Abstract
Silver/manganese dioxide (Ag@MnO2) nanorods are synthesized and characterized by scanning electron microscopy, transmission electron microscopy, energy dispersive X-ray spectroscopy, X-ray powder diffraction, and X-ray photoelectron spectroscopy. It was discovered that Ag@MnO2 nanorods can realize hydrogen-borrowing reactions in high yields and are also effective for the synthesis of tert-butyl esters from aryl cyanides and tert-butyl hydroperoxide in a short period of time. Mechanistic experiments revealed that this catalytic system acts as a Lewis acid in hydrogen-borrowing reactions, while the synthesis of tert-butyl esters occurs through a radical pathway. This is the first report on the excellent catalytic activity of Ag@MnO2 nanorods as a catalyst.

Introduction
Hydrogen-borrowing methodology, also known as hydrogen auto-transfer, is a powerful strategy for reduction reactions that avoids using molecular hydrogen directly and the synthesis of complexes using other reactions.1–3 This methodology has been developed for many years as a useful tool in synthetic organic chemistry. 4 It also provides a sustainable and cost-effective approach for the synthesis of substituted amines and ketones, which are important groups in many bioactive molecules (Figure 1), without the need for toxic alkyl halides or borohydrides. 5 It has been demonstrated that transition-metal complexes are effective catalysts for hydrogen-borrowing reactions, as are noble and non-noble metals.6–15 Besides, biocatalysts, such as known enzymes and Aspergillus oryzae, are also used in hydrogen-borrowing reactions along with the development of enzymes. 16 Although some homogeneous noble metals are effective catalysts, there are catalyst-product separation and recycling issues involved in the use of these catalytic systems. Therefore, heterogeneous catalysts with recyclability have been developed such as supported catalysts. Our group has engaged in developing heterogeneous catalysts for dehydrogenation and hydrogen-borrowing reactions that are recyclable, such as polymer Zr-CIA, 17 POP-Ir, 18 Cu(binap)I]2, 19 TTA-Au-NG, 20 NQ-MR, 21 and Au@HT. 22 However, some catalytic systems are effective as catalysts, but demonstrate poor recyclability and stability, for example, catalysts with noble metal cores, such as iridium and gold, while others have reduced catalytic ability after several cycles. Inspired by this, we decided to study nanorods as heterogeneous catalysts in hydrogen-borrowing reactions and in the synthesis of tert-butyl esters, a functional group present in many natural products, polymers, and pharmaceuticals. 23
Examples of biologically active amines.
Nanoparticles, in the form of nanorods, are novel sustainable nano catalysts that solve many issues in modern synthesis and catalysis.24,25 Such nano catalytic systems have effective active sites and usually demonstrate much better catalytic performance and good recovery characteristics. 26 Manganese oxides (MnO2) nanorods are known as inexpensive and nontoxic materials, which can be used as environmental friendly catalysts.27–30 A recent survey of manganese oxide (MnO2) nanorods indicates that when incorporating noble metals, for example, Pt, Au, and Ag, the nano particles have improved activity. In addition, Ag nano particles have been shown to have outstanding ability as catalyst in many organic reactions.30–33 Based on our recent discoveries on hydrogen-borrowing reactions34–38 and the properties of manganese oxides (MnO2) nanorods and Ag nanoparticles, we herein report the synthesis and application of Silver/manganese dioxide (Ag@MnO2) nanorods as a catalyst, which has higher catalytic activity after several cycles and which is much cheaper than other catalysts. The Ag@MnO2 nanorods are characterized by several physical techniques. This synthetic catalytic system was successfully utilized in hydrogen-borrowing reactions and for the synthesis of tert-butyl esters.
Results and discussion
Characterization of the Ag@MnO2 nanorod
The supported Ag@MnO2 nanorods were synthesized by a hydrothermal method
39
and were characterized through scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy (EDX), X-ray powder diffraction (XRD), transmission electron microscopy (TEM), and X-ray photoelectron spectroscopy (XPS). The SEM and TEM images in Figure 2 reveal that silver nanoparticles are supported on α-MnO

((a) and (b)) SEM images of Ag@MnO2. ((c) and (d)) TEM images of Ag@MnO2.

EDS images of Ag@MnO2.
Figure 4 shows the X-ray diffraction patterns of the synthetic α-MnO2 and Ag@MnO2. By comparing to previous wok,40–42 the XRD pattern of Ag@MnO2 revealed the presence of α-MnO2 (JCPDFS 44-0141) and metallic silver (JCPDFS 87-0597). There is no Ag, AgOx, or other silver oxide species’ diffraction signal, which may reveal good dispersion of silver particles on the surface of α-MnO2. From the two patterns delineated in Figure 4, the diffraction peaks at 38.12°, 44.28°, 64.43°, and 81.54° were assigned to the (111), (200), (220), and (222) crystal faces of silver indicating that silver nanoparticles had been successfully supported on the α-MnO2 nanorods.

