Abstract
A facile method for oxidation of primary benzylic alcohols to the corresponding aldehydes is reported using NaNO3/P2O5 under high-speed ball-milling conditions. This approach is clean, efficient, and exhibits broad functional group compatibility.

Introduction
The selective oxidation of alcohols to the corresponding aldehydes/ketones plays a central role in the synthetic chemist’s toolkit and has received increasing attention in recent years.1,2 Many reagents have been developed for oxidation of benzylic alcohols to the corresponding carbonyl compounds, such as chromium reagents,3,4 manganese oxidizing reagents,5–8 2,2,6,6-tetramethylpiperidyl-1-oxy (TEMPO)-related reagents,9–11 and nitrite- and nitrate-related reagents.12–16 However, these reactions usually suffer from disadvantages, including the application of heavy-metal-based reagents, harsh reaction conditions, long reaction times, and toxic solvents. With ever-increasing environmental concerns, the development of a general and efficient synthetic protocol for the oxidation of benzylic alcohols is still highly desirable.
Mechanochemical organic methodologies have recently attracted significant attention as a powerful tool for organic chemists.17–25 Mechanochemistry is generally performed under solvent-free conditions, and as the reagent concentrations are high, this often leads to accelerated reactions and alterations in product selectivity, typically in favor of the discovery of new chemical transformations. In addition, the technique also covers multiple aspects of green chemistry, which can avoid bulk solvents and reduce environmental pollution. 26
In our previous work in the area of mechanochemical synthesis, the combination of Bi(NO3)3·5H2O and MgSO4 was successfully employed for the nitration of aromatic compounds. 27 Recently, we reported that the combination of Fe(NO3)3·9H2O and P2O5 could convert deactivated arenes into the corresponding nitrated arenes in excellent yields under high-speed ball-milling (HSBM) conditions. 28 We proposed that the N2O4/NO2, which was produced in situ from the reaction of nitrates and the auxiliary, should play a major role in these mechanochemical nitrations. Since N2O4/NO2 species are also well-known as oxidants,29–31 we set out to explore the oxidative nature of the combination of nitrates and auxiliary. In this paper, a combination of NaNO3 and P2O5 is employed as a simple, stable, and inexpensive oxidant for the oxidation of primary benzylic alcohols to aldehydes under HSBM conditions.
Results and discussion
Initially, 4-methylbenzyl alcohol (
Optimization of the reaction conditions. a

N.D.: not detected.
The side products are known compounds and were determined by 1H NMR (see ESI for details).
Reaction conditions:
Molar ratio of
Yields were determined by 1H NMR analysis of the crude reaction mixture using dibromomethane as an internal standard.
We noticed that the nitrate/P2O5 molar ratio had a strong influence on the outcome of the reactions during our exploration of these HSBM reactions.28,32 To obtain the best yield of the product, the HSBM reaction conditions were further optimized by adjusting the molar ratio of NaNO3 and P2O5 in relation to
The conversion of
Oxidation of

Reaction conditions:
Yields were determined by 1H NMR analysis of the crude reaction mixture using dibromomethane as an internal standard.
With optimized reaction conditions in hand, we next explored the scope of our oxidation system on a series of benzylic primary alcohols. Benzyl alcohol (
Oxidation of different primary aromatic alcohols to the corresponding aldehydes. a

N.D.: not detected.
Reaction conditions: primary aromatic alcohol
Yields were determined by 1H NMR analysis of the crude reaction mixture using dibromomethane as an internal standard.
Reaction was run for 4 h.
