Abstract
An efficient palladium-catalyzed Hiyama reaction between various pyrimidin-2-yl tosylates with organosilanes has been developed. The use of CuCl with TBAF as additive is essential for promotion of the construction of the carbon–carbon bond. This procedure shows wide functional group tolerance for electrophilic pyrimidin-2-yl tosylates and has been extended to aromatic amino-substituted pyrimidin-2-yl tosylates, affording the desired C2-aryl and alkenyl pyrimidine derivatives in good to excellent yields.
This method proved to be tolerant of various electrophilic pyrimidin-2-yl tosylates as well as nucleophilic trimethoxy(phenyl) silane and vinyltrimethoxysilane.

Introduction
Transition-metal-catalyzed Hiyama coupling of organosilanes with halides has been developed as a routine tool for construction of new C–C bonds including the synthesis of the non-symmetrical biaryls,1,2 which are important materials for the preparation of polymers, agrochemicals, pharmaceutical intermediates, bioactive molecules, and electronic materials.3–6 Compared to other organometallic reagents,7–10 organosilicon reagents have emerged as more competitive nucleophilic partners for cross-coupling reactions due to their high chemical stability, low toxicity, and good compatibility.11–13 Among the electrophiles employed in Hiyama coupling reactions,14–18 halides exhibit relatively high reactivity, but they suffer from some drawbacks including harsh conditions for their preparation, with the formation of by-products and environmental pollution. 19 Thus, there is still demand for new electrophilic partners instead of halides for Hiyama reactions.
Enol- and phenol-based tosylates have garnered a great deal of interest in recent years because of their easy preparation from phenols, the good cleavage ability of C–O bonds and the consequent reduction of halide waste.20–22 Indeed tosylates have been effectively employed as new electrophilc partners for classical cross-coupling reactions.23–25 Nevertheless, only a few examples of Hiyama reactions of tosylates with silanes have been reported due to the weak polarization of the carbon–silicon bond in organosilicon reagents.26,27 In 2008, Wu and Zhang reported the Pd(OAc)2 catalyzed Hiyama coupling of aryl tosylates with silanes for the first time; vinyl tosylates also reacted well under the same conditions. 28 Subsequently, in 2010, Sarkar and co-workers efficiently achieved the Hiyama reaction of benzyl tosylates and aryltrialkoxysilane in PEG-600 with nanoparticles as the catalyst. 29 In 2013, Hiyama and co-workers described Ni(PPh3)2Cl2 catalyzed Hiyama reactions of aryl tosylates with aryl (trialkyl)silanes (Scheme 1). 30 Despite the great successes of aryl and benzylic tosylates in these reactions, the Hiyama coupling of heterocyclic tosylates, especially pyrimidin-2-yl tosylates is scarcely reported in the literature and remains a challenging task.

Hiyama reaction of tosylates with organosilanes.
Results and discussion
Pyrimidin-2-yl tosylates were prepared according to the literature.
