Abstract
A potential candidate material for the construction of Mars habitats is concrete made from the Martian regolith and sulfur extracted from the regolith itself. Sulfur concrete, which has excellent mechanical properties, can be prepared at a low temperature (<150 °) and without water (unlike Portland-cement concrete). The surface of Mars has a much higher concentration of sulfur than those of the Earth, the Moon, or the asteroids. Sulfur on Mars, however, exists not as elemental sulfur—which is needed in concrete production—but as sulfates (usually hydrated) and sulfides. This paper surveys thermochemical and electrochemical methods that might be used to produce elemental sulfur from its compounds contained in the minerals on Mars. Possible methods include chemical or electrochemical oxidation or decomposition of sulfides, which include sulfides that exist naturally on Mars as well as sulfides that are produced via chemical or electrochemical reduction of sulfates. Some of the methods to obtain elemental sulfur—such as chemical or electrochemical oxidation or decomposition of metal sulfides or hydrogen sulfide—have already been demonstrated. The methods of producing elemental sulfur from sulfur-containing minerals on Mars will have the added benefit of generating byproducts (e.g. water, hydrogen, oxygen, and metals) that are useful for explorations of the Red Planet. In the future, chemical processes for the production of elemental sulfur may also have important industrial applications on Earth.

Introduction
Sulfur and the exploration of Mars—the ISRU approach
In human exploration of Mars, in which a round-trip duration is expected to be two and a half years, there is a need to protect the astronauts from solar and cosmic radiation. One solution during the Mars stopover (lasting for approximately 500 days) is to construct a habitat (hab) with walls thick enough to serve as a radiation shield (Figure 1). Because structural materials for such a hab would be prohibitively massive to be launched from Earth, in-situ construction materials have been considered. One candidate material is sulfur concrete, which in theory can be made from the Martian regolith and has promising mechanical properties.1,2

Our artist’s rendering of hab structure made of Martian concrete.
Sulfur is abundantly available on Mars, usually as sulfates, and, to a lesser extent, as sulfides. Unfortunately, elemental sulfur, which is required for preparation of sulfur concrete, has not been found. The literature does not address the problem of obtaining elemental sulfur from sulfur compounds on Mars, raising doubts among some researchers as to the feasibility of using sulfur concrete for Mars exploration. This paper will address the serious gap in the literature, and explore the feasibility of producing elemental sulfur using in-situ resource utilization (ISRU) techniques on Mars.
The most famous example of Mars ISRU is probably the production of methane/oxygen propellants from the Martian atmosphere (which is mostly carbon dioxide (CO2)). 3 Other well-studied ISRU examples include production of water from the hydrated minerals in the Martian regolith and production of oxygen and hydrogen from the water by electrolysis. 3 As reported in a recent review, 4 many of the Mars ISRU concepts have reached the stages of technology demonstrations. In-situ production of oxygen, for example, is currently tested in the Mars Oxygen ISRU Experiment (MOXIE) on board the Perseverance rover, which has successfully landed on Mars in February 2021. This oxygen generation technology is based on high-temperature electrolysis of CO2 in a solid oxide electrolyte (SOXE).5,6
The ISRU approach to production of sulfur on Mars poses new challenges, calling for advances in chemical and chemical engineering research. The literature on producing sulfur from sulfur-containing minerals is limited, in part because elemental sulfur is readily available on Earth, both naturally and as a byproduct of the oil and gas industry. Production of sulfur by the petrochemical industry replaced sulfur mining as the main source of sulfur, because the industrial production of sulfur is cheaper than sulfur mining, which also causes contamination of the soil and surface water in the area of the mine.7,8 As such, there has been no impetus for studies of artificial sulfur production from sulfur-containing minerals. The interest in sulfur formation from sulfur-containing minerals has been largely limited to biogeochemical processes. The objective of this paper is to survey and evaluate methods to obtain elemental sulfur from sulfur compounds found on the Red Planet.
Sulfur concrete—a candidate material for construction of Mars habitats
A round trip to Mars will take two and a half years, because the orbital motions of Earth and Mars provide limited windows for the outbound and return trips. During the Mars stopover lasting ~500 days, the crew habitats (habs) must protect the astronauts from such hazards as low pressure, low temperature, dust storms, and solar and cosmic radiation. The cosmic radiation is particularly troublesome, as the only proven mitigation method is to employ radiation shields of a few meters in thickness. Transporting the building blocks for construction of such habitats on a spaceship from Earth is prohibitively expensive. Thus, the habitats will have to be constructed, to a large extent, from materials found on Mars.
Potential in-situ construction materials for Mars habs include cold-pressed unaltered regolith and cast or sintered basalt prepared at high temperatures. Magnesium, aluminum, iron, titanium, and their alloys have also been proposed as candidate materials.
There are several types of concrete and concrete-like products that have been considered for Mars applications. The first example is the ordinary Portland-cement concrete, in which the cement is produced through high-temperature processing of regolith whereas the aggregate is produced by physical processing of rocks. The second example is geopolymer concrete, which can be made by mixing the regolith with volcanic ash and activating the mixture with alkali, for example, sodium hydroxide. The third example is polymer concrete, which may contain 5%–20% of polymer (e.g. epoxy resin, unsaturated polyester resin, polyethylene, or polypropylene) transported from Earth, and can be reinforced with glass, carbon, or steel fibers. 9 A particularly intriguing construction material for Mars—where water is scarce but sulfur is abundant—is sulfur concrete, which (unlike Portland-cement concrete) can be prepared without water. Sulfur concrete, which has been used since ancient times, is light, strong, and durable. Compared with Portland-cement concrete, sulfur concrete may have comparable (if not higher) compressive strength, bending strength, and modulus of elasticity, and it develops those strengths over a much shorter period; sulfur concrete also has lower porosity and higher resistance to frost.10,11 Unlike production of metals or alloys, the fabrication of sulfur concrete is performed at a relatively low temperature of below 150 °C, 12 and it does not require a reaction of the starting materials with reductants, such as carbon or magnesium or electrolysis of a melt. Unlike polymer concrete, sulfur concrete is highly resistant to radiation and to embrittlement at low temperatures. All these features make sulfur concrete an attractive candidate for use as an in-situ construction material on Mars. Sulfur concretes typically contain 5%–30% of sulfur by weight,13–15 and have high strength and low susceptibility to cracking even at sulfur content as low as 5%. The elemental sulfur used in sulfur concrete may be pure, or it may be modified to contain a plasticizer, such as a 1:1 mixture of monomeric dicyclopentadiene and dicyclopentadiene oligomers at a concentration of up to 5% of the weight of the sulfur. The addition of the plasticizer improves the quality of the concrete and further increases its resistance to cracking. 15
Abundance and speciation of sulfur on Mars
A major advantage of sulfur concrete as a candidate material for the construction of Mars habitats is that Mars has long been considered a sulfur-rich planet. 16 Average Martian soil has been reported to contain 6.16% sulfur on an SO3 basis, corresponding to 2.47% of elemental sulfur.17,18 Further information is provided in the works of King and McLennan 16 and Yen et al. 19 In certain Martian surface deposits and rocks, the content of sulfur is as high as 37% on an SO3 basis or 14.8% as elemental sulfur.16,20 (In general, 1% on an elemental sulfur basis corresponds to 2.497% on an SO3 basis.)
The sulfur content in the surface regolith on Mars is much higher than that on the Moon, where concentrations of sulfur are 0.05%–0.08% in lunar fines, 21 0.16%–0.27% in basalt, 0.07%–0.10% in breccia, and 0.06%–0.13% in soils. 22 The sulfur content in the Martian regolith is also much higher than the sulfur content on our home planet, where the concentration of sulfur has been reported to be ~0.035% in the Earth’s crust 23 and 0.02%–0.03% in soil. 24
The observation that the Martian surface is rich in sulfur is a reflection of a geochemical history of the surface that has been dominated by the sulfur cycle, which includes sulfur output from the surface or shallow subsurface onto rising magmas. 25 The dominance of the sulfur cycle on the Martian surface is analogous to the dominance of the carbon cycle on the Earth surface. (On the Earth’s surface, the sulfur cycle is not a dominant process due to the low sulfur content of terrestrial soils, largely caused by the leaching of a very large fraction of sulfates from eroded rocks and soils into the oceans.)
Identified sulfur-containing minerals on Mars are either sulfates or sulfides. In general, sulfate minerals are more abundant than sulfide minerals, in agreement with the strongly oxidized character of the Martian surface. Monohydrated magnesium sulfate (MgSO4∙H2O) and polyhydrated magnesium sulfate, most probably starkeyite (MgSO4∙4H2O), have been reported to be the most common and abundant hydrated sulfates observed on Mars. Several sulfate minerals belong to the (Fe,Mg)SO4∙nH2O group that includes kieserite (MgSO4∙H2O), szomolnokite (FeSO4∙H2O), and Fe(II), Fe(III), and Mg polyhydrate sulfates. Another group of sulfate minerals has the general formula CaSO4∙
Among the sulfide minerals found on Mars, pyrrhotite (Fe1-
The high sulfur content of the Martian surface may appear to suggest that producing sulfur concrete on Mars would be easy. However, there is a problem: preparation of sulfur concrete requires the elemental form of sulfur. Unfortunately, no evidence has been reported for existence of elemental sulfur on the surface of Mars.16,25
The difficulty in obtaining elemental sulfur on Mars
Until recently, elemental sulfur on Earth has been recovered as a byproduct of mining of other minerals, commonly by the Frasch process (in which superheated water is pumped underground to melt and extract sulfur). 32 There are two types of locations where deposits of elemental sulfur are found. The first type of location is salt domes containing hydrocarbons, where sulfur was formed through reduction of sulfates (such as gypsum, CaSO4∙2H2O, or anhydrite, CaSO4) by methane through the action of sulfate-reducing bacteria (SRB) on the sulfate minerals. The second type of locations is areas around hot springs and volcanoes, where veins of elemental sulfur have formed through microbial oxidation of hydrogen sulfide (H2S) emitted during volcanic activity by oxygen, as shown in equation (1)