Many hydrolyzable and oxidatively sensitive functional groups such as amido, ester, and formyl are tolerated under these ball-milling conditions, indicating that our NaNO3/P2O5 system is an effective combination for obtaining aldehydes, and can be used widely for the oxidation of various substituted benzylic primary alcohols. In addition, 2-naphthalenemethanol (
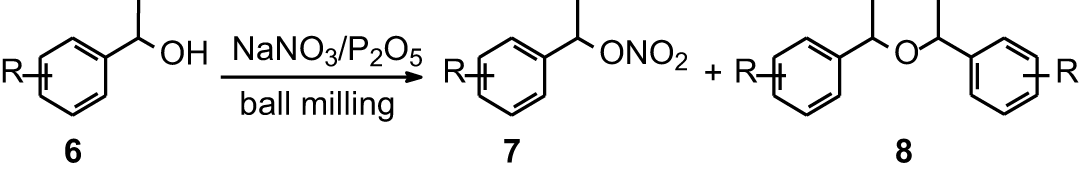
We also examined the properties of NaNO3/P2O5 as an oxidant reagent for secondary benzylic alcohols. In contrast to the behavior of primary benzylic alcohols,
Oxidation of different secondary aromatic benzylic alcohols. a
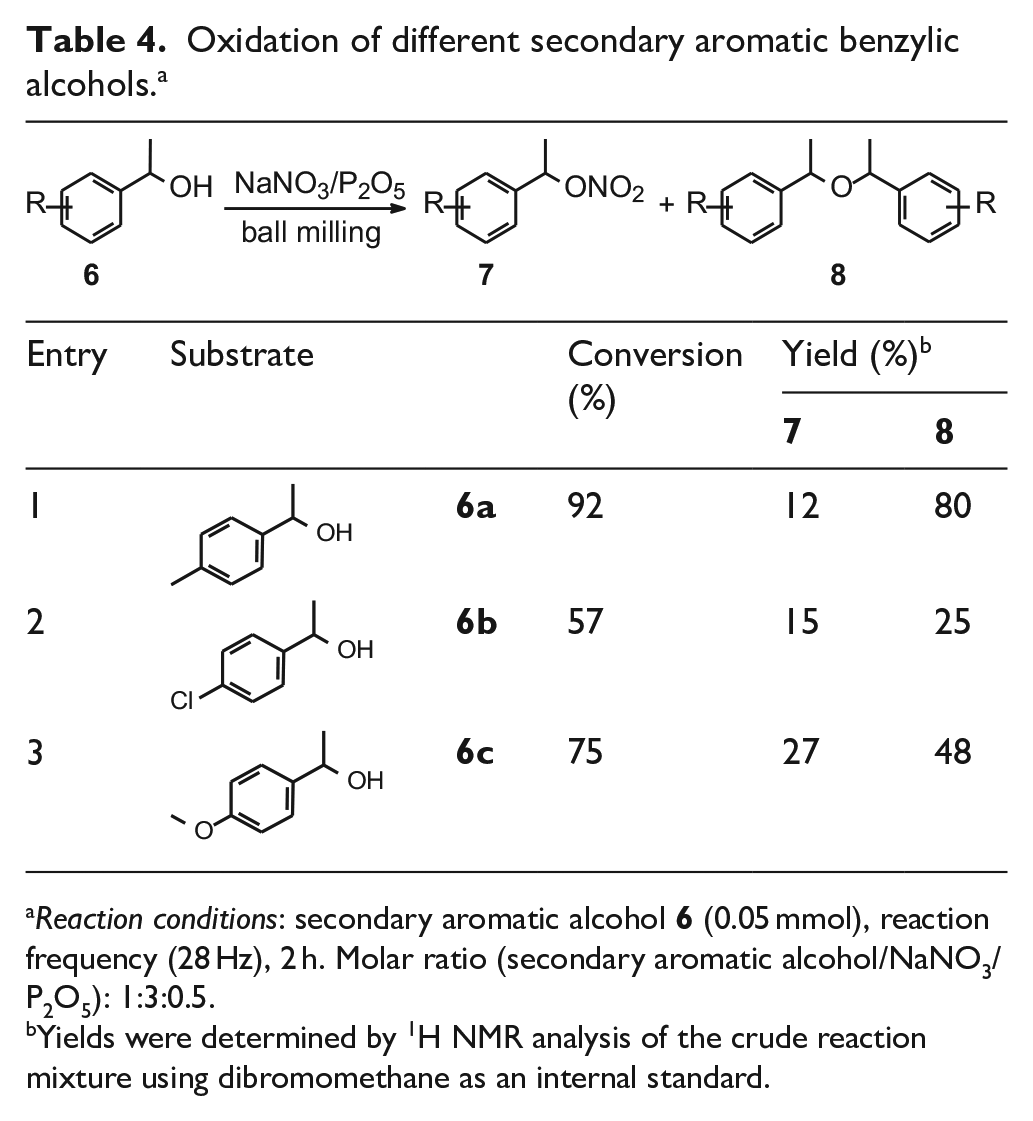
Reaction conditions: secondary aromatic alcohol
Yields were determined by 1H NMR analysis of the crude reaction mixture using dibromomethane as an internal standard.
