2H). 13C NMR (126 MHz, CDCl3): δ 166.0 (d, J = 257.5 Hz), 162.6, 150.8, 150.1, 135.9 (d, J = 3.3 Hz), 134.2, 131.0 (d, J = 9.8 Hz), 119.4, 118.1, 118.1, 117.0 (d, J = 22.6 Hz), 42.7. HRMS (EI): m/z [M]+ calcd for C14H12NO2FS: 277.0573; found: 277.0553.
2-Isopropyl-4-benzenesulfonylpyridine (3l ):
Pale yellow oil; 44% yield; 1H NMR (500 MHz, CDCl3): δ 8.71 (d, J = 5.1 Hz, 1H), 7.97 (d, J = 8.2 Hz, 2H), 7.66 (s, 1H), 7.64 (t, J = 7.5 Hz, 1H), 7.58-7.52 (m, 3H), 3.15 (hept, J = 6.9 Hz, 1H), 1.31 (d, J = 6.9 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 169.6, 150.3, 150.2, 140.0, 134.0, 129.6, 128.1, 118.1, 117.6, 35.9, 22.3. HRMS (EI): m/z [M]+ calcd for C14H15NO2S: 261.0824; found: 261.0820.
2-Cyclopentyl-4-benzenesulfonylpyridine (3m ):
Pale yellow oil; 46% yield; 1H NMR (500 MHz, CDCl3): δ 8.67 (d, J = 5.1 Hz, 1H), 7.94 (d, J = 7.2 Hz, 2H), 7.62 (s, 1H), 7.59 (t, J = 7.4 Hz, 1H), 7.53-7.50 (m, 2H), 7.50-7.48 (m, 1H), 3.21 (p, J = 8.3 Hz, 1H), 2.09-1.99 (m, 2H), 1.83-1.75 (m, 2H), 1.75-1.69 (m, 2H), 1.68-1.59 (m, 2H). 13C NMR (126 MHz, CDCl3): δ 168.7, 150.5, 149.8, 140.0, 134.0, 129.5, 128.0, 118.5, 117.8, 48.7, 33.5, 25.8. HRMS (EI): m/z [M]+ calcd for C16H17NO2S: 287.0980; found: 287.0963.
2-Isopropyl-4-p-toluenesulfonylquinoline (3n ):
Pale yellow solid; 38% yield; 1H NMR (500 MHz, CDCl3): δ 8.56 (d, J = 8.5 Hz, 1H), 8.15 (s, 1H), 8.09 (d, J = 7.1 Hz, 1H), 7.87 (d, J = 8.4 Hz, 2H), 7.65 (t, J = 8.4 Hz, 1H), 7.52 (t, J = 7.1 Hz, 1H), 7.24 (d, J = 7.7 Hz, 2H), 3.33 (hept, J = 6.9 Hz, 1H), 2.29 (s, 3H), 1.41 (d, J = 6.9 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 167.2, 149.1, 145.4, 144.9, 137.5, 130.1, 130.1, 130.0, 127.8, 127.7, 124.0, 120.8, 120.0, 37.3, 22.2, 21.5. HRMS (EI): m/z [M]+ calcd for C19H19NO2S: 325.1137; found: 325.1139.
2-Cyclohexyl-4-p-toluenesulfonylquinoline (3o ):
Pale yellow solid; 35% yield; m.p. 188–190 °C (lit. 13 ); 1H NMR (500 MHz, CDCl3): δ 8.54 (dd, J = 8.7, 1.4 Hz, 1H), 8.14 (s, 1H), 8.09 (d, J = 9.8 Hz, 1H), 7.87 (d, J = 8.4 Hz, 2H), 7.68 (t, J = 7.7 Hz, 1H), 7.53 (t, J = 8.4 Hz, 1H), 7.28 (d, J = 8.6 Hz, 2H), 3.00 (tt, J = 12.0, 3.5 Hz, 1H), 2.35 (s, 3H), 2.04 (d, J = 14.2 Hz, 2H), 1.91 (dt, J = 13.0, 3.4 Hz, 2H), 1.79 (d, J = 14.5 Hz, 1H), 1.68 (qd, J = 12.6, 3.4 Hz, 2H), 1.51-1.42 (m, 2H), 1.38-1.32 (m, 1H). 13C NMR (126 MHz, CDCl3): δ 166.4, 149.2, 145.2, 144.8, 137.7, 130.1, 130.0, 130.0, 127.8, 127.6, 124.0, 120.8, 120.3, 48.0, 32.5, 26.4, 25.9, 21.6. HRMS (EI): m/z [M]+ calcd for C22H23NO2S: 365.1450; found: 365.1436.
