Abstract
Isoxazoline derivatives of the natural products eugenol, 1’-S-acetoxychavicol acetate and sclareol are prepared through 1,3-dipolar cycloaddition reactions in an aqueous buffered system. The compounds are evaluated for their antitubercular and antibacterial activities. Compounds
Keywords

Introduction
Tuberculosis (TB) is a contagious respiratory disease caused by Mycobacterium tuberculosis. According to the World Health Organization (WHO), TB was ranked among the top 10 most common causes of death due to an infectious agent, with approximately 10 million people being infected and 1.4 million people dying of TB in 2019; although both TB incidence and TB mortality rates slightly decreased globally in 2020, they did not reach the 2020 milestone.1,2 Typically, TB is prevalent in tropical regions with dense populations, such as India, Indonesia, Nigeria, Bangladesh, the Philippines and Pakistan.1–3 In addition, concern has grown with the emergence of multidrug-resistant strains that remain a public threat, with approximately >200,000 reported cases of resistance in 2019.1,2,4–6 Therefore, identifying novel antitubercular agents is imperative.
Natural products, extracted and isolated from medicinal herbs, marine organisms, or microorganisms, display a variety of biological activities and are invaluable sources for drug discovery; examples of such products include artemisinin, penicillin, quinine, paclitaxel and amphotericin B.7–9 Diverse primary or secondary metabolites can be derived from natural sources, leading to the discovery of new bioactive ingredients that are useful in medicinal chemistry and pharmaceutical chemistry research.8,10 Chemical transformations involving the use of natural products as starting materials have been extensively applied in the fields of natural product research and medicinal chemistry.11,12 Such transformations entail the use of natural products as building blocks, and the resulting natural product derivatives exhibit enhanced biological activities, thus leading to the discovery of new drug candidates. In nature, various natural products contain terminal alkene moieties, with some being reported to have antitubercular activities. These moieties can act as dipolarophiles, reacting with a nitrile oxide to form an isoxazoline ring, for example, phenylpropenes

Structures of antitubercular isoxazolines and examples of natural products containing an olefin moiety: eugenol (
According to our review of the literature, is oxazolines are heterocyclic compounds that are considered to be among the most potent antimicrobial agents.23–27 However, studies on antitubercular agents containing an isoxazoline nucleus are scarce. In addition, the literature does not contain a molecular modelling study on isoxazoline derivatives as InhA inhibitors. Accordingly, in this study, natural compounds – namely, eugenol (
Results and discussion
Chemistry
The isoxazoline derivatives of natural products
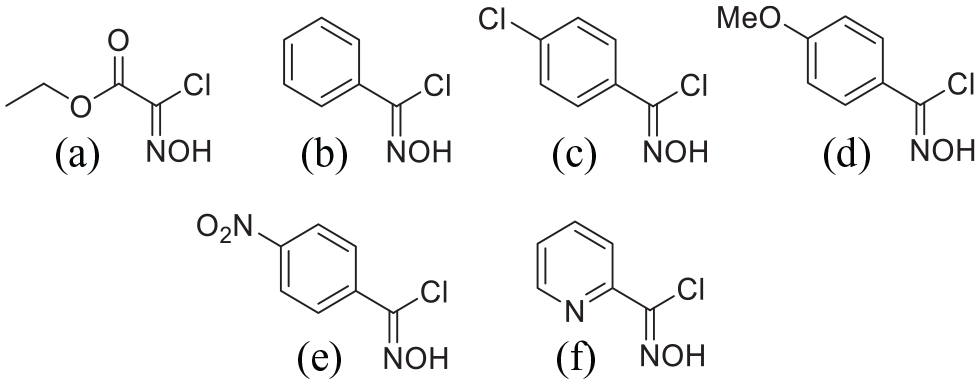
Structures of oxime chlorides
Synthesis of the isoxazoline derivatives.

KPi: K2HPO4/KH2PO4.
Starting material (1.0 equiv.).
Oxime chloride (1.2 equiv.).
Isolated yield.
d.r.: diastereoisomeric ratio (major/minor) based on 1H NMR analysis.
All reactions were performed in aqueous phosphate buffer.
For the eugenol series
The isoxazoline derivatives
Antitubercular and antibacterial activities of the isoxazoline derivatives.