Mechanistically, a nitrate ester could be a candidate intermediate toward the formation of aromatic aldehydes.33,34 Since small amount of nitrate ester
Considering that NaNO3/P2O5 system works effectively for the oxidation of primary benzylic alcohols, we therefore turned our attention to primary non-benzylic alcohols. Interestingly, while the oxidation of primary benzylic alcohols proceeded well, 2-(4-methylphenyl)ethanol (

Oxidation of 2-(4-methylphenyl) ethanol.
Since excellent oxidation of the benzylic primary hydroxy group was observed, while an aliphatic primary alcohol was inert under this NaNO3/P2O5 system, we decided to evaluate the selectivity of the reagent. The selective oxidation of diols containing both a primary benzylic and an aliphatic hydroxy group was studied as shown in Scheme 2. The diol

Chemoselective oxidation of diol
Currently, we do not understand the precise mechanism of this reaction. Nishiguchi and Strazzolini have presumed a radical mechanism for the oxidation of alcohols.12,15 Thus, we propose a radical pathway for our oxidation of benzylic alcohols since our results are comparable with those of Strazzolini and Nishiguchi (Scheme 3). First, NaNO3 would react with P2O5 to generate nitric acid, which could be decomposed to generate N2O4/NO2 under the mechanical force, as described in our previous work.27,28 Next, the in situ-produced N2O4/NO2 should be responsible for the oxidation, most probably, following the radical-type mechanism reported by Nishiguchi and Strazzolini.12,15

A proposed mechanism for the oxidation of primary benzylic alcohols.
Conclusion
In conclusion, we have reported a methodology for the selective oxidation of primary benzylic alcohols to the corresponding aldehydes using a combination of NaNO3 and P2O5 under HSBM conditions. Chemoselective oxidation of a diol containing both a primary benzylic and a primary aliphatic hydroxy group was also performed. This procedure offers several advantages, including good to excellent yields of aldehydes, compatibility with a broad range of functional groups, the use of simple and inexpensive oxidants, and solvent-free reaction conditions. These features make the present method a useful alternative to current methods for the oxidation of benzylic alcohols.
Experimental
General
All chemicals and reagents were purchased from Meryer (Shanghai, China), Alfa Aesar (Ward Hill, MA, USA), or Energy Chemical (Shanghai, China), and were used as received. The reactions were conducted in a stainless steel milling beaker (2.5 mL) using a stainless steel milling ball in an MM400 mixer ball mill (Retsch GmbH, Germany). Column chromatography was performed using 100–200 mesh silica gel purchased from Qingdao Haiyang Chemical Co. Ltd. (China). Appropriate solvent systems for chromatography were selected according to thin-layer chromatography (TLC) analysis using UV light (254 and 365 nm) to visualize the reaction components. Melting points (m.p.) were obtained on a digital melting point apparatus and are uncorrected. Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AV 400 spectrometer (Billerica, MA, USA) at 400 MHz ( 1 H NMR) and at 101 MHz ( 13 C NMR). The peak patterns are designated as follows: s, singlet; d, doublet; t, triplet; q, quartet; dd, doublet of doublets; dt, doublet of triplets; m, multiplet. Coupling constants, J, are reported in Hz. The product was dissolved in the appropriate deuterium reagent (CDCl3 or dimethyl sulfoxide (DMSO)) and tetramethylsilane (TMS) or the solvent peak was used as a standard. An APEX II (Bruker Inc.) mass spectrometer was used for high-resolution mass spectrometry (HRMS) analysis.