31
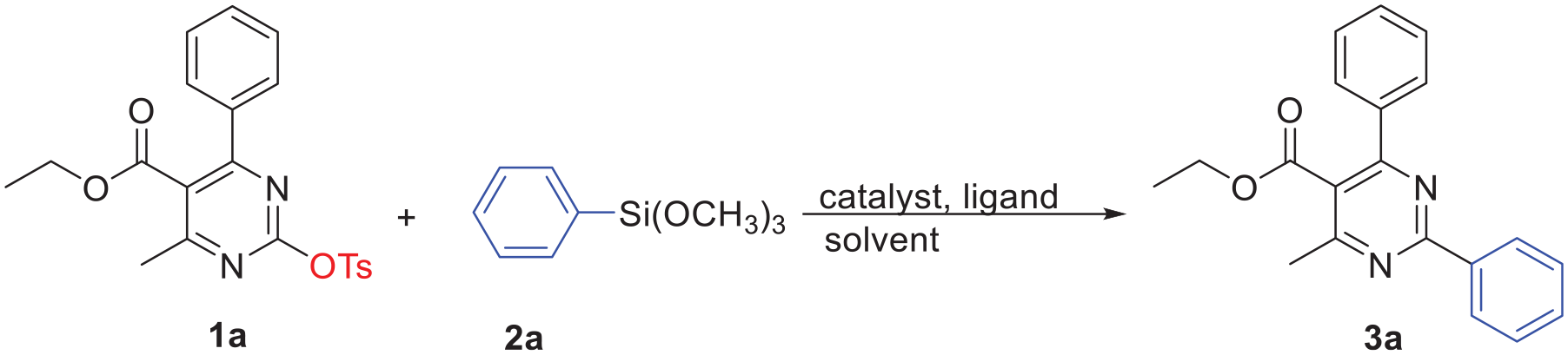
A set of trial experiments were carried out using pyrimidin-2-yl tosylate
Optimization of the Hiyama reaction of pyrimidin-2-yl tosylate

Reaction conditions: pyrimidin-2-yl tosylate
Isolated yield after column chromatography.
Subsequently, the necessity of PdCl2 and CuCl to promote the reaction was also tested, the result showed that no product was detected in the absence of PdCl2 (Table 1, entry 23) and the reaction conversion was also very low without CuCl (Table 1, entry 24), both PdCl2 and CuCl were indispensable for the Hiyama reaction of pyrimidin-2-yl tosylates with trimethoxy(phenyl)silane.
Under the optimized conditions, the scope of this reaction was investigated using a range of pyrimidin-2-yl tosylates as the electrophile, and the results are summarized in Table 2. For most cases, the pyrimidin-2-yl tosylates
The Hiyama reaction of pyrimidin-2-yl tosylates with phenyl and vinyl trimethoxysilanes.

The Hiyama reaction was not restricted to the use of trimethoxy(phenyl)silane as nucleophile, and allowed for the arylation of other substituted organosilanes. Notably, the desired products
To expand further the scope of this novel methodology, aromatic amino-substituted pyrimidin-2-yl tosylates were also tested as electrophilic partners. As illustrated in Table 3, the reaction of the aminopyrimidin-2-yl tosylate
The Hiyama reaction of aromatic amino pyrimidin-2-yl and pyridin-2-yl tosylates with phenyltrimethoxysilane.

Conclusion
In summary, we have developed an efficient protocol for the C2-arylation of pyrimidine derivatives via Pd-catalyzed Hiyama cross-coupling reactions of heteroaromatic tosylates with organosilanes in dioxane at 110 °C. The catalyst system including PdCl2 and PCy3 exhibits good tolerance for various substituted pyrimidin-2-yl tosylates bearing both electron-donating groups or electron-withdrawing ones. Meanwhile, the nucleophile partner is not restricted to the use of trimethoxy(phenyl)silane, vinyltrimethoxysilane can also give the desired products in moderate yields. In addition, CuCl and TBAF·3H2O are crucial for activating the C–Si bond in organosilanes and promoting the Hiyama reaction.
Experimental
Infrared (IR) spectra were recorded on a Nicolet Avatar 36 Fourier transform spectrophotometer in KBr disk. 1H NMR and 13C NMR data analyses were performed on a Varian Mercury plus-400 or plus-600 instrument using tetramethylsilane (TMS) as the internal standard, CDCl3 as solvent. The 1H NMR data are reported as follows: chemical shift, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, and m = multiplet), coupling constant (J-values) in Hz, and integration. The chemical shifts for 1H NMR and 13C NMR spectra were recorded in units (ppm). High-resolution mass spectra (HRMS) (electrospray ionization (ESI)) were obtained with a Bruker Daltonics APEX II 47e and an Orbitrap Elite mass spectrometer. Column chromatography was generally performed on silica gel (200–300 mesh) and analytical thin-layer chromatography (TLC) was conducted on silica gel GF254 plates according to standard techniques. Melting points were measured with an XT-4 apparatus. Solvents were dried by standard procedures. Pyrimidin-2-yl tosylates as starting materials were prepared by previously reported procedures of our group, 31 and other chemical reagents were purchased and used without further purification.