Mtb: M. tuberculosis H37Ra strain: MIC (µM) of rifampin (0.01).
Bacillus cereus: MICs (µM) of chloramphenicol (12.38) and tetracycline∙HCl (4.16–8.32).
Staphylococcus aureus: MICs (µM) of chloramphenicol (1.55) and tetracycline∙HCl (0.26).
Pseudomonas aeruginosa: MICs (µM) of chloramphenicol (24.76) and tetracycline∙HCl (8.32).
Escherichia coli: MICs (µM) of chloramphenicol (6.19) and tetracycline∙HCl (2.08–4.16).
Inactive at >100 µg/mL for Mtb and >400 µg/mL for the four bacterial strains (maximum concentration).
Cytotoxic activity of the isoxazoline derivatives.

Inactive at 50 µg/mL (maximum concentration).
Relative inhibitory activity of the 1’-S-acetoxychavicol acetate isoxazoline series

Biological evaluation
The derivatives were evaluated for their antitubercular and antibacterial activities (Table 2). Derivatives
The MIC values of all the isoxazolines were determined using the broth microdilution method
31
against both Gram-positive bacteria (Bacillus cereus and Staphylococcus aureus) and Gram-negative bacteria (Escherichia coli and Pseudomonas aeruginosa) (Table 2). The natural product eugenol (
InhA inhibitory activity
Many antitubercular agents inhibit InhA as the biomolecular target.21,22 In particular, five-membered heterocyclic compounds exhibit potent antitubercular activity; however, studies investigating isoxazoline derivatives for their InhA inhibitory activities are scarce.
32
Accordingly, in this study, the compounds were tested for their InhA inhibitory activities using the drug triclosan as a positive control (Table 4). The relative inhibitory potency of 1 µM of triclosan against the InhA enzyme was 50.49%. The 1’-S-acetoxychavicol acetate series
Molecular docking of isoxazoline derivatives to the InhA enzyme
Molecular modelling was employed to investigate the binding action of the isoxazoline derivatives. The docking protocol was first validated to ensure its reliability and reproducibility. The co-crystallised ligand was removed from the crystal structure of InhA, its energy was minimised, and it was redocked to the InhA binding site (see section “Experimental”). The ChemPLP scoring function was used for docking. The results revealed that ChemPLP demonstrated good overlays of heavy atoms between the co-crystallised ligand and the best ranked docked conformation with a root-mean-square deviation (RMSD) value of 1.9 Å calculated using DockRMSD 33 (Figure S24, Supporting Information). The RMSD value of <2.0 Å is considered acceptable and discriminates well-docked from poorly docked ligands with respect to crystallographic data. 34
Table S1 (Supporting Information) lists the calculated molecular docking scores, which were similar to those of triclosan. The molecular docking results provided insights into the possible binding interactions and binding modes of the isoxazoline derivatives. Compounds