General procedure for the synthesis of compounds 2 and 11
For all the experiments, a mixture of alcohol (
4-Methylbenzaldehyde (2a):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 86% yield (62.01 mg); colorless liquid (lit. 35 ); 1H NMR (400 MHz, CDCl3): δ 9.97 (s, 1H), 7.78 (d, J = 8.0 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 2.44 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 192.1, 145.6, 134.2, 129.9, 129.8, 21.9; HRMS (ESI): m/z [M + H]+ calcd for C8H8O: 121.0648; found: 121.0641.
Benzaldehyde (2b):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 83% yield (52.85 mg); colorless liquid (lit. 36 ); 1H NMR (400 MHz, CDCl3): δ 10.03 (s, 1H), 7.89 (d, J = 7.4 Hz, 2H), 7.64 (t, J = 7.4 Hz, 1H), 7.54 (t, J = 7.6 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 192.5, 136.4, 134.5, 129.8, 129.0; HRMS (ESI): m/z [M + H]+ calcd for C7H6O: 107.0491; found: 107.0495.
4-Biphenylcarboxaldehyde (2c):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 89% yield (97.31 mg); light yellow solid; m.p. 55–57 °C (lit. 37 55 °C); 1H NMR (400 MHz, CDCl3): δ 10.06 (s, 1H), 7.96 (d, J = 8.2 Hz, 2H), 7.76 (d, J = 8.2 Hz, 2H), 7.64 (d, J = 7.3 Hz, 2H), 7.49 (t, J = 7.6 Hz, 2H), 7.42 (t, J = 7.4 Hz, 1H); 13C NMR (101 MHz, CDCl3): δ 192.0, 147.3, 139.8, 135.2, 130.3, 129.1, 128.5, 127.7, 127.4; HRMS (ESI): m/z [M + Na]+ calcd for C13H10O: 205.0624; found: 205.0629.
4-Fluorobenzaldehyde (2d):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 92% yield (68.51 mg); colorless liquid (lit. 38 ); 1H NMR (400 MHz, CDCl3): δ 9.97 (s, 1H), 7.92 (dd, J = 8.7, 5.5 Hz, 2H), 7.22 (t, J = 8.5 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 190.6 (s), 166.6 (d, J = 256.8 Hz), 133.0 (d, J = 2.7 Hz), 132.3 (d, J = 9.7 Hz), 116.4 (d, J = 22.3 Hz); 19F NMR (376 MHz, CDCl3): δ −102.34; HRMS (ESI): m/z [M + H]+ calcd for C7H5FO: 125.0397; found: 125.0394.
4-Chlorobenzaldehyde (2e):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 89% yield (75.06 mg); light yellow solid; m.p. 44–45 °C (lit. 39 44–46 °C); 1H NMR (400 MHz, CDCl3): δ 9.99 (s, 1H), 7.83 (d, J = 8.4 Hz, 2H), 7.52 (d, J = 8.4 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 190.9, 141.0, 134.8, 131.6, 131.0, 129.5, 129.0; HRMS (ESI): m/z [M + Na]+ calcd for C7H5ClO: 162.9921; found: 162.9924.