XRD pattern of Ag@ MnO2.
The chemical composition and surface chemical states of the Ag@MnO2 composite were also examined by XPS. Four elements (C, Ag, Mn, and O) were identified from the full-survey-scan spectrum of the Ag@MnO2 composite (Figure 5(a)). The Ag 3d spectrum shows two obvious peaks located at 368.1 eV for Ag 3d3/2 and 374.2 eV for Ag 3d5/2, with a spin energy separation of 6.1 eV, which explains that silver is present in the metallic state.
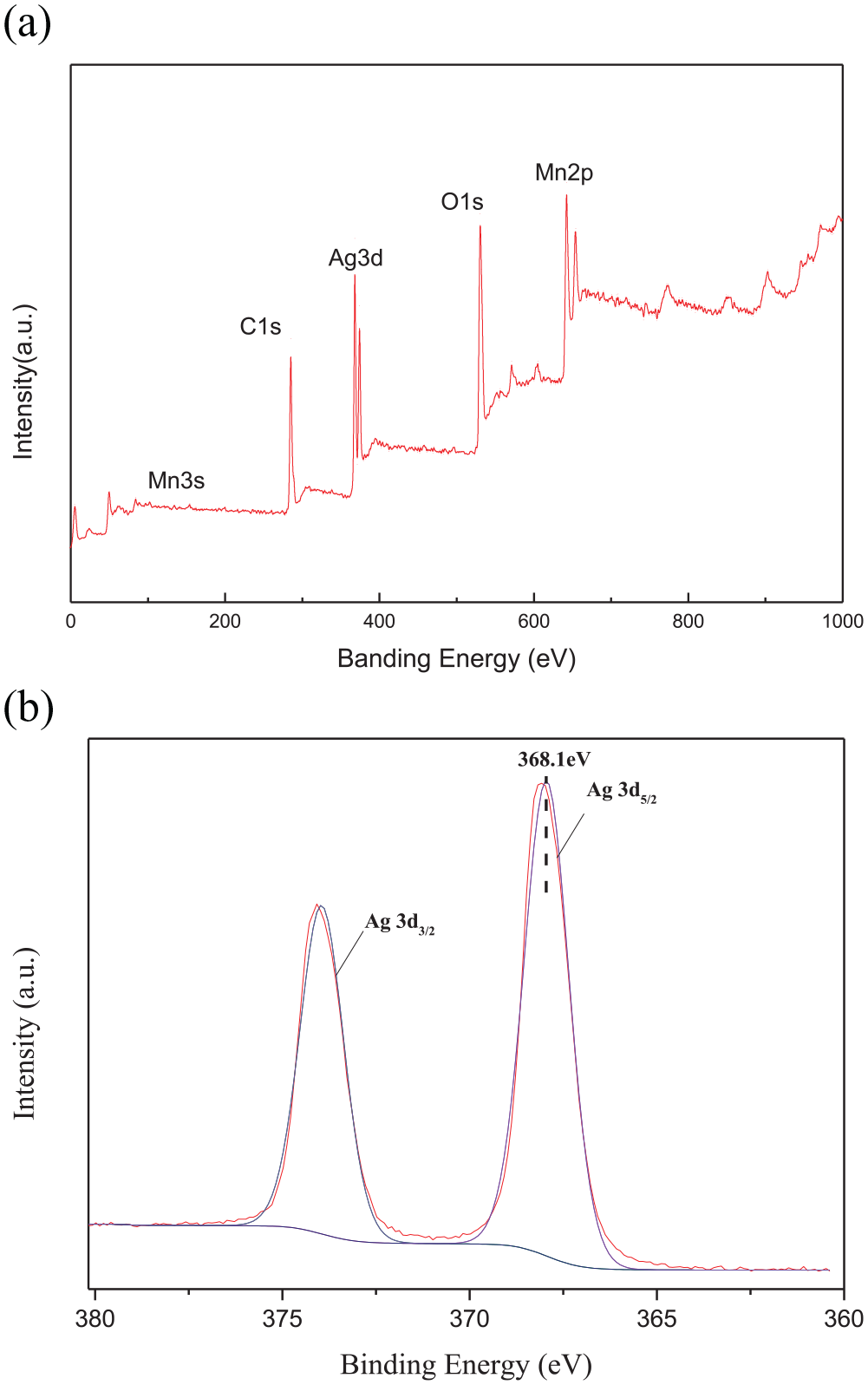
(a) XPS wide-scan spectrum of Ag@MnO2 and (b) high-resolution spectrum of Ag.
Catalytic activity
In order to test the catalytic activity of the Ag@MnO2 nanorods, the classical hydrogen-borrowing reaction between benzyl alcohol (
DCM: dichloromethane; DMF: dimethylformamide; DCE: dichloroethane.
Conditions:
Isolated yields.
Having established the optimum reaction conditions, the substrate scope of this hydrogen-borrowing reaction was explored. As shown in Table 2, all of the results are similar to the previous work,
43
different amines and alcohols could be employed, and it was found that this transformation was not influenced to any great extent by donating or withdrawing groups on the aromatic ring of the amine substrates such as methoxy (–OCH3), hydroxyl (–OH), chloro (–Cl), bromo (–Br), and fluoro (–F) (Table 2,
Conditions:
Isolated yields.
Considering the importance and application of tert-butyl esters in organic synthesis,
23
the reaction of aryl cyanides with tert-butyl hydroperoxide (TBHP) using the Ag@MnO2 catalytic system was explored. A range of phenylacetonitriles and TBHPs containing electron-rich or electron-deficient groups were employed. The desired products were obtained through C–CN bond cleavage and esterification in good to excellent yields and the results are summarized in Table 3. Phenylacetonitrile derivatives bearing electron-rich groups, like methoxy, methyl, and hydroxyl, reacted smoothly and were transferred into the corresponding esters
Conditions:
Isolated yields.
Control experiments and activity explorations
To gain further insight into the mechanism, control experiments were performed (Scheme 1). Therefore, 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO) was used in the Ag@MnO2 nanorod catalyzed reactions as a radical scavenger, and the experiments revealed that nearly the same yields of the desired products were obtained. However, totally different results were observed for the reaction of arylcyanide