On Mars, there are no bacteria, no free oxygen, and negligibly low concentrations of methane. Therefore, the bacterial processes that dominated the production of sulfur on Earth could not have occurred on Mars. This assumption is consistent with the observation that sulfur on Mars is present in the form of sulfates or sulfides but not as elemental sulfur. 33
Thus, obtaining elemental sulfur on Mars requires not only identification of locations with high concentrations of sulfur minerals and extraction of such minerals from the regolith, but also conversion of these minerals to elemental sulfur.
Although the literature regarding such conversion is limited, there is one study 9 which suggests that elemental sulfur can be produced on Mars, in theory, from gypsum or anhydrite using the following series of reactions



Equation (2) takes place upon heating the gypsum to a temperature around 200 °C. 34 At equilibrium, the pressure of the gaseous decomposition products of CaSO4 reaches 610 Pa (i.e. the atmospheric pressure on Mars) at a temperature of ~1200 °C. Thermodynamic calculations and experimental measurements35–37 suggested that sulfur can be obtained, in theory, from CaSO4 via equations (3) and (4).
In practice, however, the process as outlined in equations (2)–(4) is not likely to be implementable. In an experiment conducted under standard atmospheric pressure, 7% of the calcium sulfate obtained from equation (2) decomposed after heating for 3 h at a temperature of 1300 °C.37,38 The gas phase at equilibrium with heated CaSO4 contained a small fraction of SO3 and O2 relative to SO2, which amounted to less than 1% (e.g. 0.8% at 927 °C and 0.45% at 1371 °C), 36 where the equilibrium state is expressed as

As can be seen here, the amount of SO2 being produced is too small to produce elemental sulfur. In fact, in studies of thermal decomposition of CaSO4, no elemental sulfur was detected in the products even when gypsum or anhydrite were heated to temperatures as high as 1400 °C.35–37 Heating pure SO2 to 1550 °C yielded a gaseous mixture with the partial pressures shown in Table 1. 39 It can be seen in the table that the principal products of dissociation of SO2 at this high temperature are SO and O2, while the combined partial pressures of S and S2 are negligibly low, being <3 × 10−6 (i.e. <0.0003%). Accordingly, the experimental evidence shows that heating of gypsum or anhydrite (at least for cases without reductants added) is not a viable option for obtaining elemental sulfur.
Partial pressures obtained after heating pure SO2 to 1550 °C. 37