2-Allyl-4-p-toluenesulfonylquinoline (3p ):
Pale yellow solid; 34% yield; 1H NMR (500 MHz, CDCl3): δ 8.58 (d, J = 8.6 Hz, 1H), 8.13 (s, 1H), 8.11 (d, J = 9.5 Hz, 1H), 7.88 (d, J = 8.4 Hz, 2H), 7.74-7.69 (m, 1H), 7.57 (t, J = 7.1 Hz, 1H), 7.29 (d, J = 8.2 Hz, 2H), 6.22-6.10 (m, 1H), 5.24 (d, J = 7.2 Hz, 2H), 3.86 (d, J = 6.8 Hz, 2H), 2.36 (s, 3H). 13C NMR (126 MHz, CDCl3): δ 163.2, 149.2, 145.4, 145.0, 137.4, 134.4, 130.3, 130.0, 130.0, 128.0, 127.9, 124.1, 121.6, 120.8, 118.2, 43.1, 21.6. HRMS (EI): m/z [M]+ calcd for C19H17NO2S: 323.0980; found: 323.0981.
2-n-Propyl-4-p-toluenesulfonylquinoline (3q ):
Pale yellow solid; 36% yield; 1H NMR (500 MHz, CDCl3): δ 8.57 (d, J = 9.9 Hz, 1H), 8.11 (s, 1H), 8.09 (d, J = 1.0 Hz, 1H), 7.88 (d, J = 8.4 Hz, 2H), 7.70 (t, J = 8.4 Hz, 1H), 7.59-7.53 (m, 1H), 7.29 (d, J = 8.6 Hz, 2H), 3.08-3.02 (m, 2H), 2.35 (s, 3H), 1.94-1.84 (m, 2H), 1.03 (t, J = 7.3 Hz, 3H). 13C NMR (126 MHz, CDCl3): δ 162.6, 149.3, 145.1, 144.9, 137.5, 130.2, 130.0, 130.0, 127.9, 127.7, 124.0, 121.6, 120.7, 40.5, 22.9, 21.6, 13.9. HRMS (EI): m/z [M]+ calcd for C19H19NO2S: 325.1137; found: 325.1143.
2-Cyclopropyl-4-p-toluenesulfonylquinoline (3r ):
Pale yellow solid; 24% yield; 1H NMR (500 MHz, CDCl3): δ 8.51 (d, J = 8.7 Hz, 1H), 8.04 (s, 1H), 7.98 (d, J = 10.1 Hz, 1H), 7.88 (d, J = 8.4 Hz, 2H), 7.67-7.61 (m, 1H), 7.51-7.45 (m, 1H), 7.28 (d, J = 8.0 Hz, 2H), 2.35 (s, 3H), 2.33-2.27 (m, 1H), 1.27-1.23 (m, 2H), 1.18-1.13 (m, 2H). 13C NMR (126 MHz, CDCl3): δ 163.3, 149.4, 144.9, 144.6, 137.6, 130.1, 130.0, 129.8, 127.9, 127.1, 124.0, 120.6, 120.5, 21.6, 18.3, 11.3. HRMS (EI): m/z [M]+ calcd for C19H17NO2S: 323.0980; found: 323.0981.
2-Phenyl-4-p-toluenesulfonylquinoline (3s ):
Pale yellow solid; 27% yield; m.p. 170–173 °C (lit. 13 ); 1H NMR (500 MHz, CDCl3): δ 8.71 (s, 1H), 8.61 (d, J = 8.7 Hz, 1H), 8.27-8.23 (m, 3H), 7.91 (d, J = 8.4 Hz, 2H), 7.76 (t, J = 8.4 Hz, 1H), 7.63-7.59 (m, 1H), 7.59-7.55 (m, 2H), 7.53 (t, J = 7.2 Hz, 1H), 7.31 (d, J = 8.2 Hz, 2H), 2.38 (s, 3H). 13C NMR (126 MHz, CDCl3): δ 156.8, 149.6, 145.8, 145.0, 138.1, 137.5, 130.8, 130.5, 130.3, 130.1, 129.1, 128.3, 127.9, 127.6, 124.1, 121.1, 119.3, 21.6.
2-Benzyl-4-p-toluenesulfonyl-7-chloroquinoline (3t ):
Pale yellow solid; 37% yield; 1H NMR (500 MHz, CDCl3): δ 8.54 (d, J = 9.1 Hz, 1H), 8.16 (d, J = 2.1 Hz, 1H), 7.97 (s, 1H), 7.78 (d, J = 8.4 Hz, 2H), 7.54 (dd, J = 9.1, 2.2 Hz, 1H), 7.37-7.32 (m, 2H), 7.32-7.26 (m, 5H), 4.40 (s, 2H), 2.39 (s, 3H). 13C NMR (126 MHz, CDCl3): δ 162.3, 149.6, 145.8, 145.2, 137.8, 136.9, 136.5, 130.1, 129.2, 129.1, 128.9, 128.9, 127.9, 127.0, 125.4, 121.8, 119.1, 45.4, 21.6. HRMS (EI): m/z [M]+ calcd for C23H18ClNO2S: 407.0747; found: 407.0750.