General procedure for synthesis 3a–r and 5a–c
The pyrimidin-2-yl tosylate (0.2 mmol), PdCl2 (5 mol%, 0.01 mmol), PCy3 (6 mol%, 0.012 mmol), CuCl (20 mol%, 0.04 mmol), and TBAF·3H2O (2.0 equiv., 0.4 mmol) were respectively added to the sealed Schlenk tube. The organosilane (2.0 equiv., 0.4 mmol) and toluene (2 mL) were injected into the sealed tube by syringe under a nitrogen atmosphere and the mixture was stirred at 110 °C for 36 h. After completion of the reaction as monitored by thin-layer chromatography (silica gel), the mixture was cooled to ambient temperature, diluted with saturated aqueous NH4Cl (5 mL), and extracted with ethyl acetate (3 × 10 mL). The combined organic layers were dried with anhydrous MgSO4 and concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography (ethyl acetate:petroleum ether = 1:50) to afford products
Ethyl 4-methyl-2,6-diphenylpyrimidine-5-carboxylate (3a ):
White solid 36 ; 80% yield; m.p. 66–86 °C (66–67 C); IR (KBr) v (cm−1): 3059, 2978, 2925, 1724, 1535, 1495, 1430, 1259, 1174, 1078; 1H NMR (600 MHz, CDCl3): δ 8.56-8.55 (m, 2H), 7.76-7.75 (m, 2H), 7.50-7.47 (m, 6H), 4.21 (q, J= 7.2 Hz, 2H), 2.70 (s, 3H), 1.08 (t, J= 7.2 Hz, 3H); 13C NMR (150 MHz, CDCl3): δ 168.5, 165.5, 163.7, 163.6, 138.3, 137.2, 131.1, 130.0, 128.7, 128.6, 128.5, 128.5, 123.4, 61.8, 22.9, 13.7; HRMS (ESI+) m/z: calcd for C20H19N2O2 [M + H]+: 319.1441; found: 319.1447.
Ethyl 4-methyl-2-phenyl-6-(p-tolyl)pyrimidine-5-carboxylate (3b ):
White solid 36 ; 83% yield; m.p. 67–69 °C (66–67 °C); IR (KBr) v (cm−1): 2920, 2848, 1724, 1658, 1537, 1511, 1452, 1375, 1259, 1174, 1076; 1H NMR (600 MHz, CDCl3): δ 8.56-8.54 (m, 2H), 7.68 (d, J= 8.2 Hz, 2H), 7.49-7.47 (m, 3H), 7.27 (d, J= 7.2 Hz, 2H), 4.24 (q, J= 7.2 Hz, 2H), 2.68 (s, 3H), 2.41 (s, 3H), 1.13 (t, J= 7.2 Hz, 3H); 13C NMR (150 MHz, CDCl3): δ 168.7, 165.2, 163.7, 163.4, 140.3, 137.3, 135.4, 131.0, 129.3, 128.7, 128.5, 128.5, 123.2, 61.8, 22.9, 21.5, 13.8; HRMS (ESI+) m/z: calcd for C21H21N2O2 [M + H]+: 333.1598; found: 333.1594.
