Abstract
Visible-light-excited fluorescent probes with excellent water solubility are of significant research importance in the biomedical field. In this work, a series of visible-light-responsive carbocyanine fluorescent compounds [DiSCn(3)] are synthesized from 2-methylbenzothiazole and are employed as fluorescent probes for detecting tin(II) ions. Upon addition of tin(II) ions to an aqueous solution of compounds DiSCn(3)-3 and 4, both exhibit considerable changes in their absorption and fluorescent intensity, respectively. These spectral changes may correspond to the binding between tin(II) ions and the sulfur atom in the benzothiazole unit to form a metal complex. This indicates that DiSCn(3)-3 and 4 can be used as promising fluorescent chemical sensors for detecting tin(II) ions in aqueous solution via visible-light excitation.

Introduction
Nowadays, metal ions have a significant impact in terms of environmental pollution and human health, respectively. Hence, effective detection methods for metal ions are necessary. With the development of coordination chemistry and probe technique, different instrumental methods, functional nucleic acids (FNAs)-based sensors,1,2 electrochemical biosensors, 3 aggregation-induced emission (AIE)-based luminescence probes, 4 cellulose-based sensors, 5 nanomaterials,6–8 colorimetric sensors, 9 metal-organic frameworks (MOFs) 10 and so on,11,12 have been applied to metal ion detection in recent decades.
As a heavy metal ion, the presence of too much tin(II) ions in the body is harmful to human beings.13,14 Hence, the toxicological effects of tin(II) ions have been widely studied for about half a century. In terms of the lethal median dose in rats, McRae et al. 15 found that a dose of 8 mg Sn2+ kg−1 produced significant mortality and gross kidney damage. The outcome mentioned above was largely attributed to the tissue distribution of 99mTc-pertechnetate altered in rat bodies after treated with tin(II) ions. Meanwhile, tin(II) ions are also widely used for the preparation of 99mTc radio-pharmaceuticals16–18 and their non-radioactive premise kits. 19 Moreover, stannous chloride spectrophotometry, as a method for the determination of phosphorus, has been widely applied in different environments.20–22
Carbocyanine fluorescent compounds [DiSCn(3)] can be used as simple, rapid, and specific DNA detection reagents. They represent an important alternative test method compared with existing detection methods. The sequence-specific accelerated photobleaching reaction of these compounds in the presence of a peptide nucleic acids (PNA) probe and a genomic DNA target forms a very interesting and previously unreported photochemical basis for a sensitive, rapid, simple, and potentially inexpensive method of DNA detection.23–26 However, there are no reports regarding the treatment of these [DiSCn(3)] compounds with different metal ions, especially tin(II) ions, in aqueous solution as fluorescent probes with visible-light excitation.
A series of carbocyanine-type fluorescent compounds [DiSCn(3)] have been synthesized from 2-methylbenzothiazole. Their fluorescent intensity changes revealed that the fluorescent compounds DiSCn(3)-3 and 4 had unique selectivity toward tin(II) ions in aqueous solution. Therefore, these two compounds can be used for the detection of tin(II) ions via fluorescence sensor studies.
