Abstract
Verbascoside is isolated from the whole plants of the Verbenaceae family and demonstrates significant levels of bioactivity. In this work, we report on the isolation of verbascoside, a phenolic glycoside, from the stems of Clerodendrum inerme collected in Vietnam and verify its structure by comparing its spectroscopic data with those reported in the literature. Verbascoside is then applied as a monomer for the electrochemical deposition of a layer of polyverbascoside onto glassy carbon electrodes toward Cu2+ sensing. Electrochemical results demonstrate that the polyverbascoside-modified glassy carbon electrode is highly selective for Cu2+ ions compared to other evaluated ions including Ni2+ and Co2+. The developed sensor can detect Cu2+ in a concentration range from 0 to 175 μM with a sensitivity of 0.372 µA µM−1 and a limit of detection of 50 nM Cu2+ ions.
Keywords
Introduction
Verbascoside (VBC) is considered as a triglycoside of luteolin. As a crucial member of the flavonoids, there are many reports in the literature on the biological and physiological activities of VBC, with examples including vasodilation, antioxidant, inhibition of lipid peroxidations and DNA oxidative damage, and anticarcinogen and antimutagenic effects. In general, VBC is isolated from the whole plant of Verbascum thapsus, mainly Clerodendrum inerme Gaertn. (Figure 1).1 –3 In many reports, V. thapsus is used in the indigenous system of Indian medicine for the treatment of inflammatory disease, asthma, spasmodic coughs, and migraine and has also been reported to possess antiviral activity against influenza in chicken embryos.1 –4 C. inerme Gaertn. grows near the seashore in tropical Asia and the Pacific. 3 In traditional medicine, this plant is useful for the treatment of skin diseases, topical burns, and rheumatism. 5 The constituents of this plant have been investigated by Vendantham et al. 6 (flavonoids), Achari et al. 1 (clerodane diterpenes), and Kanchanapoom et al. 7 (phenylethanoid, megastigmane, and iridoid glycosides). There are many reports in the literature on the additional bioactivities of these constituents, such as antioxidant, anti-inflammatory and photo-protective activity,8,9 and cancer inhibitions. 10 However, there have been no reports on the redox process and electrocatalytic ability of VBC.

Structure of verbascoside and the proposed interaction of verbascoside with copper(II) ions. Insert: a photo of Clerodendrum inerme Gaertn. (Verbenaceae).
Copper(II) (Cu2+) is a transition that is metal essential for life; however, at elevated concentrations, it is toxic to organisms such as algae, fungi, and many bacteria, and in humans may adversely affect the gastrointestinal, hepatic, and renal systems. It should be stressed that levels of Cu2+ concentrations in drinking water below 2 mg L−1 correspond to the World Health Organization (WHO) guidelines.11 –15 In fact, Cu2+ ions can be detected and quantified by routine methods with the most common being flame atomic absorption spectrometry with a limit of detection (LoD) at level less than 10 nM.16 –18 Other methods include the most sensitive, that is, mass spectrometry (coupled to inductively coupled plasma) with an LoD of 5 ng L−1,19,20 as well as optical21 –24 and electrochemical14,15,25 techniques. However, the above techniques are laborious and require complicated equipment. Therefore, it is important to develop a simple, sensitive, and adaptive method for the determination of Cu2+ ions in aqueous media.
In this paper, we describe the isolation of a phenolic glycoside, that is, VBC, from the stems of C. inerme collected in Vietnam. The structure of this compound was identified by comparison with reported data. For the development of an application, VBC was subjected to electrochemical oxidative deposition to form a polymer (polyverbascoside (polyVBC)) on a glassy carbon electrode (GCE) surface using cyclic voltammetry (CV). The obtained polyVBC/GCE has been used as an electrochemical sensor for sensing heavy metal ions, during which Cu2+, Co2+, and Ni2+ ions have also been investigated. The obtained results suggest that polyVBC/GCEs can be used for highly selective and sensitive Cu2+ ion sensing in a linear range from 0 to 175 μM, with a sensitivity of 0.372 µA µM–1 and an LoD of 50 nM Cu2+ ions.