Ethyl 4-(4-methoxyphenyl)-6-methyl-2-phenylpyrimidine-5-carboxylate (3c ):
White solid 37 ; 87% yield; m.p. 62–36 °C (64–65 °C); IR (KBr) v (cm−1): 3062, 2924, 2839, 1724, 1537, 1464, 1365, 1259, 1174, 1076; 1H NMR (400 MHz, CDCl3): δ 8.46-8.44 (m, 2H), 7.67 (d, J= 8.8 Hz, 2H), 7.38-7.37 (m, 3H), 6.88 (d, J= 8.8 Hz, 2H), 4.15 (q, J= 7.2 Hz, 2H), 3.72 (s, 3H), 2.56 (s, 3H), 1.05 (t, J= 7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 168.7, 165.0, 163.4, 162.5, 161.2, 137.1, 130.8, 130.3, 130.0, 128.4, 128.4, 122.7, 113.8, 61.7, 55.2, 22.7, 13.7; HRMS (ESI+) m/z: calcd for C21H21N2O3 [M + H]+: 349.1547; found: 349.1542.
Ethyl 4-(4-fluorophenyl)-6-methyl-2-phenyl pyrimidine-5-carboxylate (3d ):
White solid 36 ; 81% yield; m.p. 84–86 °C (85–86 °C); IR (KBr) v (cm−1): 3060, 3055, 2922, 1724, 1600, 1537, 1506, 1427, 1363, 1259, 1159, 1076; 1H NMR (600 MHz, CDCl3): δ 8.55-8.53 (m, 2H), 7.78-7.76 (m, 2H), 7.51-7.49 (m, 3H), 7.17 (t, J= 8.4 Hz, 2H), 4.24 (q, J= 7.2 Hz, 2H), 2.69 (s, 3H), 1.14 (t, J= 7.2 Hz, 3H); 13C NMR (150 MHz, CDCl3): δ 168.4, 165.5, 164.0 (d, J= 248.9 Hz, 1C), 163.7, 163.1, 162.3, 137.0, 134.3 (2C), 131.1, 130.6 (d, J= 8.7 Hz, 1C), 128.6 (d, J= 7.2 Hz, 1C), 123.2, 115.6 (d, J= 21.8 Hz, 1C), 61.9, 22.9, 13.8; 19F NMR (564 MHz, CDCl3): δ −110.6; HRMS (ESI+) m/z: calcd for C20H18FN2O2 [M + H]+: 337.1347; found: 337.1352.
Ethyl 4-(4-chlorophenyl)-6-methyl-2-phenyl pyrimidine-5-carboxylate (3e ):
White solid 36 ; 76% yield; m.p. 83–85 °C (83–84 °C); IR (KBr) v (cm−1): 3051, 3014, 2928, 1724, 1536, 1363, 1255, 1174, 1078; 1H NMR (400 MHz, CDCl3): δ 8.48-8.45 (m, 2H), 7.63 (d, J= 8.4 Hz, 2H), 7.43-7.37 (m, 5H), 4.16 (q, J= 7.2 Hz, 2H), 2.62 (s, 3H), 1.07 (t, J= 7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 168.2, 165.6, 163.7, 162.3, 136.8, 136.6, 136.4, 131.2, 129.9, 128.7, 128.6, 128.5, 123.2, 61.9, 22.8, 13.7; HRMS (ESI+) m/z: calcd for C20H18ClN2O2 [M + H]+: 353.1051; found: 353.1047.
Ethyl 4-(2-chlorophenyl)-6-methyl-2-phenyl pyrimidine-5-carboxylate (3f ):
White solid 38 ; 72% yield; m.p. 101–310 °C (101–102 °C); IR (KBr) v (cm−1): 3053, 2979, 2922, 1724, 1595, 1543, 1432, 1360, 1252, 1178, 1082; 1H NMR (600 MHz, CDCl3): δ 8.52-8.51 (m, 2H), 7.51-7.47 (m, 4H), 7.42-7.36 (m, 3H), 4.10 (q, J= 7.2 Hz, 2H), 2.79 (s, 3H), 0.96 (t, J= 7.2 Hz, 3H); 13C NMR (150 MHz, CDCl3): δ 166.7, 166.6, 164.0, 163.8, 138.0, 137.0, 132.3, 131.3, 130.2, 130.2, 129.6, 128.9, 128.6, 126.7, 123.9, 61.5, 23.7, 13.5; HRMS (ESI+) m/z: calcd for C20H18ClN2O2 [M + H]+: 353.1051; found: 353.1049.