(a) The predicted binding interactions of 1’-S-acetoxychavicol acetate (
Because the isoxazoline derivatives
Physicochemical properties of the 1’-S-acetoxychavicol acetate (
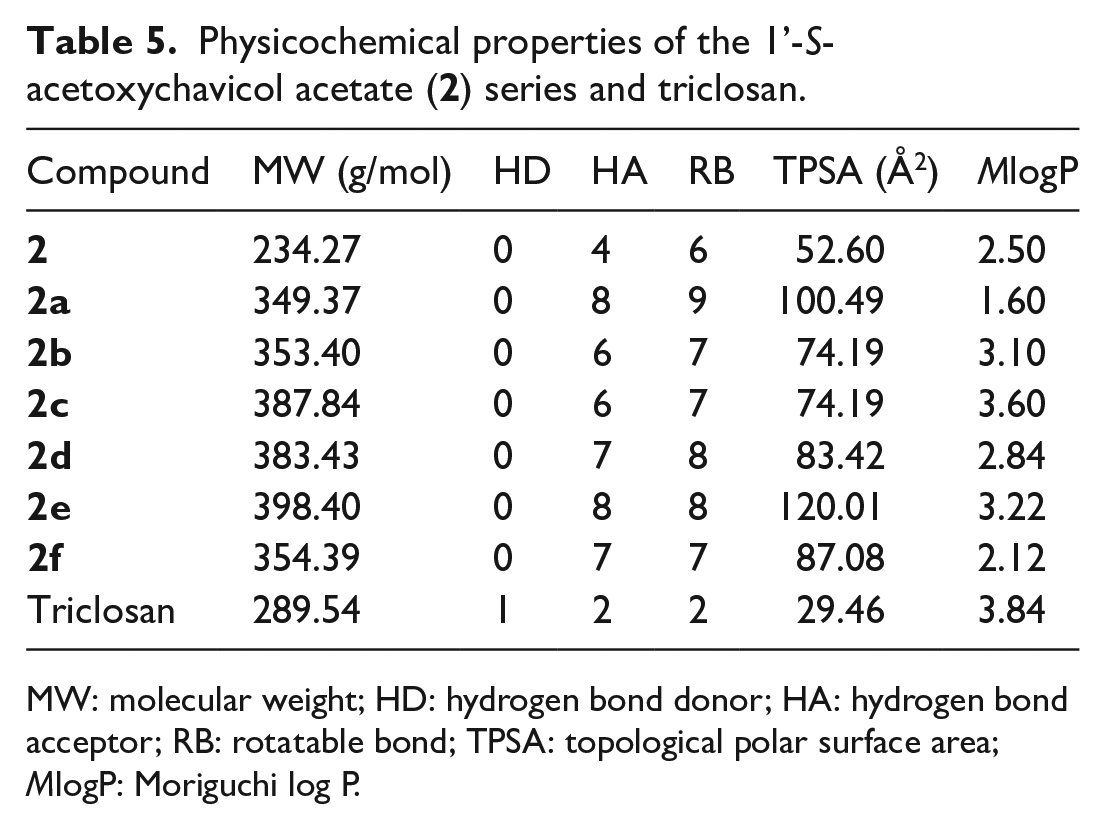
MW: molecular weight; HD: hydrogen bond donor; HA: hydrogen bond acceptor; RB: rotatable bond; TPSA: topological polar surface area; MlogP: Moriguchi log P.
Conclusion
In this study, 18 isoxazoline derivatives of natural products
Experimental
Chemicals and instrumentation
All commercially available chemicals and solvents were purchased from Sigma Aldrich and Santa Cruz and were used without purification. 1’S-acetoxychavicol acetate (
The NMR spectra of the studied compounds were recorded on a Bruker Avance 300 spectrometer ( 1 H NMR at 300 MHz and 13C NMR at 75 MHz). The values of the chemical shifts (δ) are reported in ppm, with coupling constants (J) being reported in Hertz. The abbreviations used in the NMR assignments are as follows: s = singlet; d = doublet; t = triplet; q = quartet; m = multiplet; br = broad. HRMS data were obtained using a Bruker micro time-of-flight mass spectrometer. IR spectra were recorded on a Perkin Elmer Spectrum ONE spectrophotometer using the universal attenuated total reflectance technique. Melting points (m.p.) were determined in open capillary tubes using a Büchi 535 melting point apparatus without correction. Flash column chromatography was executed using columns packed with silica gel 60 (Merck, 0.063–0.200 mm), eluting with the stated solvent system. Thin-layer chromatography was performed on Merck DC-Alufolien Kieselgel 60F254 aluminium-backed silica plates. Each plate, after being developed with an appropriate solvent system, was visualised under ultraviolet light (λ = 254 or 365 nm).
Synthesis of isoxazoline derivatives 1a–f, 2a–f and 3a–f
Oxime chloride
Ethyl 5-(4-hydroxy-3-methoxybenzyl)-4,5-dihydroisoxazole-3-carboxylate (1a)
Colourless oil (52.5 mg, 75%). IR (UATR): 3449, 2940, 1717, 1589, 1515, 1465, 1433, 1378, 1269, 1254, 1125 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.44 (s, 1H, OH), 6.92 (d, J = 1.8 Hz, 1H, ArH), 6.84–6.60 (m, 2H, 2×ArH), 5.04 (ddt, J = 6.5, 8.3, 10.8 Hz, 1H, CH), 4.25 (q, J = 7.1 Hz, 2H, OCH2), 3.83 (s, 3H, OCH3), 3.21 (dd, J = 10.8, 17.5 Hz, 1H, CH2), 2.96 (dd, overlapping, 1H, CH2), 2.95 (dd, overlapping, 1H, CH2), 2.84 (dd, J = 6.7, 13.9 Hz, 1H, CH2), 1.28 (t, J = 7.1 Hz, 3H, CH3). 13C NMR (75 MHz, acetone-d6): δ 161.3, 152.4, 148.3, 146.3, 128.9, 122.8, 115.7, 113.8, 85.2, 62.0, 56.2, 40.8, 38.5, 14.3. HRMS (APCI): m/z [M + H]+ calcd for C14H18NO5: 280.1180; found: 280.1190.
2-Methoxy-4-((3-phenyl-4,5-dihydroisoxazol-5-yl)methyl)phenol (1b)
