Abstract
(E)-4-[2-(Pyridin-4-yl)vinyl]benzaldehyde, containing both a 4-vinylpyridine and an aldehyde functionality, is utilized to develop new, highly conjugated chalcone compounds and a bis-Schiff base azine compound. The chalcone-containing compounds are further explored for their protonation, methylation and silver(I) coordination chemistry using the pyridine moiety. In parallel, a cyano-containing analogue, (E)-4-[2-(pyridin-4-yl)vinyl]benzonitrile is also synthesized and studied for its silver(I) coordination chemistry. These new compounds are fully characterized by mass spectrometry, elemental analysis and spectroscopic techniques. The methylated product of (E)-1-(9-anthryl)-3-{4-[2-(pyridin-4-yl)vinyl]phenyl}prop-2-en-1-one and a silver complex of (E)-4-[2-(pyridin-4-yl)vinyl]benzonitrile are structurally determined by X-ray crystallography.

Introduction
4-Vinylpyridine (VP) has long been known as an essential building block in polymer chemistry. In 1984, Frechet and de Meftahi 1 published a brief review article entitled ‘Poly(vinylpyridine)s: Simple Reactive Polymers with Multiple Applications’. Since then, a significant increase in research efforts has been dedicated to the structural modification and materials applications related to poly(4-vinylpyrdine)s (PVP). 2 While being structurally similar to the polymer, polystyrene (PS), PVP is particularly attractive with regard to its unique functionality because of the presence of nitrogen atoms within the aromatic pendants. Pyridine is one of the important structural units in metal coordination chemistry and is a hydrogen-bonding acceptor in supramolecular chemistry due to its strong electron-donating ability; it also provides scope for rich chemical modifications on the nitrogen atom. Thus, the incorporation of pyridyl units in polymer materials will certainly bring versatile physical and chemical properties along with applications in optical, electronic, magnetic, catalytic, conductive and chemosensing materials.2,3 Remarkable recent progress in PVP chemistry includes applications in solar cells,4,5 organic–inorganic hybrid functional materials,6-10 nanomaterials,11,12 catalysis/electrocatalysis,13-15 heavy metal absorption/removal16-18 and environmental sensors.19-21
In addition to its main applications in polymer materials, 4-VP is also an important synthon in organic synthetic chemistry as well as in small molecular materials.22-24 Assuming that studies on the synthesis and modification of small molecules derived from 4-VP could be helpful for understanding the properties and behaviour in the polymeric state, we were interested in the synthesis of novel organic compounds derived from 4-VP and further studying the derivatization and coordination chemistry utilizing the pyridine functionality. In particular, the incorporation of an additional important chromophores, such as chalcone and anthracene, into 4-VP would extend the conjugation and may result in interesting photonic and electronic properties. Herein, we report the synthesis and derivatization of an aldehyde-containing 4-VP with chalcone, anthracence and imine functionalities and further modification through protonation, methylation and silver coordination. A novel silver coordination complex of cyano-derived 4-VP was also synthesized and structurally characterized (Scheme 1).

The structures of 4-vinylpyridine derivatives
Results and discussion
Synthesis of 4-PV derivatives 3 -5
The benzaldehyde-containing 4-PV (
In addition, we studied the reaction of
Protonation, methylation and metalation of compounds 3 and 4
Next, we explored the modification of new chalcone-containing 4-VP
While we were unable to isolate pure product from the protonation of compound
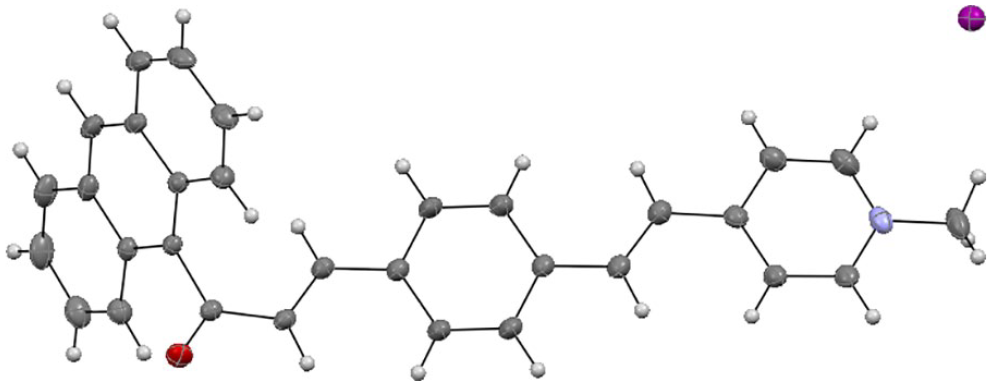
The ORTEP representation of the X-ray structure of (
Synthesis and crystal structure of Ag(2 )2(ClO4)
The silver coordination chemistry of the 4-PV ligand was further explored using the related compound