Ethyl 4-(4-bromophenyl)-6-methyl-2-phenyl pyrimidine-5-carboxylate (3g ):
White solid 36 ; 69% yield; m.p. 88–90 °C (87–89 °C); IR (KBr) v (cm−1): 3059, 2984, 2896, 1724, 1587, 1539, 1370, 1259, 1174, 1084; 1H NMR (400 MHz, CDCl3): δ 8.51-8.49 (m, 2H), 7.78 (d, J= 8.4 Hz, 2H), 7.44-7.38 (m, 5H), 4.19 (q, J= 7.2 Hz, 2H), 2.63 (s, 3H), 1.06 (t, J= 7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 168.5, 165.4, 163.7, 163.1, 137.1, 137.0, 131.0, 128.9, 128.8, 128.6, 128.5, 127.8, 127.2, 127.2, 123.2, 61.8, 22.9, 13.7; HRMS (ESI+) m/z: calcd for C20H18BrN2O2 [M + H]+: 397.0546; found: 397.0541.
Ethyl 4-methyl-6-(3-nitrophenyl)-2-phenyl pyrimidine-5-carboxylate (3h ):
White solid 38 ; 81% yield; m.p. 120–112 °C (123–125 °C); IR (KBr) v (cm−1): 3052, 2983, 2922, 1718, 1581, 1535, 1352, 1261, 1178, 1074; 1H NMR (600 MHz, CDCl3): δ 8.66 (s, 1H), 8.55 (s, 2H), 8.37 (s, 1H), 8.11 (s, 1H), 7.68 (s, 1H), 7.53 (s, 3H), 4.30 (q, J= 7.2 Hz, 2H), 2.73 (s, 3H), 1.19 (t, J= 7.2 Hz, 3H); 13C NMR (150 MHz, CDCl3): δ 167.8, 166.2, 164.1, 161.0, 148.3, 139.8, 136.6, 134.5, 131.5, 129.6, 128.7, 128.7, 124.7, 123.7, 123.4, 62.2, 23.1, 13.8; HRMS (ESI+) m/z: calcd for C20H18N3O4 [M + H]+: 364.1292; found: 364.1296.
Methyl 4-methyl-2,6-diphenylpyrimidine-5-carboxylate (3i ):
White solid 38 ; 89% yield; m.p. 87–89 °C (87–89 °C); IR (KBr) v (cm−1): 3059, 2949, 1726, 1603, 1535, 1495, 1257, 1174, 1079; 1H NMR (600 MHz, CDCl3): δ 8.56 (s, 2H), 7.76 (s, 2H), 7.50-7.48 (m, 6H), 3.74 (s, 3H), 2.69 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 169.1, 165.5, 163.8, 163.4, 138.1, 137.1, 131.1, 130.1, 128.7, 128.6, 128.6, 128.4, 123.0, 52.6, 23.0; HRMS (ESI+) m/z: calcd for C19H17N2O2 [M + H]+: 305.1285; found: 305.1280.
Methyl 4-methyl-2-phenyl-6-(p-tolyl) pyrimidine-5-carboxylate (3j ):
White solid 38 ; 83% yield; m.p. 63–56 °C (63–65 °C); IR (KBr) v (cm−1): 3060, 3030, 2947, 2850, 1728, 1612, 1537, 1512, 1390, 1263, 1172, 1078; 1H NMR (600 MHz, CDCl3): δ 8.56-8.54 (m, 2H), 7.69 (t, J= 7.8 Hz, 2H), 7.51-7.49 (m, 3H), 7.29 (d, J= 8.4 Hz, 2H), 3.76 (s, 3H), 2.67 (s, 3H), 2.43 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 169.3, 165.4, 163.7, 163.3, 140.5, 137.2, 135.2, 131.1, 129.4, 128.7, 128.5, 128.4, 122.8, 52.6, 23.0, 21.5; HRMS (ESI+) m/z: calcd for C20H19N2O2 [M + H]+: 319.1441; found: 319.1447.