The fact that the only proposed method to extract sulfur on Mars has proved infeasible is a problem, since reserves of elemental sulfur have not been located on Mars. 33 Given the current state of knowledge, some researchers 40 doubt the feasibility of preparing Martian sulfur concrete. Other studies9,41 that point out the advantages of the Martian sulfur concrete—including its good mechanical properties and durability under Martian conditions, as well as the large abundance of sulfur compounds on Mars—have ignored the problem of obtaining the sulfur in elemental form. The possibility of using the Frasch process 32 to produce elemental sulfur on Mars was mentioned, 42 but such use is contingent upon the existence of accessible subsurface deposits of sulfur on Mars. Such deposits have not been shown so far to exist. SO3 was also mentioned as a possible source of elemental sulfur, but no methods for obtaining SO3 on Mars and converting it to sulfur were proposed. 42 Recently, the possibility of obtaining sulfur from sulfide minerals was briefly mentioned, but no specific methods for achieving this goal were discussed. 43
Prospective producers of sulfur concrete on Mars thus face a problem similar to the mariner in Samuel Taylor Coleridge’s famous poem
Developing a method for producing elemental sulfur for sulfur concrete may also offer a potential side benefit in an effort to sustain habitable environments on our home planet. The main objective of the campaign against the Earth’s climate change is to reduce reliance on the natural gas and oil, which are currently the predominant source of elemental sulfur. The amount of sulfur reserves available for mining is limited, and sulfur mining causes contamination of both the soil and the surface water around the area of the mine7,8 Accordingly, technologies to produce sulfur from sulfate minerals (e.g. gypsum) and sulfide minerals (e.g. pyrite) may become environmentally friendly alternatives in the future. Elemental sulfur is likely to have an important role in the development of not just sulfur concrete, but also other environmentally friendly materials (e.g. sulfur-based polymeric materials)44,45 and novel products for storage of energy from renewable sources (in particular, lithium–sulfur (Li–S) batteries). 46 Sulfur is also needed for its traditional applications as a source material for the production of sulfuric acid and for use as a fungicide. It has been pointed out that sulfur is present on Earth predominantly in the form of sulfates, in particular calcium sulfates (gypsum and anhydrite). Such sulfates, as well as sulfur contained in coal or in shale (i.e. oil shale and shale rich in organic matter), have not been used as sources of elemental sulfur and their utilization would require development of low-cost methods of extraction. Only elemental sulfur in evaporite and volcanic deposits, as well as sulfur associated with natural gas, petroleum, tar sands, and metal sulfides, have been used as economic resources for the production of elemental sulfur. 47 As mentioned above, the production of fossil fuels is expected to decline in the near future, and the mining of sulfur in elemental form or in sulfide form poses serious environmental problems. Accordingly, the current study, although focused on production of elemental sulfur on Mars, is relevant to the development of future methods that will assure the continuity of the supply of elemental sulfur.
Processes leading to the formation of elemental sulfur and their potential relevance to sulfur production on Mars
To determine whether it is possible to produce elemental sulfur on Mars using locally available minerals, it is helpful to be reminded that significant amounts of native sulfur are present on Earth, because understanding how elemental sulfur came to exist naturally on Earth will provide clues as to how elemental sulfur may be produced artificially on Mars. More specifically, it is necessary to look for ways to replace the processes and conditions that exist on Earth (e.g. the bacterial processes in water-containing environments) with those that may be achievable on Mars (e.g. inorganic catalysts under elevated temperatures). Accordingly, this paper describes processes known to lead to the formation of elemental sulfur on Earth and discusses the possibility of using modified versions of these processes in sulfur production under Martian conditions.
Before discussing chemical processes involved in extraction of sulfur from the sulfur-containing minerals on Mars, it should be noted that mechanical separation techniques, such as those based on differences in particle size or density, are widely used for solid–solid separations by the mining and mineral processing industries. The sulfate minerals can be separated from the regolith using differences in melting point, just as iron is separated from slag during its production from iron ore. Furthermore, extracted elemental sulfur need not be completely free of regolith impurities because the sulfur will be remixed with the regolith to make sulfur concrete. The bottom line is that the fact that the sulfur-containing minerals are surrounded by the Martian regolith would not likely pose a major difficulty. There is even a chance that a material separation mechanism is unnecessary at some locations on Mars, where such sulfate minerals as gypsum and anhydrite have been observed to form well-defined veins in locations, for example, Endeavor Crater 48 and Gale Crater, 49 which could be used for obtaining sulfates at a high degree of purity.
Chemical oxidation of H2S to elemental sulfur in the Claus process
Both on Earth and Mars, there are more sulfates than sulfides, but sulfides can be converted more easily and directly to elemental sulfur than sulfates can. Today, most of the terrestrial elemental sulfur is recovered from leftover dross from the production of oil and gas in areas of sedimentary deposits. During recovery of sour (sulfur-rich) crude oil and natural gas, which are trapped in salt domes, H2S and some limited quantities of elemental sulfur in those salt domes are removed via the Claus process. 50 This process consists of oxidation of H2S by oxygen at elevated temperatures to produce SO2, which is subsequently reacted with additional H2S to yield elemental sulfur. As such, the Claus process not only removes H2S from the products of the petrochemical industry, but also serves as the major source of production of elemental sulfur.
The Claus process consists of two steps of H2S oxidation. The first step, applied to one-third of the H2S, is a thermal step, performed at a temperature around 1050 °C

The required temperature for the oxidation of H2S by oxygen is much lower, around 250 °C, in the presence of catalysts, such as iron or vanadium on a silica support.51,52 The catalysts used in the first step (equation (6)) have no effect on the second step (equation (7)), which represents a catalytic reaction between SO2 formed during the first step and the remaining two-thirds of the H2S

This second step, which typically uses aluminum oxide as a catalyst, is carried out at a temperature between about 200 °C and 315 °C.
Using a dual-reactor system, it is also possible to reduce SO2 in stack gases to elemental sulfur without hydrogen. This process uses CO as a reducing agent, with alumina coated with copper used as a catalyst at temperatures above 427 °C. Starting from the excess concentration of CO over the concentrations of oxygen and SO2 (at a ratio of 1.03 and 1.25, respectively), SO2 can be reduced to elemental sulfur with efficiencies of 92%–97%. 53 Advanced catalysts, such as TiO2/SiO2, have been introduced in an effort to eliminate remaining traces of H2S from the gas stream. 54
Other processes of oxidation of H2S to elemental sulfur
In addition to the Claus reaction, there are other ways in which sulfides (which are obtained by bacterial or thermal reduction of sulfate or from other sources) can be oxidized to elemental sulfur. In geological environments with low levels of oxygen, the oxidation to SO2 that forms the first step of the Claus reaction does not occur naturally. Instead, a partial oxidation of H2S to S may take place through the following process 55

Utilization of H2S by phototrophic green or purple sulfur bacteria can also result in oxidation by CO2 to elemental sulfur. 56
On Earth, sulfide (e.g. H2S) can be oxidized to elemental sulfur not only by oxygen, but also by microbiological processes, where possible oxidants include sulfate in the form of H2SO4 or CaSO4∙2H2O, carbonate in the form of CO2 or HCO3-, acetate in the form of CH3COO- or CH3COOH, and N2. 57 On Mars, however, production of elemental sulfur cannot rely on SRB and sulfide-oxidizing bacteria. Even if bacteria could be introduced to and propagated on Mars—a highly unrealistic proposition given the current standards of planetary protection—a bacterially mediated process cannot be expected to produce the necessary quantities of sulfur within a reasonable time. It should be noted, however, that oxidation of sulfides under natural conditions on Earth can also take place through chemical, rather than microbiological, processes. Oxidizers that can cause sulfide oxidation through both types of processes include not only oxygen, but also nitrate, ferric iron,58–62 MnO2,60,61 V(V), 62 Cu(II), 62 and so on. In both types of processes, the oxidation of sulfide often stops at a redox level of S(0)60,61 and does not go all the way to S(VI).
The Martian atmosphere consists of 95% CO2, but there is virtually no oxygen. As such, of particular interest for Mars exploration is production of sulfur through oxidation of H2S by CO2. (The process is carried out at elevated temperatures of >600 °C.) It has been found that when H2S is oxidized by CO2 to obtain S, H2O is one of the products63,64

or one of the reactants 65

The catalysts that have been shown to be effective in these reactions include MoS2, 66 MgO, CaO, γ-Al2O3, 63 and FeS. 65 The roles of these catalysts can be shown as reactions involving the catalysts. For example, the catalytic role of FeS in equation (10) stems from a cycle consisting of the two reactions 65
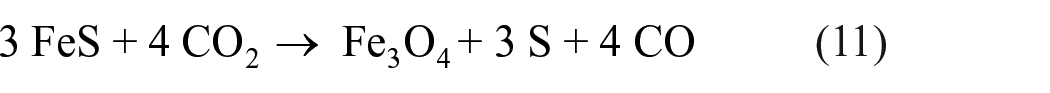