Experimental
Materials and reagents
NMR spectra were recorded on a Bruker Avance 500 MHz spectrometer with tetramethylsilane (TMS) as an internal standard. MS was performed on an ESI-LC/MS/MS-Xevo TQMS instrument (Agilent Technologies, Waldbronn, Germany). For column chromatography, silica gel 60 (0.04–0.063 mm, Merck, Kenilworth, NJ, USA), YMC GEL (ODS-A, 12 nm S–150 µm, YMC Co., Ltd., Kyoto, Japan), and Sephadex LH-20 (25–100 µm, Sigma-Aldrich, St. Louis, MO, USA) were used. TLC was performed on Merck pre-coated TLC DC-Alufolien silica gel 60F254 and RP-18F254S, and compounds were detected by UV fluorescence or by spraying with 1% vanillin-H2SO4, followed by heating at 100 °C for 1–2 min.
Plant materials
The stems of C. inerme (Verbenaceae) were collected from the semi-mangrove areas of Thai Binh Province in May 2018. The species was identified by Professor Dr Tran Huy Thai, Institute of Ecology and Biological Resources, Vietnam Academy of Science and Technology (VAST). A voucher specimen (no. BK-N02) has been deposited at the Hanoi University of Science and Technology, Vietnam.
Extraction and isolation
The dry powdered stems (5.0 kg) of C. inerme were extracted with 80% aqueous methanol at 50 °C (each 3 × 7 L) by sonication for 60 min. After evaporation of the solvent under reduced pressure, the residue (185.0 g) was suspended in water (3 L) and defatted with n-hexane. The aqueous layer was further extracted with ethyl acetate to give ethyl acetate (17.0 g) and water (95.5 g) residues, after removal of the solvents. The ethyl acetate extract (17.0 g) was subjected to silica gel column chromatography with solvents of increasing polarity (ranging from 0% to 100% methanol in dichloromethane) to give 10 fractions (F1–F10). Fraction F9 (2.9 g), eluted with dichloromethane–methanol (7:3, v/v), was re-chromatographed using Sephadex LH-20 column with methanol to give four sub-fractions F9.1–F9.4. Purification of sub-fraction F9.4 (0.84 g) over YMC GEL using water–methanol (3:2, v/v) afforded compound
VBC (
HR-ESI-MS: m/z [M + Na]+ calcd for C29H36O15Na: 647.1946; found: 647.1928, ESI-MS: m/z = 647 [M + Na]+.
1 H-NMR (500 MHz, CD3OD): δ 7.63 (1H, d, J = 16.0 Hz, H-γ’), 7.07 (1H, d, J = 2.0 Hz, H-2’’’), 6.98 (1H, dd, J = 8.0, 2.0 Hz, H-6’’’), 6.80 (1H, d, J = 8.0 Hz, H-5’’’), 6.72 (1H, d, J = 2.0 Hz, H-2), 6.70 (1H, d, J = 8.0 Hz, H-5), 6.59 (1H, dd, J = 8.0, 2.0 Hz, H-6), 6.30 (1H, d, J = 16.0 Hz, H-β’), 5.21 (1H, d, J = 2.0 Hz, H-1’’), 4.94 (1H, t, J = 9.5 Hz, H-4’), 4.40 (1H, d, J = 8.0 Hz, H-1’), 4.09-4.04 (1H, m, H-α), 3.93 (1H, m, H-2’’), 3.83 (1H, t, J = 9.0 Hz, H-3’), 3.77-3.72 (1H, m, H-α), 3.65-3.41 (7H, m, protons of glucose and rhamnose), 2.82 (2H, t, J = 7.0 Hz, H-β), 1.12 (3H, d, J = 6.0 Hz, H-6’’).
13 C NMR (125 MHz, CD3OD): δ 168.3 (C-α’), 149.7 (C-4’’’), 148.0 (C-γ’), 146.8 (C-4), 146.1 (C-3’’’), 144.7 (C-3), 131.5 (C-1), 127.7 (C-1’’’), 123.2 (C-6’’’), 121.2 (C-6), 117.1 (C-2), 116.5 (C-5’’’), 116.3 (C-5), 115.2 (C-2’’’), 114.7 (C-β’), 104.2 (C-1’), 103.0 (C-1’’), 81.6 (C-3’), 76.2 (C-2’), 76.0 (C-5’), 73.8 (C-4’’), 72.3 (C-2’’), 72.2 (C-α), 72.0 (C-3’’), 70.6 (C-4’), 70.4 (C-5’’), 62.4 (C-6’), 36.5 (C-β), 18.4 (C-6’’).