Methyl 4-(4-methoxyphenyl)-6-methyl-2-phenylpyrimidine-5-carboxylate (3k ):
White solid; 85% yield; m.p. 89–91 °C; IR (KBr) v (cm−1): 3057, 2997, 2945, 2830, 1714, 1606, 1539, 1508, 1253, 1174, 1076; 1H NMR (600 MHz, CDCl3): δ 8.56-8.54 (m, 2H), 7.77 (d, J= 9.0 Hz, 2H), 7.50-7.59 (m, 3H), 7.00 (d, J= 8.4 Hz, 2H), 3.88 (s, 3H), 3.78 (s, 3H), 2.66 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 169.4, 165.2, 163.6, 162.6, 161.3, 137.3, 131.0, 130.4, 130.0, 128.6, 128.5, 122.4, 114.0, 55.4, 52.6, 22.9; HRMS (ESI+) m/z: calcd for C20H19N2O3 [M + H]+: 335.1390; found: 335.1395.
Methyl 4-(4-fluorophenyl)-6-methyl-2-phenyl pyrimidine-5-carboxylate (3l ):
Yellow oil 38 ; 78% yield; IR (KBr) v (cm−1): 3060, 2924, 1726, 1604, 1539, 1508, 1390, 1263, 1157, 1076; 1H NMR (600 MHz, CDCl3): δ 8.55-8.53 (m, 2H), 7.78-7.56 (m, 2H), 7.50-7.49 (m, 3H), 7.17 (t, J= 8.4 Hz, 2H), 3.75 (s, 3H), 2.67 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 169.0, 165.6, 164.8, 164.0 (d, J= 249.3 Hz, 1C), 163.2, 162.2, 137.0, 134.2 (2C), 131.2, 130.4 (d, J= 8.6 Hz, 1C), 128.6 (d, J= 9.3 Hz, 1C), 122.8, 115.7 (d, J= 21.8 Hz, 1C), 52.6, 22.9; 19F NMR (564 MHz, CDCl3): δ −110.4; HRMS (ESI+) m/z: calcd for C19H16FN2O2 [M + H]+: 323.1190; found: 323.1194.
Methyl 4-(4-chlorophenyl)-6-methyl-2-phenyl pyrimidine-5-carboxylate (3m ):
Colorless oil; 61% yield; IR (KBr) v (cm−1): 3060, 2920, 2848, 1722, 1539, 1489, 1390, 1265, 1171, 1088; 1H NMR (600 MHz, CDCl3): δ 8.55-8.53 (m, 2H), 7.71 (d, J= 8.4 Hz, 2H), 7.51-7.50 (m, 3H), 7.47 (d, J= 8.4 Hz, 2H), 3.76 (s, 3H), 2.68 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 168.8, 165.7, 163.9, 162.2, 136.9, 136.5, 136.4, 131.20, 129.8, 128.8, 128.6, 128.5, 122.4, 52.7, 22.9; HRMS (ESI+) m/z: calcd for C19H16ClN2O2 [M + H]+: 339.0895; found: 339.0889.
Isopropyl 4-methyl-2,6-diphenylpyrimidine-5-carboxylate (3n ):
White solid 38 ; 88% yield; m.p. 81–28 °C (82–84 °C); IR (KBr) v (cm−1): 3060, 3055, 2976, 2920, 2848, 1720, 1583, 1543, 1267, 1174, 1099; 1H NMR (600 MHz, CDCl3): δ 8.56-8.54 (m, 2H), 7.77-7.75 (m, 2H), 7.49-7.46 (m, 6H), 5.12 (m, 1H), 2.69 (s, 3H), 1.11 (d, J= 6.0 Hz, 6H); 13C NMR (150 MHz, CDCl3): δ 168.0, 165.2, 163.7, 163.5, 138.3, 137.3, 131.0, 130.0, 128.7, 128.6, 128.5, 128.5, 123.9, 69.7, 22.8, 21.4; HRMS (ESI+) m/z: calcd for C21H21N2O2 [M + H]+: 333.1598; found: 333.1595.