Results and discussion
Organic synthesis
The synthetic route to the target compounds
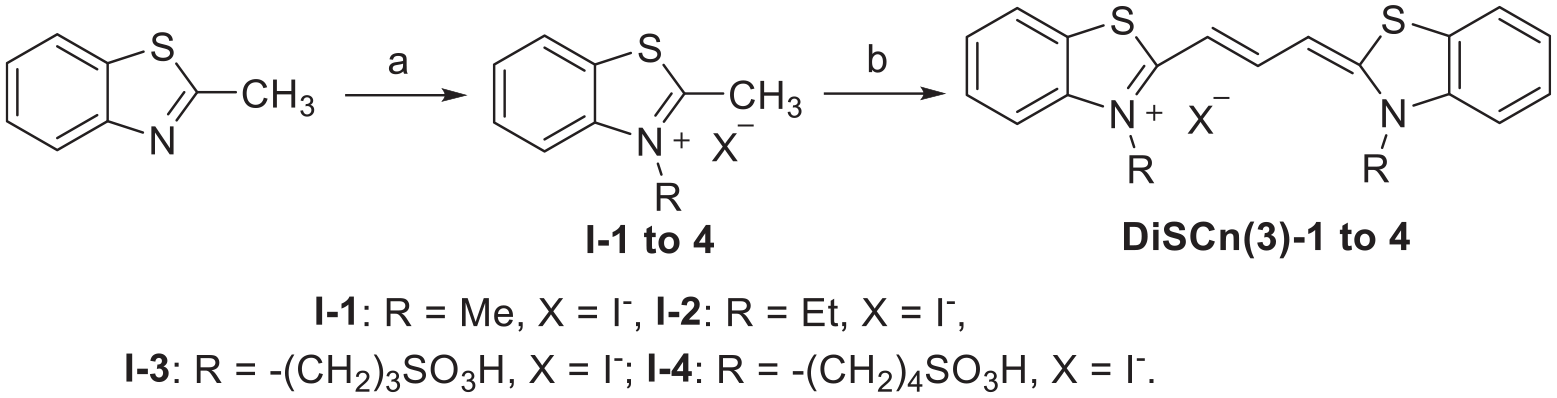
Synthesis of
Optical properties of DiSCn(3)-1 to 4
Both the UV-Vis absorption and fluorescence spectra of
Optical characteristics of compounds

The results shown in Table 1 reveal similar excitation wavelengths, emission wavelengths, and stokes shift for compounds
Response of the compounds to metal ions
The response properties of

The selectivity of
As shown in Figure 1, tin(II) ions quenched the fluorescence intensity of
Spectral properties of DiSCn(3)-3 to 4
Due to the pendent sulfonic groups, compounds
As shown in Figure 2 (left), the fluorescence spectra were measured during dropwise addition of a stannous chloride solution an aqueous solution of the prepared compounds. Because no metal ion response was observed on excitation at 556 nm, the fluorescence spectra of the compounds were obtained on excitation at 515 nm. The fluorescent intensity of compound
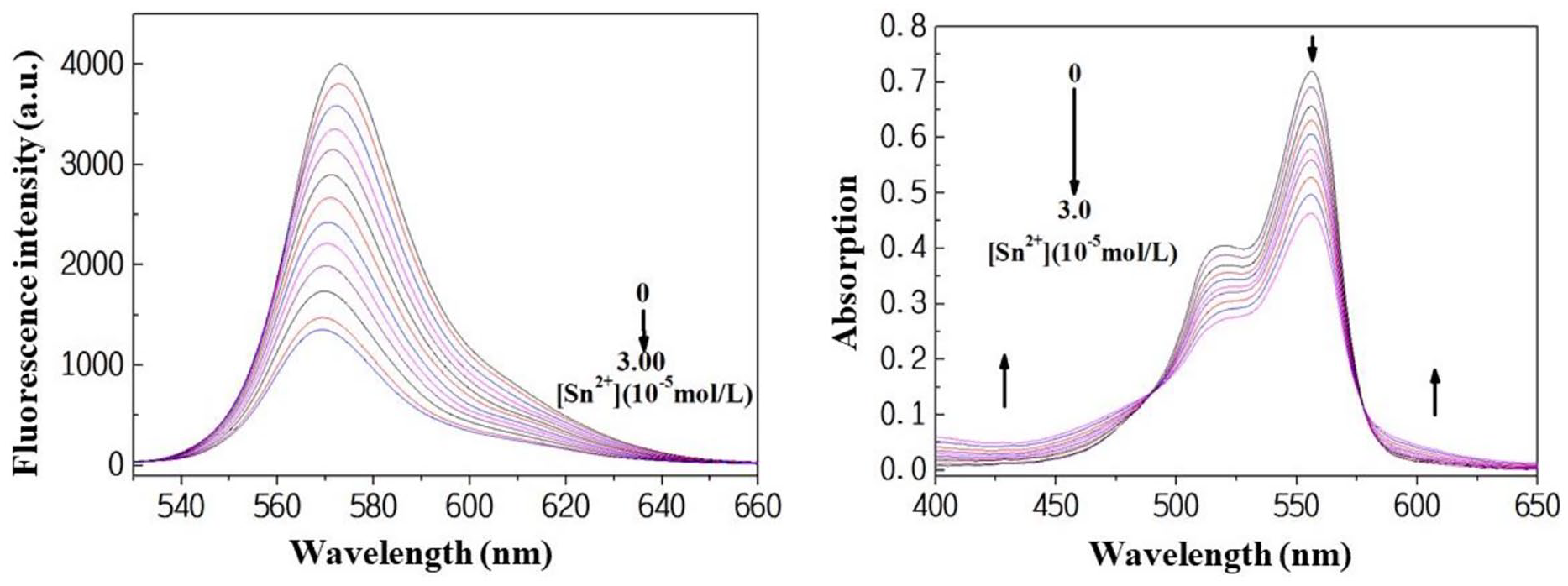
Fluorescence emission spectra of
The process described above can also be monitored by UV-Vis absorption spectroscopy. As shown in Figure 2 (right), the maximum absorption of
Similar to

Fluorescence emission spectra of
The decreasing intensity of the fluorescence and UV-Vis absorption spectra as well as the formation of two isosbestic points at 487 and 579 nm in the UV-Vis absorption spectra on dropwise addition of tin(II) ions indicated that two components existed in the system. Hence, the interaction between
These measurements suggest the formation of a chelate complex between tin(II) ions and
A plausible complexation mechanism for DiSCn(3)-3 and 4 upon addition of SnCl2
The proposed mechanism for the metal complex formation is shown in Scheme 2. The prepared probe, which has two different resonance structures, forms a chelate complex with tin(II) ions in the most stable conformation. The equilibrium between compounds

The proposed mechanism for complexation of tin(II) ions.
Conclusion
