Abstract
The first palladium-catalyzed cross-coupling of various substituted benzyltitaniums with aryl triflates is presented for the synthesis of diarylmethanes in yields of up to 94% through highly selective C–O bond functionalization. The benzyltitaniums act as nucleophiles to realize the C(sp2)–C(sp3) cross-coupling with high efficiency in short reaction times. The reactions proceed at 60°C and show excellent functional groups tolerance.

Introduce
Palladium-catalyzed cross-coupling plays an important role in the industrial manufacture of pharmaceuticals, fine chemicals, and materials.1–3 Researchers have found that a significant number of bioactive molecules contain a methylene functional group, which makes research on diarylmethane-containing compounds important (Figure 1).4,5 The preparation of diarylmethanes is usually accomplished by four common methods: (a) electrophilic substitution of aromatics, which is often referred to as Friedel–Crafts benzylation6,7 but suffers from poor regioselectivity; (b) nucleophilic aromatic substitution reactions with benzyl derivatives as nucleophiles; 8 (c) C–H activation of aromatic hydrocarbons;9–11 and (d) the most commonly used strategy to prepare diarylmethanes through transition metal-catalyzed coupling by employing either electrophilic benzyl derivatives12,13 or nucleophilic benzyl reagents,14–16 such as reactions using organoboron derivatives, 17 organozinc, 18 or Grignard reagents. 19 More recently, organotitanium reagents have been used as organometallic partners in C(sp2)–C(sp2) and C(sp2)–C(sp3) cross-coupling reactions.20–28 Organotitanium reagents have lower nucleophilicity, basicity, and better functional group tolerance compared with Grignard reagents. Moreover, compared with organozinc reagents, organotitanium reagents have higher metal transfer rates. Studies by Dastbaravardeh and Knochel 21 have shown that the reactivity of organotitanium reagents as nucleophiles is much higher than that of the corresponding organozinc counterparts.
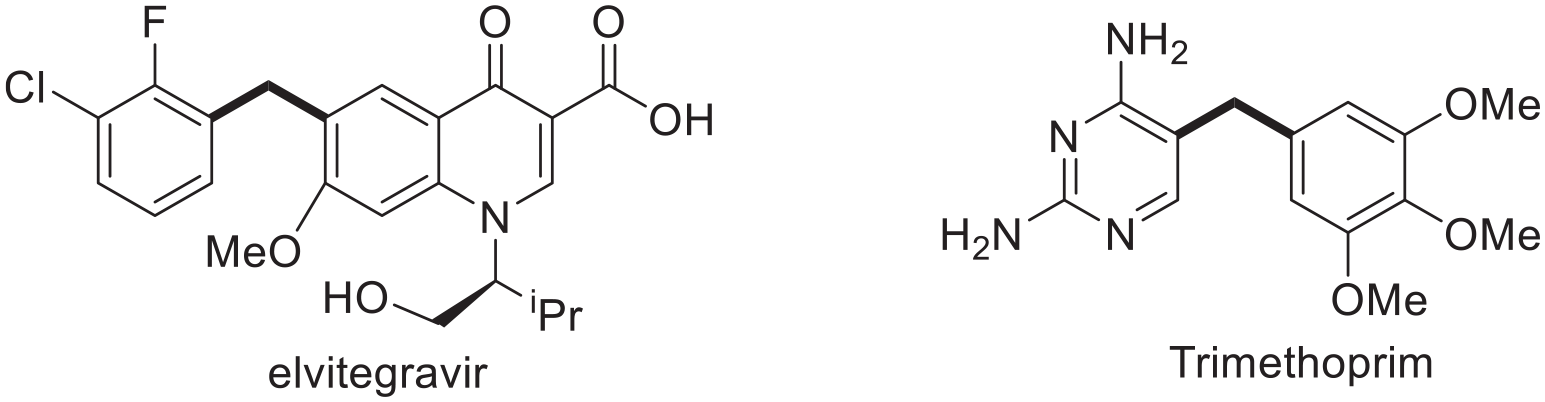
Examples of the diarylmethane motif in pharmaceuticals.
Interestingly, although many exciting achievements have been made in nucleophilic addition of organotitanium reagents to carbonyl compounds (Scheme 1a),29,30 by comparison, much less attention has been devoted to the employment of such reagents in C(sp2)–C(sp2) cross-coupling transformations (Scheme 1b).20–28 Furthermore, there is only one successful example of the employment of such reagents in C(sp2)–C(sp3) cross-coupling transformations (Scheme 1c). 21 To the best of our knowledge, this is the first example using organotitanium nucleophiles to generate diarylmethane units by C(sp2)–C(sp3) cross-coupling reactions (Scheme 1d).