Ethyl 4-isopropyl-2,6-diphenylpyrimidine-5-carboxylate (3o ):
Colorless oil 38 ; 80% yield; IR (KBr) v (cm−1): 3058, 2971, 2922, 2847, 1722, 1587, 1536, 1265, 1174, 1089; 1H NMR (600 MHz, CDCl3): δ 8.62-8.60 (m, 2H), 7.77-7.75 (m, 2H), 7.49-7.45 (m, 6H), 4.19 (q, J= 7.2 Hz, 2H), 3.30 (m, 1H), 1.42 (d, J= 6.6 Hz, 6H), 1.06 (t, J= 7.2 Hz, 3H); 13C NMR (150 MHz, CDCl3): δ 173.0, 168.7, 163.8, 163.6, 138.5, 137.6, 133.9, 133.7, 131.0, 129.9, 128.7, 128.5, 128.5, 122.82, 61.8, 33.4, 22.0, 13.6; HRMS (ESI+) m/z: calcd for C22H23N2O2 [M + H]+: 347.1754; found: 347.1761.
Methyl 4-methyl-6-phenyl-2-vinylpyrimidine-5-carboxylate (3p ):
Colorless oil; 64% yield; IR (KBr) v (cm−1): 3055, 2948, 1726, 1648, 1536, 1251, 1175, 1078; 1H NMR (400 MHz, CDCl3): δ 7.69-7.66 (m, 2H), 7.48-7.46 (m, 3H), 7.00-6.90 (m, 1H), 6.78-6.73 (m, 1H), 5.82-5.79 (m, 1H), 3.71 (s, 3H), 2.62 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 169.7, 165.9, 164.4, 161.9, 161.2, 133.8, 130.7, 130.0, 123.9, 121.5, 52.5, 21.0; HRMS (ESI+) m/z: calcd for C15H15N2O2 [M + H]+: 255.1128; found: 255.1126.
Methyl 4-(4-methoxyphenyl)-6-methyl-2-vinyl pyrimidine-5-carboxylate (3q ):
Colorless oil; 58% yield; IR (KBr) v (cm−1): 3059, 2941, 2866, 1723, 1647, 1533, 1246, 1176, 1077; 1H NMR (400 MHz, CDCl3): δ 7.60 (d, J= 9.2 Hz, 2H), 6.90 (d, J= 8.8 Hz, 2H), 6.84-6.80 (m, 1H), 6.68-6.63 (m, 1H), 5.72-5.69 (m, 1H), 3.79 (s, 3H), 3.68 (s, 3H), 2.51 (s, 3H); 13C NMR (100 MHz, CDCl3): δ 168.2, 163.9, 162.4, 161.6, 16 0.3, 135.3, 129.2, 128.9, 123.9, 121.7, 113.1, 54.4, 51.6, 21.7; HRMS (ESI+) m/z: calcd for C16H17N2O3 [M + H]+: 285.1234; found: 285.1230.
Ethyl 4-(4-methoxyphenyl)-6-methyl-2-vinyl pyrimidine-5-carboxylate (3r ):
Colorless oil; 61% yield; IR (KBr) v (cm−1): 3057, 2920, 2865, 1724, 1647, 1536, 1464, 1365, 1252, 1174, 1078; 1H NMR (400 MHz, CDCl3): δ 7.67 (d, J= 8.8 Hz, 2H), 6.97 (d, J= 8.8 Hz, 2H), 6.93-6.85 (m, 1H), 6.75-6.74 (m, 1H), 5.80-5.70 (m, 1H), 4.24 (q, J= 7.2 Hz, 2H), 3.86 (s, 3H), 2.60 (s, 3H), 1.15 (t, J= 7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 168.6, 164.8, 162.6, 159.2, 158.8, 136.3, 130.0, 129.3, 123.1, 121.5, 114.4, 61.7, 55.4, 22.7, 13.8; HRMS (ESI+) m/z: calcd for C17H19N2O3 [M + H]+: 299.1390; found: 299.1395.