Chemical oxidation of metal sulfides to elemental sulfur
In addition to H2S, metal sulfides can also serve as sources for elemental sulfur production. The use of CaS and other sulfides to form elemental sulfur upon oxidation by SO2 is described in detail in the “Reduction of SO2 to elemental sulfur” section. Oxidation of iron sulfides is another known source of sulfur: as practiced since ancient times in China, oxidation of pyrite (FeS2) heated to above 600 °C in the presence of atmospheric oxygen yields elemental sulfur as a product. 67
In Earth’s geology, sulfur is obtained through oxidation of sulfides, suggesting a possible route to artificially obtain sulfur on the Red Planet. It has been shown that elemental sulfur is a main product of weathering of pyrrhotite in an atmosphere containing CO2 and H2O. 68 Observations on geological formations in the Arctic, where temperatures are not much higher than on Mars, show that permafrost provides an environment where oxidation to sulfate is slowed down; the central zone of these formations contains a combination of pyrite and elemental sulfur, while the surrounding surface is dominated by gypsum and jarosite. 69 As these studies show, the lack of elemental sulfur on the Martian surface can be explained by the lack of water on the Martian surface.
Water on Mars may exist as underground ice, however. The Phoenix mission and more recently the SWIM project have detected the indication that some subsurface locations on Mars harbor a significant amount of frozen water. 70
Thermal decomposition of sulfides
Decomposition of H2S into hydrogen and sulfur requires temperatures above 1000 °C, 71 but the use of catalysts permits carrying out the reaction at much lower temperatures. In the presence of transition metal sulfides, H2S heated at temperatures above 400 °C decomposes into hydrogen and sulfur. 72
Thermochemical decomposition of H2S can be promoted by the use of metals or metal sulfides with low sulfur-to-metal ratios. Upon reaction with H2S, such metals or low sulfur sulfides would form sulfides with high sulfur-to-metal ratios, which in turn decompose to produce sulfur and regenerate sulfides with low sulfur-to-metal ratios73,74
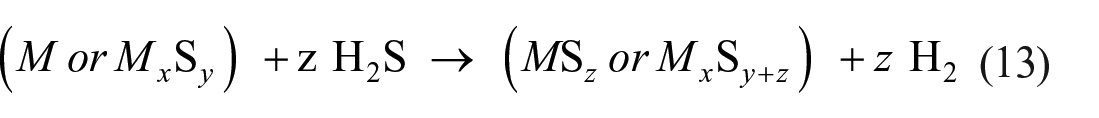

where

The decomposition of H2S by a closed-loop thermochemical decomposition in combination with iron sulfide has been studied. The loop consists of two steps. In the first step, H2S decomposes by reacting with iron sulfide to produce hydrogen and a solid solution consisting of FeS and S2. In the second step, the solid solution is decomposed at high temperature to recover sulfur and iron sulfide. The iron sulfide is then reused in the first step. 75
Overall, the decomposition of H2S has not attracted much interest in the literature, but it is now the subject of extensive research because of its potential for use in large-scale production of hydrogen. In recent years, novel catalysts have been developed to facilitate this reaction.76–81 The decomposition of H2S was also effected under ambient conditions using non-thermal plasma 82 or photocatalytic methods.83,84
Alkali sulfides and alkaline earth sulfides are not viable sources for elemental sulfur, because they are stable enough to prevent their thermal decomposition. For example, Na2S, K2S, MgS, and CaS melt at temperatures of 1176 °C, 840 °C, 2000 °C, and 2525 °C, respectively—all without being decomposed. Sulfides of transition metal ions, on the other hand, decompose at much lower temperatures. Thus, FeS2 (pyrite or marcasite), FeS (troilite), and NiS (millerite), which are all present on Mars, can be thermally decomposed to yield elemental sulfur.73,85 On Earth, some elemental sulfur is produced by the mining industry from sulfide minerals, for example, pyrite or marcasite (forms of FeS2) and pyrrhotite (Fe1-



As it turns out, the amounts of FeO from equation (17) and Fe3O4 from equation (18) are comparable. The iron oxide products of these reactions can be used as a source of iron metal if they are reduced with hydrogen at elevated temperatures. 89
Both pyrite and FeS heated on carbon supports in a helium environment decompose to yield elemental sulfur. The onset of the major part of the decomposition takes place at temperatures of 420 °C–500 °C. 90
Electrochemical oxidation and decomposition of sulfides
In addition to the thermochemical methods, H2S can be decomposed also by electrochemical, photochemical, and plasma methods. 73 It was found that electrochemical oxidation of dissolved H2S gas, present in wastewater, could be used to produce sulfur and water at 900 °C using anode electrocatalysts consisting of transition metal sulfides. 91 Dissolved metal sulfides, such as Na2S, were decomposed in electrochemical membrane cells with titanium anodes to yield hydrogen and elemental sulfur. 92 Sulfides contained in wastewater were oxidized electrochemically by either using oxygen generated by the electrolysis or by injecting oxygen into the electrolytic cell. In both cases, the oxidation took place on the surface of titanium electrodes coated with metal oxides (e.g. TaO2/IrO2 or SnO2).93,94
A substantial body of literature describes processes leading to oxidation of sulfide to elemental sulfur and to higher oxidation states of sulfur. As discussed earlier, many of the processes described in the literature concern either biochemical processes (which are irrelevant to the Martian applications) or industrial processes involving oxidation of H2S (a byproduct of natural gas production). Some of these studies involved electrochemical processes. Electrochemical studies have shown that the discharge of Li–S batteries (i.e. Li2S) can be re-oxidized to elemental sulfur. The re-oxidation is catalyzed by certain metal sulfides (in particular, TiS2, CoS2, and VS2). Such catalytic activity is associated with polysulfide binding on the surface of those metal sulfides. 95
When sulfides, such as pyrite (FeS2) or galena (PbS), were electrochemically oxidized in a neutral solution, the products were found to include elemental sulfur and polysulfides. 96
The most relevant conditions for electrochemical reduction of metal sulfates in Martian environments involve, as detailed in the “Electrochemical reduction of sulfate” section, electrolysis of molten salts in non-aqueous systems. A similar type of electrolysis may be used for oxidation of sulfides. Thus, electrolysis of tungsten disulfide (WS2) in the form of pellets serving as a cathode in a NaCl–KCl melt at 700 °C produced metallic W and bubbling of gaseous S2, which, upon cooling, condensed to form solid sulfur (S8) as the final anodic product.
97
Electrochemical sulfur removal from chalcopyrite (CuFeS2) was achieved in a similar NaCl–KCl melt at 700 °C with CuFeS2 being used as a cathode. Following formation of intermediate
Sulfate reduction—processes and products
On the Earth surface, elemental sulfur exists only in small quantities relative to those of sulfides and sulfates. On the Martian surface, elemental sulfur has not been found, but sulfides and sulfates are abundant. Just as on Earth, much more sulfur is present in sulfates than in sulfides on or near the surface of Mars. To obtain elemental sulfur on Mars, therefore, methods to reduce sulfates to elemental sulfur must be investigated.
Given its important role in the evolution of the geology and biology of the Earth system, sulfate reduction has been extensively studied. In nature, there are two types of sulfate reductions: bacterial sulfate reduction (BSR) and thermochemical sulfate reduction (TSR).
BSR processes, which are common in geologic settings at temperatures between 0 °C and 60 °C–80 °C, take place in the presence of SRB, which are abundant under anaerobic conditions on Earth. 102 Although there are no bacteria on Mars, the BSR processes on Earth are still informative, because they demonstrate that a given reaction can take place if a suitable catalyst can be identified. TSR processes, which are common in geologic settings at temperatures of 100 °C−180 °C (usually <140 °C), 55 are also carried out in industrial operations (at much higher temperatures). The technological maturity of an artificial TSR indicates its potential in the Mars exploration scenarios.
While oxidation of sulfides often stops at the stage of elemental sulfur formation,60,61 a large majority of the processes of sulfate reduction proceed to the formation of sulfide or SO2. To obtain elemental sulfur, therefore, these processes must be followed by oxidation of sulfide (which is discussed in the following titled “Chemical oxidation of H2S to elemental sulfur in the Claus process,” “Other processes of oxidation of H2S to elemental sulfur,” “Chemical oxidation of metal sulfides to elemental sulfur,” “Thermal decomposition of sulfides,” and “Electrochemical oxidation and decomposition of sulfides”) or reduction of SO2 (which is discussed in the “Reduction of SO2 to elemental sulfur” section).
Sulfate reduction by hydrogen
A comprehensive study of the reduction of anhydrous sulfates by hydrogen gas at temperatures 200 °C−900 °C showed that five types of reduction products may be obtained 103 :
CuSO4, HgSO4, and Fe2(SO4)3 are reduced to Cu2SO4, Hg2SO4, and FeSO4, respectively.
Cu2(SO4)3 and Ag2SO4 are reduced to metallic Cu and Ag, respectively.
NiSO4 and CoSO4 are reduced to mixtures of Ni3S2 + SO2 and Co3S8 + SO2, respectively, while CdSO4 is reduced to CdS, and Na2SO4 is reduced to Na2S.
Al2(SO4)3 and MgSO4 are reduced to mixtures of Al2O3 + SO2 and MgO + SO2, respectively.
MnSO4, ZnSO4, PbSO4, and FeSO4 are reduced to mixtures of MnO + MnS + SO2, ZnO + ZnS + SO2, PbO + PbS + SO2, and FeO + FeS + SO2, respectively.
Types 1 and 2 do not involve reduction of the sulfate anion. However, since it is easier to convert SO2 and sulfides to elemental sulfur than to reduce the original sulfates to sulfur, the sulfates that belong to Type 3 (and, to a lesser extent, Types 4 and 5) should be studied as potential sources of elemental sulfur under dry conditions. Whether any of these five types are applicable on Mars will depend on whether sufficient quantities of those sulfates are available at the production site.
Hydrogen is also used in reduction of solid sodium sulfate (Na2SO4) to produce sodium sulfide. As in the case of reduction by CO at temperatures of 650 °C−750 °C, the reduction by hydrogen is slow, but it can be made much faster in the presence of sodium titanate. 104
To use hydrogen to reduce Na2SO4 to sodium sulfide, Na2SO4 is first dissolved in a molten mixture of alkali carbonates (Li2CO3 + K2CO3 + Na2CO3) and the temperature is raised to 600 °C−840 °C. This reaction can be performed using iron or sulfide as a catalyst