2-Isopropyl-4-p-toluenesulfonyl-7-chloroquinoline (3u ):
Pale yellow solid; 39% yield; 1H NMR (500 MHz, CDCl3): δ 8.50 (d, J = 9.1 Hz, 1H), 8.12 (d, J = 2.2 Hz, 1H), 8.09 (s, 1H), 7.84 (d, J = 8.4 Hz, 2H), 7.50 (dd, J = 9.1, 2.2 Hz, 1H), 7.31 (d, J = 7.8 Hz, 2H), 3.33 (hept, J = 6.9 Hz, 1H), 2.39 (s, 3H), 1.42 (d, J = 6.9 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 168.6,149.6, 145.6, 145.1, 137.3, 136.2, 130.1, 129.0, 128.6, 127.8, 125.3, 120.1, 119.2, 37.3, 22.1, 21.6. HRMS (EI): m/z [M]+ calcd for C19H18ClNO2S: 359.0747; found: 359.0731.
2-Cyclohexyl-4-p-toluenesulfonyl-7-chloroquinoline (3v ):
Pale yellow solid; 30% yield; m.p. 114–116 °C (lit. 12 ); 1H NMR (500 MHz, CDCl3): δ 8.50 (d, J = 9.1 Hz, 1H), 8.09 (s, 1H), 8.09 (s, 1H), 7.84 (d, J = 8.4 Hz, 2H), 7.48 (dd, J = 9.1, 2.2 Hz, 1H), 7.29 (d, J = 8.1 Hz, 2H), 2.97 (tt, J = 11.9, 3.5 Hz, 1H), 2.36 (s, 3H), 2.02 (d, J = 13.7 Hz, 2H), 1.91 (d, J = 16.7 Hz, 2H), 1.80 (s, 1H), 1.66 (d, J = 15.9 Hz, 2H), 1.46 (d, J = 12.9 Hz, 2H), 1.35 (d, J = 16.4 Hz, 1H). 13C NMR (126 MHz, CDCl3): δ 167.8, 149.7, 145.4, 145.1, 137.4, 136.1, 130.1, 129.1, 128.4, 127.8, 125.3, 120.5, 119.2, 47.3, 32.4, 26.3, 25.9, 20.8. HRMS (EI): m/z [M]+ calcd for C22H22ClNO2S: 399.1060; found: 399.1042.
2-n-Propyl-4-p-toluenesulfonyl-7-chloroquinoline (3w ):
Pale yellow solid; 31% yield; 1H NMR (500 MHz, CDCl3): δ 8.51 (d, J = 9.1 Hz, 1H), 8.09 (d, J = 2.2 Hz, 1H), 8.05 (s, 1H), 7.84 (d, J = 8.4 Hz, 2H), 7.50 (dd, J = 9.1, 2.2 Hz, 1H), 7.30 (d, J = 8.5 Hz, 2H), 3.05-3.00 (m, 2H), 2.38 (s, 3H), 1.92-1.83 (m, 2H), 1.03 (t, J = 7.3 Hz, 3H). 13C NMR (126 MHz, CDCl3): δ 164.0, 150.8, 145.3, 145.2, 137.2, 136.3, 130.1, 128.9, 128.6, 127.8, 125.4, 121.7, 119.1, 41.1, 22.7, 21.6, 13.9. HRMS (EI): m/z [M]+ calcd for C19H18ClNO2S: 359.0747; found: 359.0749.
General procedure C: synthesis of 2,6-di-n-butylpyridine
A three-necked round-bottom flask was charged with the: 4-sulfonylpyridine (1 mmol) and anhydrous THF (2 mL). The air atmosphere was replaced with nitrogen and the flask was cooled using liquid nitrogen. When the reaction temperature dropped to −30 °C, n-butyl lithium reagent (3 mmol) was slowly added dropwise to the flask at −30 °C. The reaction mixture was stirred at −30 °C for 1 h. After completion of the reaction, the mixture was poured into saturated ammonium chloride solution (20 mL) and extracted with ethyl acetate (3 × 20 mL). The combined organic layers were dried over anhydrous Na2SO4. After removal of the solvent in vacuo, the obtained residue was purified by column chromatography on silica gel to give the desired product.
2,4-Di-n-butylpyridine (6 ):
Pale yellow oil; 1H NMR (500 MHz, CDCl3): δ 8.34 (d, J = 5.0 Hz, 1H), 6.92 (s, 1H), 6.87 (d, J = 3.5 Hz, 1H), 2.75-2.69 (m, 2H), 2.55-2.49 (m, 2H), 1.70-1.63 (m, 2H), 1.59-1.52 (m, 2H), 1.39-1.27 (m, 4H), 0.89 (td, J = 7.4, 3.1 Hz, 6H). 13C NMR (126 MHz, CDCl3): δ 162.1, 151.9, 148.7, 122.7, 121.0, 37.9, 34.9, 32.4, 32.0, 22.4, 22.2, 13.8, 13.7.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221103502 – Supplemental material for A straightforward coupling of 4-sulfonylpyridines with Grignard reagents
Supplemental material, sj-docx-1-chl-10.1177_17475198221103502 for A straightforward coupling of 4-sulfonylpyridines with Grignard reagents by Liu-Yi Song, Meng-Ke Chen, Jian Wang and Jing-Hua Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (no. 22078300) and the Basic Scientific Research Funds of the Department of Education of Zhejiang Providence (no. KYQN202006).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