4-Bromobenzaldehyde (2f):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 98% yield (108.79 mg); light yellow solid; m.p. 56–58 °C (lit. 39 57–59 °C); 1H NMR (400 MHz, CDCl3): δ 9.98 (s, 1H), 7.72 (dd, J = 25.5, 8.5 Hz, 4H); 13C NMR (101 MHz, CDCl3): δ 191.1, 135.1, 132.5, 131.0, 129.8; HRMS (ESI): m/z [M + Na]+ calcd for C7H5BrO: 206.9416; found: 206.9419.
2,4-Dibromobenzaldehyde (2g):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 100% yield (158.35 mg); white crystalline solid; m.p. 81–84 °C (lit. 40 84–85 °C); 1H NMR (400 MHz, CDCl3): δ 10.30 (s, 1H), 7.85 (s, 1H), 7.78 (d, J = 8.3 Hz, 1H), 7.59 (d, J = 8.3 Hz, 1H); 13C NMR (101 MHz, CDCl3): δ 190.8, 136.4, 132.4, 131.5, 130.8, 129.9, 127.5; HRMS (ESI): m/z [M + Na]+ calcd for C7H4Br2O: 284.8521; found: 284.8526.
4-Formylbenzoic acid (2h):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 10:1) in 78% yield (70.26 mg); yellow crystalline solid; m.p. 246–247 °C (lit. 41 245–246 °C); 1H NMR (400 MHz, DMSO): δ 13.41 (s, 1H), 10.10 (s, 1H), 8.12 (d, J = 7.6 Hz, 2H), 8.01 (d, J = 7.7 Hz, 2H); 13C NMR (101 MHz, DMSO-d6): δ 192.8, 166.4, 138.7, 135.4, 129.7, 129.3; HRMS (ESI): m/z [M − H]− calcd for C8H6O3: 149.0244; found: 149.0249.
1,4-Benzenedicarboxaldehyde (2i):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 10:1) in 100% yield (80.48 mg); white solid; m.p. 114–115 °C (lit. 42 114–115 °C); 1H NMR (400 MHz, CDCl3): δ 10.14 (s, 2H), 8.05 (s, 4H); 13C NMR (101 MHz, CDCl3): δ 191.5, 140.1, 130.2; HRMS (ESI): m/z [M + Na]+ calcd for C8H6O2: 157.0260; found: 157.0265.
2-Nitrobenzaldehyde (2j):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 20:1) in 98% yield (88.86 mg); yellow crystalline solid; m.p. 42–43 °C (lit. 43 43 °C); 1H NMR (400 MHz, CDCl3): δ 10.44 (s, 1H), 8.13 (d, J = 7.9 Hz, 1H), 7.97 (d, J = 7.6 Hz, 1H), 7.75-7.82 (m, 2H); 13C NMR (101 MHz, CDCl3): δ 188.1, 149.6, 134.1, 133.7, 131.4, 129.7, 124.5; HRMS (ESI): m/z [M + H]+ calcd for C7H5NO3: 152.0342; found: 152.0347.
4-Nitrobenzaldehyde (2k):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 20:1) in 87% yield (78.88 mg); yellow crystalline solid; m.p. 105–106 °C (lit. 43 105 °C); 1H NMR (400 MHz, CDCl3): δ 10.16 (s, 1H), 8.40 (d, J = 8.6 Hz, 2H), 8.08 (d, J = 8.7 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 190.3, 151.2, 140.1, 130.5, 124.4; HRMS (ESI): m/z [M + Na]+ calcd for C7H5NO3: 174.0162; found: 174.0168.
4-Methoxybenzaldehyde (2l):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 25:1) in 74% yield (60.45 mg); colorless liquid (lit. 35 ); 1H NMR (400 MHz, CDCl3): δ 9.89 (s, 1H), 7.84 (d, J = 8.7 Hz, 2H), 7.01 (d, J = 8.6 Hz, 2H), 3.89 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 190.9, 164.6, 132.0, 130.0, 114.4, 55.6; HRMS (ESI): m/z [M + H]+ calcd for C8H8O2: 137.0597; found: 137.0602.