In this equation,
Under anaerobic conditions, SRB can reduce sulfate with such agents as hydrogen gas 50 according to the equation

Potential use of sulfate reduction by hydrogen on Mars
The need for availability of hydrogen for the reduction of sulfates appears at first to be a serious obstacle. However, hydrogen is going to be required in any case for in-situ production of rocket propellant. 3 Given the advantage of in-situ methane propellant production, NASA has even considered an exploration scenario in which a hydrogen tank would be shipped from Earth. 4 In recent years, however, it is increasingly considered plausible to obtain hydrogen on Mars, that is, by electrolysis of melted water ice or of water expelled from hydrated minerals.3,4
There are also ways to mitigate the need for hydrogen. The stoichiometry of a reaction leading to reduction of sulfates to sulfur with hydrogen, whether such reaction consists of a single step or multiple steps, could be described by the equation
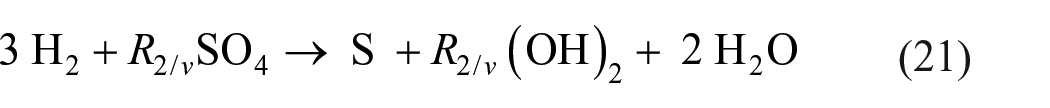
In this equation,

Accordingly, sulfate reduction requires only 6 g of hydrogen gas to obtain 32 g of elemental sulfur. Furthermore, the water generated in the reaction may be captured and electrolyzed to recover two-thirds of the hydrogen used. The rest of the hydrogen can be recovered through decomposition of the hydroxide.
Fortunately, most of the sulfate minerals on Mars are hydrated: there is a strong correlation between the locations of sulfur compounds and H2O across the surface of Mars.25,108,109 If, as might be expected, the reaction with hydrogen requires heating to a temperature at which the water is expelled from the hydrated sulfate, this water could be captured and electrolyzed to produce hydrogen. In the case of hydrated salts, the mixture of sulfate salts used in the reaction can be represented in the form of
Bedrocks exposed in craters in the region explored by the Opportunity rover were sedimentary rocks with a high concentration of sulfur in the form of calcium and magnesium sulfates. Sulfates that may be present include hydrated sulfate salts, such as kieserite (MgSO4∙H2O), bassanite (2CaSO4∙H2O), epsomite (MgSO4∙7H2O), and gypsum (CaSO4∙2H2O). The water of hydration of these minerals can be very useful in electrochemical processes because this water can serve as a source of hydrogen (as discussed above).
Sulfate reduction by carbon monoxide
It was found that carbon monoxide can be used to remove heavy metals from sulfate-rich wastewater by reducing the sulfate in the presence of SRB to form insoluble metal sulfides. 110 Biological reduction of sulfate has also been performed using both the hydrogen and CO components of producer gas (a combination of H2 + CO + CO2), and the resulting H2S product was oxidized with Fe3+ ions to produce elemental sulfur. 111 Sodium sulfate can be thermally reduced by CO at temperatures of about 930 °C−1040 °C. 112 At temperatures of 670 °C−750 °C, the reaction is slow, but it becomes much faster in the presence of sodium titanate. 113 The product of the reaction is sodium sulfide, which can be subsequently oxidized to elemental sulfur.
Potential use of sulfate reduction by carbon monoxide on Mars
CO2, which is the predominant component of the Martian atmosphere, has been shown in space applications (e.g. SOXE mentioned above) to be readily decomposed to CO and oxygen.5,6 In addition to the electrochemical SOXE method,5,6 photochemical or photoelectrochemical reduction of CO2 to CO under mild conditions using sunlight and trinuclear nickel catalysts, such as Pt(Ni3)2, has been proposed as a method for generating CO from CO2 on Mars. 114
Sulfate reduction by methane and other organic compounds
As mentioned above, H2S and limited quantities of elemental sulfur found in salt domes are a major source of sulfur on Earth. H2S and elemental sulfur in these formations were formed upon reduction of sulfates by methane through the action of SRB.
Native sulfur deposits on Earth are often formed in basins that contain hydrocarbons or in areas exposed in the past to volcanism. 57 In such basins, hydrocarbon-bearing beds lie under beds of CaSO4 or CaSO4∙2H2O. When combinations of hydrocarbons, bacteria, and water rise into the calcium sulfate beds, they reduce sulfate ions to H2S, and alter gypsum (CaSO4∙2H2O) to CaCO3.115,116 The formation of H2S during a sulfate/hydrocarbon reaction can be represented by the reaction 115
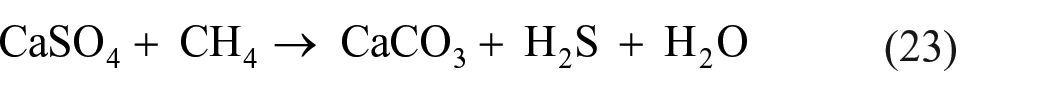
H2S in equation (23) is subsequently converted into polysulfides, which in anaerobic environments can be oxidized by CO2 to elemental sulfur. 115 In aerobic environments (e.g. oxygenated surface water), H2S can be oxidized by O2. As can be seen in equation (23), the bacterial reduction of sulfates to H2S is achieved through sulfate–hydrocarbon interaction. Another source of elemental sulfur deposits is thermal, rather than biological, oxidation of H2S formed in a sulfate–hydrocarbon reaction. Volcanic activities facilitate such thermal oxidation of H2S taking place at temperatures above 140 °C. 116 Elemental sulfur may be obtained from volcanic H2S being oxidized by atmospheric oxygen, by oxygen dissolved in groundwater, or by volcanic SO2 in combination with water.
Sulfate reduction to sulfide by methane is a key process in anaerobic (e.g. marine) environments,117,118 but SRB processes can reduce sulfates even in aerobic environments. 119 Sulfate reduction by methane in geological formations on Earth takes place not only by BSR, but also by TSR, which is more relevant to applications on Mars. The product of the reaction is H2S. 120
Under anaerobic conditions, SRB can reduce sulfate with such agents as methane, methanol, and various organic molecules. 121 Sulfate reduction by methanol proceeds as
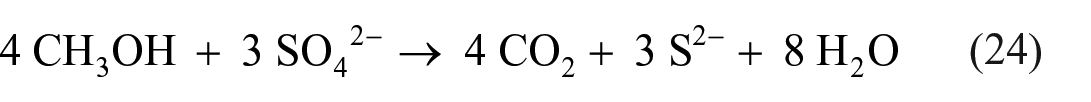
Bacteria are not available on Mars, but thermochemical alternatives may be feasible. Indeed, TSR by natural gas to H2S and polysulfides is a well-known process, starting at temperatures as low as 175 °C 122 or even 140 °C. 123 As mentioned above, H2S and, in particular, polysulfides can be readily oxidized to elemental sulfur.
Hydrocarbons other than methane can also cause sulfate reduction through BSR or TSR. In geological settings, the main organic reactants for BSR are organic acids and other products of aerobic or fermentative biodegradation. The main organic reactants for TSR are branched and
Potential use of sulfate reduction by methane on Mars
The concentrations of organic substances (methane and halogenated hydrocarbons) on Mars are in the sub-parts-per-million range.127,128 However, methane, which is much more convenient to store and use than hydrogen, can be produced artificially. Methane is produced on the International Space Station, where CO2 exhaled by the astronauts is converted into methane and water through the Sabatier reaction 129