In summary, a series of novel fluorescence probes have been synthesized from 2-methylbenzothiazole and have been applied as fluorescence probes for detecting tin(II) ions in aqueous solution. The fluorescent compounds
Experimental
All chemicals and reagents were of the highest purity available and were purchased from Tianjin Jiangtian Chemical Technology Co., Ltd., and used without further purification. 2-Methylbenzothiazole, iodomethane, iodoethane, 1,3-propanesultone, 1,4-butane sultone, and triethoxymethane were purchased from Aladdin Reagent Database Inc.
1H NMR spectra were recorded on a Bruker DRX-400 AVANCE spectrometer. CDCl3 and D2O were used as the solvents. Steady-state fluorescence spectra were recorded on a Hitachi F-4500 spectrophotometer. UV-Vis absorption spectra were measured on a Shimadzu UV-3390 spectrophotometer. Elemental analysis was obtained on a Vario MAX CHN apparatus and mass spectrometry was accomplished on a Xevo G2 Q-tof HRMS apparatus.
DiSCn(3)-1 to 4; typical procedure
2,3-Dimethylbenzothiazole-3-ium iodide (I-1)
A solution of 2-methylbenzothiazole (5.00 g, 34.0 mmol) and iodomethane (4.97 g, 35.0 mmol) was stirred in ethanol (10.0 mL) at room temperature for 0.5 h. The mixture was then refluxed for 24 h. After cooling to room temperature, the resulting crystalline product was collected by filtration under reduced pressure and washed with diethyl ether until the solution became colorless. The purified product (yellow powder) was collected by filtration under reduced pressure.27–29 Yield: 86%, m.p. 239–242 °C. 1H NMR (400 MHz, D2O): δ = 3.04 (s, 3H, -CH3), 4.11 (s, 3H, N-CH3), 7.67 (t, J = 7.6 Hz, 1H, ArH), 7.77 (t, J = 7.6 Hz, 1H, ArH), 7.97 (d, J = 8.4 Hz, 1H, ArH), 8.07 (d, J = 8.0 Hz, 1H, ArH). HRMS-ESI: m/z [M + H]+ calcd for C9H11INS: 291.9657; found: 291.9654.
2-Methyl-3-ethylbenzothiazaole-3-ium iodide (I-2)
The product (yellow powder) was prepared using a similar procedure.27–29 Yield: 85%, m.p. 179–181 °C. 1H NMR (400 MHz, D2O): δ = 1.44 (t, J = 7.2 Hz, 3H, -CH2CH3), 3.05 (s, 3H, -CH3), 4.64 (m, 2H, -CH2-), 7.67 (t, J = 3.6 Hz, 1H, ArH), 7.77 (t, J = 3.6 Hz, 1H, ArH), 8.04 (m, 2H, ArH). HRMS-ESI: m/z [M + H]+ calcd for C10H13INS: 305.9813; found: 305.9808.