3-Methoxybenzaldehyde (2m):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 25:1) in 63% yield (51.46 mg); colorless liquid (lit. 44 ); 1H NMR (400 MHz, CDCl3): δ 9.98 (s, 1H), 7.48–7.44 (m, 2H), 7.40 (d, J = 2.0 Hz, 1H), 7.19 (dt, J = 6.8, 2.5 Hz, 1H), 3.87 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 192.2, 160.2, 137.8, 130.1, 123.6, 121.6, 112.0, 55.5; HRMS (ESI): m/z [M + Na]+ calcd for C8H8O2: 159.0417; found: 159.0421.
4-Acetoxybenzaldehyde (2n):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 70% yield (68.95 mg); colorless liquid (lit. 45 ); 1H NMR (400 MHz, CDCl3): δ 9.99 (s, 1H), 7.92 (d, J = 8.6 Hz, 2H), 7.28 (d, J = 8.5 Hz, 2H), 2.34 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 191.0, 168.8, 155.4, 134.0, 131.3, 122.4, 21.2; HRMS (ESI): m/z [M + Na]+ calcd for C9H8O3: 187.0366; found: 187.0369.
4-Acetamidobenzaldehyde (2o):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 30:1) in 55% yield (53.85 mg); yellow crystalline solid; m.p. 151–153 °C (lit. 46 149–151 °C); 1H NMR (400 MHz, CDCl3): δ 9.93 (s, 1H), 7.86 (d, J = 8.6 Hz, 2H), 7.69 (d, J = 8.3 Hz, 1H), 7.37 (s, 1H), 2.24 (s, 3H); 13C NMR (101 MHz, CDCl3): δ 191.0, 168.6, 143.5, 132.4, 131.2, 119.3, 24.8; HRMS (ESI): m/z [M + Na]+ calcd for C9H9NO2: 186.0525; found: 186.0518.
2-Naphthalenecarboxaldehyde (2p):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 25:1) in 74% yield (69.34 mg); yellow solid; m.p. 54–55 °C (lit. 47 56–59 °C); 1H NMR (400 MHz, CDCl3): δ 10.17 (s, 1H), 8.36 (s, 1H), 8.03–7.91 (m, 4H), 7.67–7.59 (m, 2H); 13C NMR (101 MHz, CDCl3): δ 192.4, 138.1, 138.0, 136.5, 134.6, 132.7, 129.6, 129.2, 129.2, 128.1, 127.1, 122.8; HRMS (ESI): m/z [M + Na]+ calcd for C11H8O: 179.0467; found: 179.0462.
4-(2-Hydroxyethyl)benzaldehyde (11):
Isolated by silica gel column chromatography (petroleum ether/ethyl acetate = 10:1) in 55% yield (49.56 mg); colorless oil (lit. 48 ); 1H NMR (400 MHz, CDCl3): δ 9.99 (s, 1H), 7.84 (d, J = 8.0 Hz, 2H), 7.41 (d, J = 7.9 Hz, 2H), 3.93 (t, J = 6.5 Hz, 2H), 2.96 (t, J = 6.4 Hz, 2H); 13C NMR (101 MHz, CDCl3): δ 192.0, 146.2, 135.0, 130.1, 129.7, 63.1, 39.3; HRMS (ESI): m/z [M + Na]+ calcd for C9H10O2: 173.0573; found: 173.0578.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221101983 – Supplemental material for Selective oxidation of primary benzylic alcohols to aldehydes using NaNO3 under ball milling
Supplemental material, sj-docx-1-chl-10.1177_17475198221101983 for Selective oxidation of primary benzylic alcohols to aldehydes using NaNO3 under ball milling by Jing-Fei Wang, Li-Yu Lu, Pu Zhang, Yu-Jun Qin and Zhi-Xin Guo in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are thankful to the Renmin University of China for providing necessary facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.