Electro-polymerization to prepare polyVBC/GCE
VBC solution (1 mM) was prepared by dissolving VBC powder in a mixture of EtOH/PBS (volume ratio, 1:14) and was subjected to ultrasonication to give a homogeneous solution. VBC was deposited onto the GCE surface by electrochemical-polymerization using a CV technique with an applied potential from 0 to 1 V (vs SCE) at a scanning rate of 0.05 V s−1 for 10 scans. The obtained film of polyVBC onto GCE surface is denoted as polyVBC/GCE.
Electroactivity of polyVBC/GCE
The electroactivity of polyVBC/GCE was tested in sodium acetate/acetic acid buffer at pH 6 (NaOAc/AcOH) solution using CV with an applied potential from 1.0 to −1.0 V (vs SCE) and a scanning rate of 0.05 V s−1 for 30 cycles to evaluate the electrochemical stability of the polyVBC film. The electroactivity of VBC in NaOAc/AcOH solution on neat GCE and polyVBC/GCE film were also investigated in the presence and absence of Cu2+ ions.
Electrochemical detection of Cu(II)
A solution containing Cu2+ ions was prepared by dissolving CuSO4 or CuCl2 in acidified distilled water (pH 6) (to prevent any hydrolysis reactions). Using polyVBC/GCE, Cu2+ ions can be recognized by electrochemical methods including CV or by square wave voltammetry (SWV). Thus, to an electrochemical cell containing 5 mL of NaOAc/AcOH (pH 6), 100 µL of the Cu2+ solution was added. The CV and SWV of the polyVBC/GCE before and after addition of Cu2+ were recorded. A change in the response of the electrochemical signals was used to monitor the Cu2+ levels in solution. For selectivity testing, Co2+ and Ni2+ ions were used instead of Cu2+.
Results and discussion
VBC identification
Compound
Electro-polymerization of VBC on GCE
Figure 2(a) shows the cyclic voltammograms (the first and fifteenth cycles of 15 cycles, v = 50 mV s−1, between 0 and 1.0 V vs SCE) corresponding to electro-oxidation of VBC (1 mM) on a GCE in EtOH/PBS (1:14) solution. The first cycle shows an oxidation wave corresponding to oxidation of the hydroxy group of the first VBC residue, while the fifteenth cycle (Figure 2(a)) shows partial passivation, which indicates that VBC has been electrodeposited onto the GCE. As can be seen in Figure 2(b), there is no obvious electrochemical response at the bare GC electrode in NaOAc/AcOH buffer (curve i), while showed there is a redox couple (Eox/Ered) at +0.28/−0.04 V (vs SCE) in the CV of GCE recorded in NaOAc/AcOH buffer containing of 1 mM of free VBC (curve ii). However, this redox couple shifts to −0.10/−0.16 V in the case of polyVBC/GCE (curve iii). The lower redox potential and smaller difference between the anodic peak potential and the cathodic peak potential of polyVBC/GCE (ΔE = 60 mV) than that of free VBC on GCE (ΔE = 240 mV) clearly indicates a significantly improved electron-transfer process for the polyVBC-modified GC electrodes. Based on the obtained electrochemical results, a reaction mechanism is proposed in Figure 2(c).

(a) The first and fifteenth cycles of the CVs recorded in PBS containing 1 mM of verbascoside at a scan rate of 50 mV s−1 of GCE; (b) CVs recorded at a scan rate of 100 mV s−1 of GCE recorded in acetate buffer (pH 6) in (i) the absence and (ii) the presence of 0.2 mM of verbascoside (VBC), and (iii) CV of polyVBC/GCE in acetate buffer (pH 6); (c) a scheme of proposed mechanism for the electrochemical redox reaction of verbascoside.