Control experiments.
Based on these experiments and the work of other groups,33–35 we propose a tentative mechanism for the hydrogen-borrowing reaction (Scheme 2). First, the alcohol is oxidized to an aldehyde via dehydrogenation by the Ag@MnO2. At the same time, a silver-hydride complex is formed by hydride coordination. Next, the aldehyde condenses with the amine to form imine. Finally, the imine intermediate is reduced to give the target product.

The proposed reaction pathway.
As for the reaction of the arylcyanide with TBHP, we inferred from the control experiment that it proceeded via a radical pathway. Although the exact mechanism is still unclear, based on the literature33,34 and the control experiments, we have proposed a tentative mechanism (Scheme 3). According to the previous work,
35
it is possible to generate tert-butoxyl and tert-butylperoxy radicals in this system. Intermediate radical

A possible reaction mechanism for the synthesis of the tert-butyl esters.
Recyclability of the catalyst
Recycling the Ag@MnO2 nanorods was explored by repeating the hydrogen-borrowing reaction of benzyl alcohol (

Recyclability of the Ag@MnO2 nanorods in hydrogen-borrowing reactions.
To help explain these observations involving the Ag@MnO2 nanorod catalyst, SEM studies on recycled nanorod catalyst showed that the recovered catalyst had excellent stability (Figure 7).

SEM images: (a) fresh Ag@MnO2 nanorods catalyst and (b) Ag@MnO2 nanorod catalyst after five runs.
Conclusion
In conclusion, we have reported the synthesis and application of Ag@MnO2 nanorods, which have been characterized through SEM, TEM, EDX, XRD, and XPS. This synthetic catalytic system was successfully applied in hydrogen-borrowing reactions and for the synthesis of tert-butyl esters in moderate-to-high yields. Further investigation on the application of this catalyst in other reactions systems is in progress.
Experimental
Synthesis of Ag@MnO2
The α-MnO2 nanorods were synthesized by hydrothermal method according to the reported procedure. 39 Next, 400 mg of α-MnO2 nanowires and 130 mg of PVP (polyvinyl pyrrolidone) were added to 100 mL of EG (ethylene glycol). The obtained suspension was transferred to a 250 mL round-bottom flask and kept under vigorous stirring at 90 °C for 20 min. Then, NaBH4 (1.0 mmol) and 2 mL of 0.1 M AgNO3 (aq., 5%) solutions were sequential added to the reaction flask. This mixture was kept under vigorous stirring for another 12 h to produce Ag@MnO2 (Ag: 5%w/w), which were washed three times with ethanol (15 mL) and water (15 mL) by successive rounds of centrifugation at 7000 r/min for 8 min and removal of the supernatant. Finally, the product was dried under vacuum condition.
General procedure for synthesis of 3a
To 20-mL colorimetric tube was added
General procedure for synthesis of 6a
To 20 mL colorimetric tube was added
Supplemental Material
sj-pdf-1-chl-10.1177_1747519821989963 – Supplemental material for Silver/manganese dioxide nanorod catalyzed hydrogen-borrowing reactions and tert-butyl ester synthesis
Supplemental material, sj-pdf-1-chl-10.1177_1747519821989963 for Silver/manganese dioxide nanorod catalyzed hydrogen-borrowing reactions and tert-butyl ester synthesis by Huanhuan Luo, Yike Yang, Bobin Yang, Zhaojun Xu and Dawei Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: The authors gratefully acknowledge the financial support of this work by the National Natural Science Foundation of China (Nos. 21776111 and 21861039).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.