The reaction requires a moderately elevated temperature of 400 °C. On Mars, there is no shortage of CO2, which constitutes 95% of the atmosphere, and hydrogen could be produced from hydrated minerals. 130 It should be noted that because methane can be used as a propellant for return rockets, in-situ production of methane from the Martian atmosphere has long been a part of NASA’s Mars exploration strategy. The fact that there is already a plan to produce a large quantity of methane using the Sabatier reaction—a proven process that has been in practice since the 19th century and already tested in space missions—is another promising aspect of using methane to process Martian regolith to obtain elemental sulfur. Sulfate reduction by organic species other than methane does not appear to have significant potential for sulfate reduction under Martian conditions.
Sulfate reduction by strong inorganic reductants
Sulfuric acid is easier to reduce than sulfate ions: in an acidic environment, the standard electrode potential of the reaction

is +0.172 V, and thus, it is much higher than the corresponding potential of −0.936 V for the reaction 131
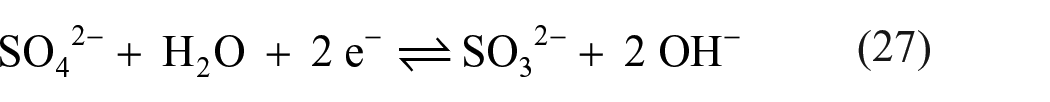
Furthermore, a reducing solution consisting of a mixture of stannous chloride and concentrated phosphoric acid was used to reduce sulfate in CO2 environments where temperatures were gradually raised from 120 °C to 300 °C. This method is effective in reducing both soluble sulfates, such as Na2SO4 and insoluble sulfates, such as BaSO4. 132 Likewise, a reducing solution consisting of a mixture of a combination of hydroiodic acid (HI) and sodium hypophosphite (NaH2PO2) at a temperature of 124 °C quantitatively reduces Na2SO4 to H2S. When this method was applied to barium sulfate (BaSO4) rather than Na2SO4, the reduction process was found to be much slower and incomplete. 133 At any rate, the use of such a strongly reducing solution (i.e. a mixture of stannous chloride and concentrated phosphoric acid) is unrealistic in a Mars expedition context, as there is no practical way to import or prepare the necessary quantity of the acidic solution on the Red Planet.
Na2SO4 is reduced to sodium sulfide by carbon using the kraft process, which is designed to recover chemicals (Na2SO4, sodium carbonate) and energy used in pulping wood by concentrating the residual pulping solution and then burning the concentrated solids in a furnace at temperatures around 850 °C. 134 The reaction is
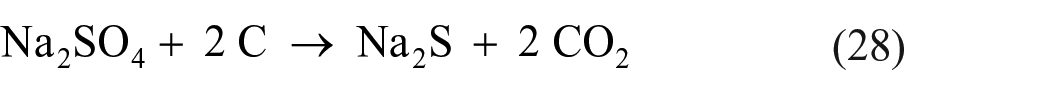
The presence of elemental carbon on Mars has not been reported, and conversion of CO2 to elemental carbon requires strong reducing conditions, such as a reaction with hydrogen over an iron catalyst at temperatures between 530 °C and 730 °C 135 or electrolytic reduction on liquid metals. 136 Thus, the use of reagents other than hydrogen, CO, methane, and H2S in sulfate reduction does not appear to be suitable for applications on Mars.
The reaction between sulfates and H2S—a combination of H2S oxidation and sulfate reduction
In geologic environments on Earth, sulfates can be reduced by H2S to elemental sulfur in the presence of water at low pH55,115,116,137


These reactions become more favorable as the temperature increases. 138 In the presence of hydrocarbons, the elemental sulfur formed in these reactions is reduced back to H2S, but if hydrocarbon levels are low, elemental sulfur can accumulate. 55
If the sulfate is present in the form of gypsum rather than sulfate ions or sulfuric acid, the reaction can be represented by the equation 57
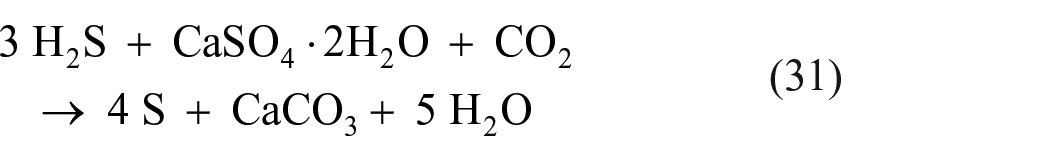
Since CO2 is the main component of the Martian atmosphere and hydrated sulfates (e.g. gypsum) are abundant, this reaction can end up producing more water than it consumes. Therefore, this reaction can be considered one of the candidate processes for producing elemental sulfur on Mars.
Reduction of SO2 to elemental sulfur
Both sulfide and sulfate minerals can be directly converted to SO2 at elevated temperatures, and thereafter used to produce elemental sulfur. Pyrite can be converted to SO2 upon roasting of pyrite or other iron sulfides in the presence of excess oxygen at temperatures of 600 °C−1000 °C 139
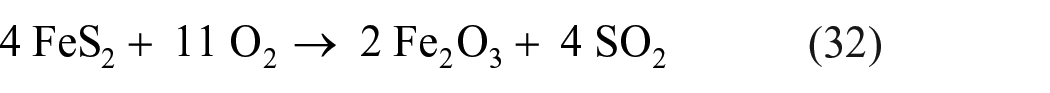
The resulting SO2 can be reduced to elemental sulfur. Equation (32), in other words, is an intermediate step to obtaining elemental sulfur from pyrite. However, in the context of sulfide oxidation, going through SO2 as an intermediate step offers no advantage over a single-step pyrolysis of pyrite in the absence of oxygen. In contrast, going through an intermediate step involving SO2 formation can be quite useful in the preparation of elemental sulfur from sulfates, since, as detailed above, direct sulfate reduction generally leads to formation of sulfides rather than elemental sulfur. Depending on the conditions of the reaction, the reduction of sulfates may lead to formation of SO2 rather than sulfides, providing an alternative route for the production of elemental sulfur. Thus, sulfate minerals can be directly converted to SO2 at high temperatures. 140 Reduction of calcium sulfate (anhydrite, which is produced upon thermal dehydration of gypsum) to SO2 with carbon monoxide, hydrogen, or methane can be performed at temperatures of 1050 °C−1150 °C 37