2-Methyl-3-(3-sulfopropyl)benzo[d]thiaol-3-ium iodide (I-3)
A solution of 2-methylbenzothiazole (1.492 g, 10.0 mL) and 1,3-propane sultone (4.48 g, 36.7 mmol) was refluxed for 8 h under a nitrogen atmosphere. After cooling to room temperature, the resulting product was collected by filtration under reduced pressure and washed with isopropanol three times. The purified product (silver-gray solid) was collected by filtration under reduced pressure.27–29 Yield: 88%, m.p. > 300 °C. 1H NMR (400 MHz, D2O): δ = 2.29 (m, 2H, -CH2-), 3.03 (t, J = 7.2 Hz, 2H, -CH2-), 3.09 (s, 3H, -CH3), 4.79 (t, J = 8.2 Hz, 2H, -CH2SO3−), 7.68 (t, J = 8.0 Hz, 1H, ArH), 7.79 (t, J = 7.8 Hz, 1H, ArH), 8.08 (m, 2H, ArH). HRMS-ESI: m/z [M + H]+ calcd for C11H15INO3S2: 399.9538; found: 399.9535.
2-Methyl-3-(4-sulfobutyl)benzo[d]thiazol-3-ium iodide (I-4)
The product (gray solid) was prepared using a similar procedure.27–29 Yield: 90%, m.p. 283–286 °C. 1H NMR (400 MHz, D2O): δ = 1.86 (m, 2H, -CH2-), 2.06 (m, 2H, -CH2-), 2.91 (t, J = 7.6 Hz, 2H, -CH2SO3−), 3.11 (s, 3H, -CH3), 4.69 (t, J = 8.0 Hz, 2H, -NCH2-), 7.70 (t, J = 8.0 Hz, 1H, ArH), 7.80 (t, J = 7.6 Hz, 1H, ArH), 8.09 (m, 2H, ArH). HRMS-ESI: m/z [M + H]+ calcd for C12H17INO3S2: 413.9695; found: 413.9691.
3-Methyl-2-((1E,3E)-3-(3-methylbenzo[d]thiazol-2(3H)-ylidene)prop-1-en-1-yl)benzo[d] thiazol-3-ium iodide (DiSCn(3)-1)
A solution of 2,3-dimethylbenzothiazole-3-ium iodide (
The symmetric tricarbocyanine dyes
3-Ethyl-2-((1E,3E)-3-(3-ethylbenzo[d]thiazol-2(3H)-ylidene)prop-1-en-1-yl)benzo[d]thiazol-3-ium iodide (
3-(3-Sulfopropyl)-2-((1E,3E)-3-(3-(3-sulfopropyl)benzo[d]thiazol-2(3H)-ylidene)prop-1-en-1-yl)benzo[d]thiazol-3-ium iodide
3-(4-Sulfobutyl)-2-((1E,3E)-3-(3-(4-sulfobutyl)benzo[d]thiazol-2(3H)-ylidene)prop-1-en-1-yl)benzo[d]thiazol-3-ium iodide (
Supplemental Material
sj-docx-1-chl-10.1177_17475198221089833 – Supplemental material for A visible-light-responsive DiSCn(3)-type fluorescent probe for the rapid, sensitive, and specific detection of tin(II) ions in aqueous solution
Supplemental material, sj-docx-1-chl-10.1177_17475198221089833 for A visible-light-responsive DiSCn(3)-type fluorescent probe for the rapid, sensitive, and specific detection of tin(II) ions in aqueous solution by Ruiji Li, Dong Wang, Xiaoyun Li, Zehui Zhang and Wei Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation (ZR2021QB124), the Outstanding Youth Fund (Overseas) Project of Rizhao City Natural Science Foundation (RZ2021ZR1), the Doctoral Scientific Research Foundation of Jining Medical University (No. 6001/600886001), and the College Students' Innovative Training Program of Jining Medical University (No. cx2020006, 202110443031).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