Comparison of reported methods and our design.
Organic halides are commonly the electrophiles,31–36 which generate halogenated waste after reactions. Also, halides are sometimes limited by reaction conditions, making it difficult to use them in some advanced synthetic intermediates; these shortcomings can be solved using alternative partners, such as C–O electrophiles.37,38 Aryl triflates are popular as electrophiles in transition metal-catalyzed cross-coupling reactions because they are derived from readily available phenols and carbonyl enolates. 39 However, phenolic hydroxy and carbonyl moieties are more common organic functional groups than non-commercially available aryl halides. 40 Herein, we present the first palladium-mediated cross-coupling of benzyltitanium reagents with aryl triflates to give the corresponding diarylmethanes in yields of up to 94%.
Results and discussion
To develop a new C(sp2)–C(sp3) cross-coupling catalyzed protocol, we initially used phenyl triflate (
Optimization of the reaction conditions a .

Reaction conditions:
GC yield calibrated against tridecane as an internal standard, n.d = not detected.
The additive (1.2 equiv.) was added and the reaction was run at 60°C.
Isolated yield is given in parentheses.
The use of various phosphine ligands subsequently tested at a temperature of −10°C using 3 mol% of Pd(OAc)2 as the catalyst. The results showed that the highest yield of product
On the basis of the optimized reaction conditions, we next investigated the scope and generality of the cross-coupling reactions of a range of aryl triflates with different benzyltitanium lithium chloride species, and the results are given in Table 2. First, we studied the influence of the functional group on the aryl triflate. Reactions with non-activated aryl triflates proceeded satisfactorily. Phenyl triflate (
Reaction conditions:
Isolated yields.
Palladium-catalyzed reactions of mixtures of phenyl triflate (

Chemoselective cross-coupling.
We propose the possible reaction mechanism shown in Scheme 3. First, oxidative addition of Pd(0) to the aryl triflate

Possible mechanism for the cross-coupling reactions.
Conclusion
In conclusion, we have developed the first examples of the palladium-catalyzed reactions of benzyltitanium reagents with aryl triflates to give diarylmethane derivatives. The procedure demonstrates broad suitability, being useful for the coupling of both electron-deficient and electron-rich aryl triflates. A large variety of functionalized diarylmethanes have been efficiently prepared using this methodology, which is based on the use of the commercially available catalytic system Pd(OAc)2/
Experimental
General
Anhydrous lithium chloride, Pd(OAc)2, Pd(PPh3)4, and Pd2(dba)3 were purchased from Sigma-Aldrich. THF, 1,4-dioxane, and 1,2-dimethoxylethane were purchased from Alfa Aesar. Other reagents are available commercially and were used without further purification, unless otherwise indicated. All reactions were carried out under an argon atmosphere with dry solvents under anhydrous conditions, unless otherwise noted. THF was dried over alumina under N2 using a Grubbs-type solvent purification system. All benzyltitanium reagents were prepared from the corresponding benzylmagnesium halides. 4-Hydroxy-N,N-dimethylbenzamide was purchased from Alfa Aesar, 4-(dimethylcarbamoyl)phenyltriflate (
Experimental section
General procedure A: benzyltitanium reagents
The glassware was oven-dried (100°C) and cooled under a stream of argon gas. Aryl Grignard reagents, such as benzylmagnesium chloride and 4-methoxybenzylmagnesium chloride, were prepared according to standard procedures. Functionalized aryl Grignard reagents, including 4-cyanobenzylmagnesium chloride, were prepared by iodine–magnesium exchange using i-PrMgCl•LiCl according to Knochel method. 50 All of the Grignard reagents were titrated before use. 51
The Grignard reagent (20 mmol) was cooled to 0°C (functionalized Grignard reagents were cooled to −20°C). A solution of Ti(OiPr)4 (4.08 mL, 15.0 mmol in 50 mL THF) and TiCl4 (0.56 mL, 5.0 mmol) were added at 0°C to a separate two-necked 500 mL round-bottomed flask under a nitrogen atmosphere. The resulting solution was warmed to room temperature and stirred for 30 min to give a solution of ClTi(OiPr)3 (100 mmol). The ClTi(OiPr)3 solution was cooled to 0°C or −20°C and the cold (0°C or −20°C) Grignard solution was added through a cannula. The reaction mixture was warmed to room temperature and stirred for 2 h (functionalized titanium reagents were warmed to −10°C and allowed to react for 4 h). The volatile material was removed completely under reduced pressure, and under a nitrogen atmosphere, the residue was extracted with n-hexane (3 × 40 mL). The combined hexane solution was concentrated and was cooled to −20°C, furnishing the aryltris(2-propoxo)titanium species as a solid product.
General procedure B: palladium-catalyzed cross-coupling of aryl triflates with benzyltitanium reagents
In a glovebox, the aryl triflate
Ethyl 4-[(4-(Dimethylamino)benzyl)]benzoate (
Ethyl 4-[(4-(Dimethylcarbamoyl)benzyl)]benzoate (
Supplemental Material
sj-doc-1-chl-10.1177_17475198221091941 – Supplemental material for A new protocol for synthesizing diarylmethanes using a benzyltitanium reagent as a nucleophile
Supplemental material, sj-doc-1-chl-10.1177_17475198221091941 for A new protocol for synthesizing diarylmethanes using a benzyltitanium reagent as a nucleophile by He Zhang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by Hebei Chemical and Pharmaceutical College.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