Upon using coke as a reductant in the presence of sand, the process produces calcium silicate cement rather than CaO 141

The reaction between H2S and SO2 (described in the “Chemical oxidation of H2S to elemental sulfur in the Claus process” section) provides a route for production of elemental sulfur through the reduction of sulfur in an oxidation state of +4 by sulfur in the −2 state.
Sulfur dioxide can also be reduced to elemental sulfur in other processes. On Earth, SO2 can be reduced to H2S by organic reducing agents, such as lactate in the presence of certain SRB. 142 Likewise, sulfur dioxide can be directly reduced to elemental sulfur by methane over ceria-based catalysts at 550 °C−750 °C at ambient pressure. 143 If a nickel–ceria catalyst is used, the products of the reaction are H2S and CO, but if the catalyst is copper–ceria, the products are elemental sulfur and CO2. 143 Sulfur dioxide can also be reduced to elemental sulfur over cobalt oxide supported on alumina or other carriers at 840 °C, 144 or over a MoS2 catalyst at 600 °C.145,146 Sulfur dioxide can also be directly reduced to elemental sulfur by CO over composite Cu–Ce–O catalysts at temperatures around 450 °C 147 or over transition metals/fluorite-type oxide catalysts. 148 Reduction of SO2 to elemental sulfur is also possible using hydrogen or hydrogen/CO mixtures in activated carbon beds at temperatures below 800 °C. 149 Reduction of SO2 using hydrogen has been performed using catalysts, such as SnO2–ZrO2 around 550 °C 150 or Co–Mo/Al2O3 around 300 °C. 151 The latter study is particularly interesting, because it leads to the conclusion that a metal sulfide phase in the catalyst hydrogenates SO2 to H2S—while the alumina support catalyzes the reaction between H2S and SO2 to form elemental sulfur. The latter reaction is the second step of the Claus reaction152,153 described in detail in the “Chemical oxidation of H2S to elemental sulfur in the Claus process” section.
In the catalytic reduction of SO2 to elemental sulfur with hydrogen or CO using sulfide-treated Co/Al2O3 or Co/TiO2 catalysts, cobalt sulfide generates H2S (with hydrogen as reductant) or COS (with CO as reductant) as an intermediate. In a second step, the intermediate reacts with SO2 over the Al2O3 or TiO2 component of the catalyst to form elemental sulfur147,154

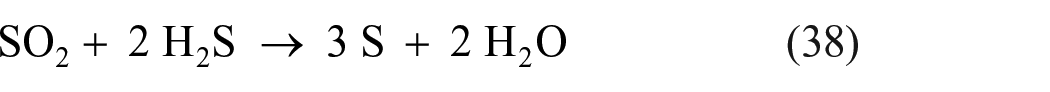
or



Another method, which has a potential application in the setting of missions to Mars, includes reduction of SO2 using methane in a radiofrequency (RF) plasma reactor. 155
Finally, there is also a method using a cyclic process involving CaS and CaSO4, 124 both of which may be found or generated on Mars (see above). This cyclic process takes place at temperatures around 730 °C−830 °C, where CaSO4 pellets used as the starting raw material are reduced by a suitable reducing agent, such as hydrogen to produce CaS pellets, which are then used to reduce SO2 to produce elemental sulfur vapor and solid CaSO4. The CaSO4 is then reduced to regenerate the CaS. If CaS is found in significant quantities on Mars, it could be used as the raw material, avoiding the need to reduce CaSO4.
Electrochemical reduction of sulfate
When dilute solutions of sulfate salts or other soluble salts are electrolyzed, the products are hydrogen and oxygen. When highly concentrated solutions of alkali sulfates (e.g. Na2SO4 in cells containing diaphragms to separate the anodic half-cell from the cathodic half-cell) are electrolyzed, the products are sulfuric acid and the corresponding hydroxide (e.g. NaOH). 156 Acidifying Na2SO4 solutions with a small amount of H2SO4 facilitates the electrolytic dissociation of concentrated Na2SO4 solutions into NaOH and H2SO4. 157 Because the reduced product of the electrolysis is sodium metal, which immediately reacts with water to form NaOH and hydrogen gas, no reduction of sulfate is achieved. Furthermore, the preparation of such solutions requires large amounts of water, which is a scarce resource on Mars. Thus, electrolysis in aqueous media is not a promising route for sulfate reduction on Mars.
Studies have shown that it is possible to reduce sulfate to sulfide in bioelectrochemical cells using carbon felt electrodes in the presence of SRB. 158 As discussed above, however, any method requiring bacteria is inapplicable to Martian environments.
Electrolysis of sulfates in non-aqueous media, especially those consisting of molten salts, cannot be ruled out as a possible route of sulfate reduction. Aluminum, for instance, is produced through electrolysis of molten reactants. 159
In the case of sulfates, measurements of electrode potentials of various metal electrode systems, including Cu(I)/Cu(0), Cu(II)/Cu(I), Pd(II)/Pd(0), and Rh(III)/Rh(0) were performed against an Ag(I)/Ag(0) reference electrode in a eutectic melt of Li2SO4 and K2SO4 (80% and 20%, respectively) at a temperature of 625 °C. 160 When the melt was electrolyzed between the two platinum electrodes, the anodic reaction appeared to be
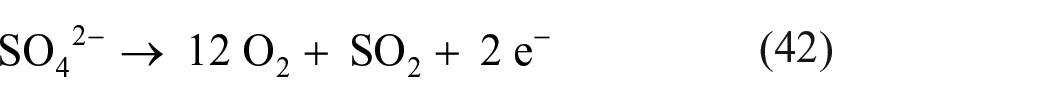
while suggested cathodic processes included


and, possibly, to a lesser extent

Thus, in the case of electrochemical (rather than regular chemical) reduction, direct formation of elemental sulfur is a possibility but elemental sulfur is not a major product. 159 As shown earlier, however, even if elemental sulfur is not produced in significant quantities, SO32- or S2- could be used as precursors of elemental sulfur. It should be noted that elemental sulfur and oxide ions (with a small amount of sulfite) were found to be the main products of electro-reduction of a molten Li2SO4–Na2SO4–K2SO4 mixtures between platinum electrodes at 550 °C, while the addition of active metals, such as Mg, caused the reaction to produce sulfide and oxide ions. 161 Theoretical calculations 162 have indicated that in the presence of Mg or another active metal in a Na2SO4 melt at a temperature below 1700 K the favored reaction is