6-Methyl-N,2-diphenylpyrimidin-4-amine (5a ):
White solid; 84% yield; m.p. 114–116 °C; IR (KBr) v (cm−1): 3377, 3055, 2918, 1606, 1537, 1496, 1446, 1367, 1328, 1192, 1084; 1H NMR (600 MHz, CDCl3): δ 8.10-88.0 (m, 2H), 7.76 (d, J= 8.4 Hz, 2H), 7.51-7.50 (m, 3H), 7.36 (d, J= 7.8 Hz, 3H), 7.06 (s, 1H), 7.04 (d, J= 7.8 Hz, 1H), 2.49 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 168.6, 164.7, 160.1, 140.0, 137.4, 130.5, 128.9, 128.8, 127.1, 122.1, 118.9, 108.0, 24.4; HRMS (ESI+) m/z: calcd for C17H16N3 [M + H]+: 262.1339; found: 262.1336.
6-Methyl-2-phenyl-N-(p-tolyl)pyrimidin-4-amine (5b ):
White solid; 86% yield; m.p. 79–81 °C; IR (KBr) v (cm−1): 3392, 2917, 2846, 1608, 1543, 1511, 1380, 1300, 1255, 1178, 1084, 1026; 1H NMR (600 MHz, CDCl3): δ 8.08-78.0 (m, 2H), 7.63 (d, J= 8.4 Hz, 2H), 7.50-7.49 (m, 3H), 7.23 (s, 1H), 7.17 (d, J= 8.4 Hz, 2H), 7.03 (s, 1H), 2.48 (s, 3H), 2.35 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 168.5, 164.7, 160.3, 137.5, 137.4, 131.6, 130.5, 129.3, 128.7, 127.1, 119.2, 107.7, 24.4, 20.8; HRMS (ESI+) m/z: calcd for C18H18N3 [M + H]+: 276.1495; found: 276.1491.
N-(4-chlorophenyl)-6-methyl-2-phenylpyrimidin-4-amine (5c ):
White solid; 87% yield; m.p. 142–144 °C; IR (KBr) v (cm−1): 3346, 3051, 2923, 1609, 1539, 1445, 1379, 1325, 1190, 1081; 1H NMR (600 MHz, CDCl3): δ 8.06-48.0 (m, 2H), 7.67 (d, J= 9.0 Hz, 2H), 7.50-7.49 (m, 3H), 7.38 (s, 1H), 7.29 (d, J= 9.0 Hz, 2H), 7.06 (s, 1H), 2.48 (s, 3H); 13C NMR (150 MHz, CDCl3): δ 168.6, 164.6, 159.9, 138.6, 137.2, 130.6, 128.8, 128.8, 127.1, 126.7, 120.1, 108.3, 24.4; HRMS (ESI+) m/z: calcd for C17H15ClN3 [M + H]+: 296.0949; found: 296.0952.
Supplemental Material
sj-doc-1-chl-10.1177_17475198211067163 – Supplemental material for Palladium-catalyzed Hiyama cross-couplings of pyrimidin-2-yl tosylates with organosilanes
Supplemental material, sj-doc-1-chl-10.1177_17475198211067163 for Palladium-catalyzed Hiyama cross-couplings of pyrimidin-2-yl tosylates with organosilanes by Hai-Peng Gong, Zheng-Jun Quan and Xi-Cun Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (no. 21362031 and 21562036) and special funds for discipline construction of Gansu Agricultural University (GAU-XKJS-2018-121).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.