Figure 3(a) shows the CVs of the GC electrode in NaOAc/AcOH solution containing VBC (0.2 mM) in the absence (curve i) and in the presence of 0.160 mM Cu2+ (curve ii), which show slight differences in the current of the oxidation peak at +0.2 V (vs SCE) and the reduction peak at −0.05 V (vs SCE). These results imply that the GC electrode can detect Cu2+ ions using free VBC in NaOAc/AcOH buffer; however, the sensitivity is poor. In contrast, the CVs of polyVBC/GCE recorded in the NaOAc/AcOH buffer (Figure 3(b)) in the absence (curve i) and the presence of 0.160 mM Cu2+ (curve ii) are completely different. The oxidation and reduction peak positions in curve ii did not change compared to those in the absence of Cu2+ ions (curve i), but the current intensities strongly increased from 0.94 to 9.80 µA (for the oxidation peak), and from 1.32 to 10.44 µA (for the reduction peak), respectively, when only a 0.160 mM concentration of Cu2+ ion was used. These results indicate that polyVBC/GCE is very sensitive to Cu2+ ions. For comparison, polyVBC/GCE has been used to recognize the presence of Co2+ and Ni2+ ions in NaOAc/AcOH using CV (Figure 3(c)). The results show that the position of the redox peak did not change when adding 0.4 mM Ni2+ (curve ii) or 0.4 mM Co2+ (curve iii) in comparison with the absence of any heavy metal ion (curve i). Meanwhile, a strong change was observed in the presence of only 0.02 mM Cu2+ (curve iv). These results highlight the high Cu2+ ions selectivity of the polyVBC/GCE sensor.26
–28 In fact, VBC can “capture” Cu2+ ions by creating bonds between unshared valence electrons on the oxygen atoms of hydroxy groups on VBC with the empty-valence atomic orbitals (AOs) of Cu2+ (see Figure 1, structure
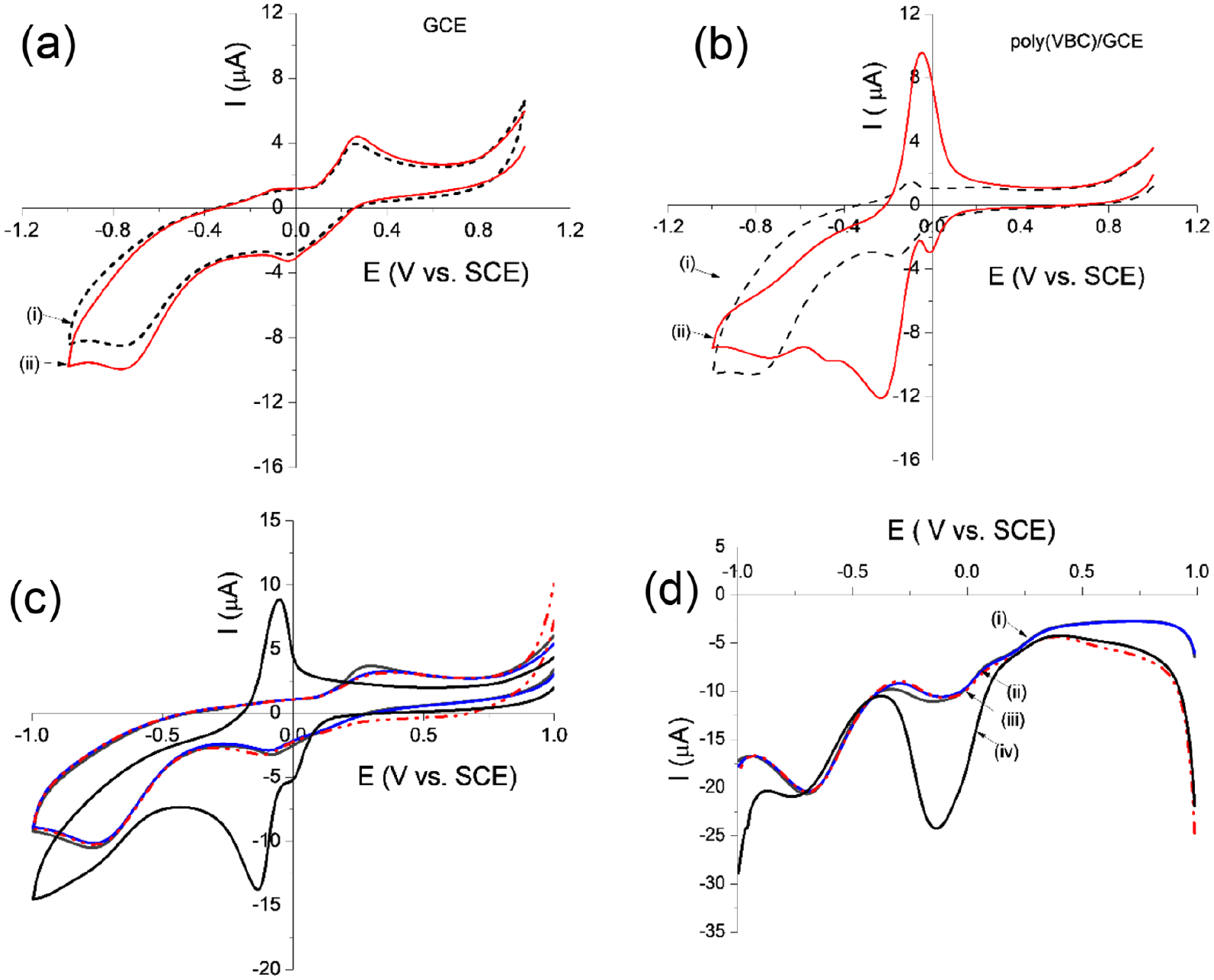
(a) CVs of GCE recorded in acetate buffer (pH 6) containing 0.2 mM verbascoside in (i) the absence and (ii) in the presence of 0.160 mM Cu2+; (b) CVs of polyVBC/GCE recorded in acetate buffer (pH 6) in (i) the absence and (ii) in the presence of 0.160 mM Cu2+; (c) CVs of polyVBC/GCE recorded in acetate buffer (pH 6) in (i) the absence and (ii–iv) in the presence of 0.4 mM Co2+ (curve ii), (iii) 0.4 mM Ni2+ (curve iii), and (iv) 0.02 mM Cu2+ (curve iv); (d) square wave voltammetry (SWV) of corresponding samples from (c). All CVs were recorded at a scan rate of 100 mV s−1, and detailed experimental conditions are described in the text.