(a) The ORTEP structure of cationic [Ag(
Conclusion
In summary, we report the derivatization of an aldehyde-containing 4-VP with chalcone and imine functionalities and have further investigated the reactions of chalcone-based 4-PVs in protonation, methylation and silver coordination chemistry. The new compounds were synthesized in good yields and fully characterized by IR, MS, elemental analysis and 2D NMR spectroscopy. The methylated product was also structurally determined by single-crystal X-ray diffraction. In addition, an analogue of a silver complex with a related cyano-based 4-PV ligand was synthesized and structurally characterized by X-ray crystallography, revealing the coordination mode of this complex as a mononuclear bis-ligand complex. The newly synthesized derivatives of 4-VP are anticipated to be utilized as potential candidates for photocyclodimerization and polymerization for preparing new PVP materials. The additional chalcone moiety included in the 4-VP structures may give rise to opportunities for regioselective olefin polymerization.
Experimental
Solvents and reagents were purchased from Fisher Scientific or Sigma-Aldrich. All reactions were performed under ambient conditions in air. Fourier transform infrared (FTIR) spectra were measured on a Shimadzu 8400S instrument with solid samples using a Golden Gate ATR accessory. 1H and 13C NMR and 2D NMR spectra were obtained at room temperature on a Bruker III 500 MHz spectrometer. Electrospray ionization mass spectra were recorded using a Finnigan MAT LCQ mass spectrometer. UV-Vis electronic absorption spectra were recorded on a Varian-Cary 5000 spectrophotometer. (E)-4-[2-(Pyridin-4-yl)vinyl]benzaldehyde (
Synthesis of (E)-1-phenyl-3-(4-(2-(pyridin-4-yl)vinyl)phenyl)prop-2-en-1-one (3 )
Acetophenone (0.600 g, 5.00 mmol) and (E)-4-[2-(pyridin-4-yl)vinyl]benzaldehyde (
Synthesis of (E)-1-(9-anthryl)-3-(4-(2-(pyridin-4-yl)vinyl)phenyl)prop-2-en-1-one (4 )
9-Acetylanthracene (1.10 g, 5.00 mmol) and
Synthesis of 4-[2-(pyridin-4-yl)vinyl]benzaldazine (5 )
To a stirred solution of
Synthesis of (3 -H)+Cl−
To a stirred solution of the ligand
Synthesis of Ag(3 )2(NO3)
Ligand
Synthesis of Ag(3 )2(CF3CO2)
Ligand
Synthesis of (4 -Me)+I−
Ligand
Synthesis of Ag(4 )2(NO3)
Ligand
Synthesis of Ag(2 )2(ClO4)
Ligand
X-ray structural determinations
Suitable crystals of compounds (
Structural Refinement Data for (
Supplemental Material
sj-cif-1-chl-10.1177_1747519821989659 – Supplemental material for 4-vinylpyridine derivatives: Protonation, methylation and silver(I) coordination chemistry
Supplemental material, sj-cif-1-chl-10.1177_1747519821989659 for 4-vinylpyridine derivatives: Protonation, methylation and silver(I) coordination chemistry by Guoqi Zhang in Journal of Chemical Research
Supplemental Material
sj-cif-2-chl-10.1177_1747519821989659 – Supplemental material for 4-vinylpyridine derivatives: Protonation, methylation and silver(I) coordination chemistry
Supplemental material, sj-cif-2-chl-10.1177_1747519821989659 for 4-vinylpyridine derivatives: Protonation, methylation and silver(I) coordination chemistry by Guoqi Zhang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors gratefully acknowledge the support from the City University of New York (CUNY) for the PSC-CUNY awards (Nr. 62154-0050 and 63809-0051). Funding for this work is also provided by a Seed grant from the Office for the Advancement of Research and the PRISM programme at John Jay College, CUNY.
Supplemental material
CCDC 1980771 and 1980772 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html, or from the Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge CB2 1EZ, UK; fax: (+44) 1223-336-033; or e-mail: ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