The studies have shown that, when molten sulfates are electrolyzed in an atmosphere of oxygen with added 0.1% SO2, sulfate ions can be directly reduced to sulfide ions and to elemental sulfur. Similarly, molten LiCl–KCl–Na2SO4 (with a melting point of 700 °C) under inert argon, a condition more relevant to that on Mars, can be electrochemically reduced. 163
Electrochemical reduction of molten sulfates does not require water, and the high temperature facilitates the electrolysis. If a process based on electrolysis of molten sulfates could be developed for sulfate reduction, electrochemical reduction would have significant advantages over other approaches. The reduction method would probably involve a relatively abundant sulfate phase, such as a calcium or magnesium sulfate compound, dissolved in a melt of alkali-containing sulfate phase. Alkali-containing sulfate minerals are preferable for use as solvents because of the high polarity of their melts and their relatively low melting temperatures. Alkali-containing sulfates found in the region of Mars explored by the Opportunity rover include glauberite (Na2Ca(SO4)2), bloedite (Na2Mg(SO4)2), and vanthofite (3Na2SO4∙MgSO4).164,165 To lower the melting point of the mixture, it may be necessary to add such non-sulfate salts as chlorides or carbonates. Chlorides, particularly in the form of halite (NaCl), have been identified on the surface of Mars166–168 with the reported chloride content in Martian soils being 0.68%.17,18 Development of a process involving addition of non-sulfate salts to reduce the melting point would require evaluation of the availability of candidate minerals on Mars. Identification of potential solvent–solute systems is also necessary.
It has been pointed out that electrolysis of magnesium sulfate minerals on Mars is a potential resource for obtaining magnesium metal. 169
Byproducts of elemental sulfur production
An additional benefit of producing elemental sulfur is that the byproducts can be extremely useful for human exploration of Mars. Table 2 summarizes potential methods of obtaining elemental sulfur and the corresponding byproducts.
Summary and possible useful byproducts of methods of elemental sulfur production.

Conclusion
Given that sulfur compounds are abundant on the Martian surface, sulfur concrete has been considered as a promising in-situ construction material for habitable structures on Mars. Preparation of sulfur concrete, which has excellent mechanical properties, is relatively easy, as all it requires is mixing molten sulfur and Martian regolith, a process that can be carried out at a moderate temperature (<150 °C). Preparation of sulfur concrete, unlike that of Portland-cement concrete, does not require water which is scarce on Mars. On the Martian surface, however, sulfur is present not as elemental sulfur, which is needed to make concrete, but in the form of sulfates and, to a lesser extent, sulfides. Thus, the key step in preparing Martian sulfur concrete is to obtain elemental sulfur from those sulfates or sulfides. The goal of this paper is to review the literature and identify candidate methods to obtain elemental sulfur in the Mars exploration scenarios.
Unfortunately, the literature concerning technological production of elemental sulfur is limited. This limitation may stem from the fact that, here on Earth, elemental sulfur can be mined from natural deposits or produced as a byproduct during oxidation of H2S emitted by the oil and gas industry. A large majority of the available information on sulfur production concerns bacterial processes, which are primarily of interest in biology and paleontology—but not applicable for the Mars exploration scenarios. However, the fact that bacterial catalysts allow elemental sulfur to be produced from sulfate minerals under mild conditions (through a bacterial reduction of sulfate minerals to sulfides by hydrocarbons followed by bacterial oxidation of the sulfides to sulfur by oxygen) indicates the likelihood of finding thermochemical alternatives.
The first category of processes to obtain elemental sulfur is chemical oxidation or decomposition of sulfides. Production of sulfur through thermal oxidation of pyrite by oxygen is well-known. Obtaining sulfur upon heating of marcasite and troilite has also been demonstrated. Sulfide minerals are present at certain locations on the surface of Mars in significant quantities (although less abundant than those of sulfates). Thermal decomposition of sulfides is a promising method, especially during the initial period of Mars exploration. Oxidation of sulfides may involve the use of oxygen or alternative oxidizing reagents, such as CO2 or sulfates, available on Mars or SO2 produced by sulfate reduction. Unlike sulfide oxidation, thermal decomposition of sulfides (including H2S) is advantageous because the process does not require an oxidizing agent and because elemental sulfur can be produced without competition with SO2 or SO3 as alternative products.
The second category of pathways to produce sulfur on Mars involves electrochemical oxidation or decomposition of molten sulfides. Multiple studies have demonstrated the feasibility of obtaining elemental sulfur in high-temperature electrolysis of sulfide melts. To apply this technique on Mars, it would be necessary to identify sulfide minerals or mixtures of minerals that do not require excessively high temperatures to produce melts suitable for electrolysis. In addition to the thermal and electrochemical processes, other sulfide decomposition methods may include photochemical and plasma processes.
The third category of processes investigated in this paper is chemical reduction of sulfates to elemental sulfur using hydrogen, carbon monoxide, or methane as a reducing agent. There are currently very few known cases (in particular, reduction by H2S) where elemental sulfur can be obtained from sulfate in a single-step process. In general, the products of known processes involving chemical reduction of sulfates include such compounds of sulfur as H2S or metal sulfides (with sulfur in the oxidation state of −2), which can be oxidized to elemental sulfur, and SO2 (with sulfur in the state of +4), which can be reduced to elemental sulfur. Since some of those two-step methods of production of sulfur involve participation of H2O, it is fortunate that sulfates on the Martian surface are typically found as hydrated minerals. There is also a very important advantage in having hydrogen, carbon monoxide, or methane used as reducing agents in the sulfate reduction processes mentioned above, because all three of them are leading candidates for ISRU on Mars. These ISRU methods are well-known and successfully tested in the past or current space missions. In addition to using the aforementioned reducing agents, elemental sulfur may be obtained using the well-characterized Claus reaction, involving the reaction of H2S with SO2 produced by sulfate reduction.
The fourth category of processes investigated is decomposition of sulfates in the molten state using electrochemical methods. To obtain a eutectic salt mixture with a relatively low melting temperature, it is preferred that the melt consists of not just one individual sulfate but a combination of several sulfates and possibly other salts. There has been a report of possible formation of sulfur as a minor product of electrochemical reduction of hydrated sulfates. As in the case of chemical reduction, the main obstacle to the use of such methods is the greater likelihood of obtaining sulfides or sulfites as products than of obtaining elemental sulfur. However, as in the case of chemical reduction, it is possible to convert the sulfide or sulfite products of the electrochemical reaction as starting materials in a second process leading to formation of elemental sulfur.
Producing elemental sulfur from sulfides can be accomplished through two approaches: using sulfides that exist naturally as minerals and using sulfides that are produced during sulfate reduction. The first approach requires mapping of the Martian surface to identify sites that are rich in sulfides, which are less common than sulfates. This approach has the advantage of relying on well-established methods. The second approach involves reducing hydrated sulfates (found naturally on Mars) to sulfides and then converting the sulfides to sulfur. The second approach requires advanced techniques, but has the advantage of being able to start from sulfates that are abundantly available on Mars, without restricting the exploration site.
It should be emphasized that the reducing or oxidizing agents—CO, methane, hydrogen, and oxygen—used to obtain elemental sulfur are already regarded as likely products of ISRU processing on Mars. Extraction of sulfur on Mars to make sulfur concrete is even more attractive in view of the fact that the byproducts of sulfur production, such as water, hydrogen, oxygen, and metals, will be very useful for the planned missions. For instance, electrolysis of magnesium sulfate minerals has already been pointed out to be a potential source of magnesium metal on Mars. Finally, the study of potential methods of producing elemental sulfur on Mars may have useful future applications on Earth.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