Figure 4(a) shows the SWV response results of polyVBC/GCE with different concentrations of Cu2+ ions. It can be seen that an increase in the concentration of Cu2+ led to an increase in the peak current (Ipeak) of the response. A change in the electrochemical response was observed even at a low concentration of Cu2+ ions (100 nM). This phenomenon becomes more obvious when the target concentration continues to rise (up to 250 µM). These results can be explained by the fact that when the amount of Cu2+ ions in the aqueous solution increases, the amount of Cu2+ ions accumulating on the surface of the polyVBC layer increases, leading to an increase in the response current in the SWV. The sensitivity of the electrochemical sensors was investigated by plotting Ipeak versus different Cu2+ concentrations (Figure 4(b)). The plot shows a linear relationship between Ipeak versus [Cu2+] in the range from 100 nM to 150 µM: Ipeak (μA) = (5.955 ± 0.926) + (0.372 ± 0.011) × [Cu2+] (µM) with a correlation coefficient of R2 = 0.992. Based on a signal-to-noise ratio characteristic of 3 (S/N = 3), 15 the LoD of Cu2+ ions on the polyVBC/GC electrode was estimated to be 50 nM. The fabricated electrochemical sensor was compared with other examples that have been reported in previous studies (Table 1). These comparisons show that the polyVBC/GCE has significant potential for the determination of Cu2+ ions in terms of simple fabrication, direct detection, rapid analysis, and high sensitivity.

(a) SWVs of polyVBC/GCE recorded in acetate buffer (pH 6) in the presence of different concentrations of Cu2+, and (b) a calibration curve for Cu2+ detection.
Comparison of the fabricated electrochemical sensor in this work with examples from prior literature.

N/A: not available.
GE: gold electrode; GEC: glassy carbon electrode; GME: gold microelectrode; CPE: carbon paste electrode.
CV: cyclic voltammetry; SWV: square wave voltammetry; DPV: differential pulse voltammetry; FET: field effect transistor; LSV: linear sweep voltammetry; SCV: stripping cyclic voltammetry; DPASV: differential pulse anodic stripping voltammetry.
Conclusion
We have reported the extraction and isolation of VBC from the stems of Clerodendrum, which can be used as a natural monomer for the electrochemical modification of a GCE for the electrochemical detection of Cu2+ ions. The obtained results indicate that the generated polyVBC/GCE is highly selective and sensitive for Cu2+ ion detection in a concentration range from 0 to 175 μM using the SWV technique, with a sensitivity of 0.372 µA µM−1 and an LoD of 50 nM (Cu2+ ions). The results demonstrate that the polyVBC/GCE is a promising material for Cu2+ ion sensing in terms of its simple preparation, rapid analysis time, and high selectivity and sensitivity for the direct detection of Cu2+ ions in aqueous samples.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.T.T. thanks the Vietnam National Foundation for Science and Technology Development (NAFOSTED) for funding (grant no. 104.01-2018.36) and H.V.T. thanks the Vietnam Ministry of Education and Training for funding (project no. CT2022.04.BKA.03).
Data availability statement
The data used to support the findings of this study are included within the article.