Off-white solid (44.8 mg, 63%); m.p. 90–91 °C (lit. 40 m.p. 100–101 °C). IR (UATR): 3421, 2937, 1765, 1602, 1514, 1448, 1432, 1356, 1268, 1237, 1153 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.71–7.63 (m, 2H, 2×ArH), 7.47–7.35 (m, 3H, 3×ArH), 7.40 (s, overlapping, 1H, OH), 6.96 (d, J = 1.1 Hz, 1H, ArH), 6.77 (d, J = 1.1 Hz, 2H, 2×ArH), 4.95 (dddd, J = 6.2, 6.8, 8.0, 10.3 Hz, 1H, CH), 3.84 (s, 3H, OCH3), 3.39 (dd, J = 10.3, 16.8 Hz, 1H, CH2), 3.14 (dd, J = 8.0, 16.9 Hz, 1H, CH2), 3.00 (dd, J = 6.2, 13.8 Hz, 1H, CH2), 2.83 (dd, overlapping, 1H, CH2). 13C NMR (75 MHz, acetone-d6): δ 157.0, 148.2, 146.1, 131.2, 130.5, 129.6, 129.4 (2), 127.3 (2), 122.7, 115.7, 113.8, 83.0, 56.2, 41.2, 39.7. HRMS (ESI+): m/z [M + Na]+ calcd for C17H17NO3Na: 306.1101; found: 306.1100.
4-((3-(4-Chlorophenyl)-4,5-dihydroisoxazol-5-yl)methyl)-2-methoxyphenol (1c)
Yellow oil (62.0 mg, 78%). IR (UATR): 3415, 2936, 2884, 1597, 1514, 1495, 1432, 1403, 1351, 1268, 1236, 1152 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.73–7.63 (m, 2H, 2×ArH), 7.52–7.41 (m, 2H, 2×ArH), 7.39 (s, 1H, OH), 6.95 (s, 1H, ArH), 6.76 (d, J = 1.1 Hz, 2H, 2×ArH), 4.97 (dddd, J = 6.2, 6.8, 8.0, 10.3 Hz, 1H, CH), 3.84 (s, 3H, OCH3), 3.40 (dd, J = 10.3, 16.9 Hz, 1H, CH2), 3.15 (dd, J = 8.0, 16.9 Hz, 1H, CH2), 3.00 (dd, J = 6.2, 13.9 Hz, 1H, CH2), 2.84 (dd, overlapping, 1H, CH2). 13C NMR (75 MHz, acetone-d6): δ 156.2, 148.2, 146.1, 135.7, 129.9, 129.6 (2), 129.5, 128.9 (2), 122.7, 115.7, 113.8, 83.3, 56.2, 41.1, 39.5. HRMS (APCI): m/z [M + H]+ calcd for C17H17NO3Cl: 318.0891; found: 318.0897.
2-Methoxy-4-((3-(4-methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)phenol (1d)
White solid (69.1 mg, 88%); m.p. 75–76 °C. IR (UATR): 3422, 2936, 2839, 1607, 1514, 1462, 1432, 1357, 1253, 1177, 1153 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.65–7.57 (m, 2H, 2×ArH), 7.37 (s, 1H, OH), 6.99–6.94 (m, 3H, 3×ArH), 6.76 (d, J = 1.1 Hz, 2H, 2×ArH), 5.01–4.78 (m, 1H, CH), 3.84 (s, 3H, OCH3), 3.83 (s, 3H, OCH3), 3.35 (dd, J = 10.2, 16.7 Hz, 1H, CH2), 3.10 (dd, J = 8.0, 16.7 Hz, 1H, CH2), 2.98 (dd, J = 6.2, 13.8 Hz, 1H, CH2), 2.84 (dd, overlapping, 1H, CH2). 13C NMR (75 MHz, acetone-d6): δ 161.8, 156.5, 148.2, 146.1, 129.7, 128.8 (2), 123.6, 122.7, 115.7, 114.8 (2), 113.8, 82.6, 56.2, 55.6, 41.2, 40.0. HRMS (APCI): m/z [M + H]+ calcd for C18H20NO4: 314.1386; found: 314.1398.
2-Methoxy-4-((3-(4-nitrophenyl)-4,5-dihydroisoxazol-5-yl)methyl)phenol (1e)
Yellow solid (70.1 mg, 85%); m.p. 128–129 °C. IR (UATR): 3495, 2937, 2846, 1600, 1574, 1513, 1432, 1343, 1268, 1236, 1152 cm−1. 1H NMR (300 MHz, acetone-d6): δ 8.31–8.16 (m, 2H, 2×ArH), 7.98–7.85 (m, 2H, 2×ArH), 7.42 (s, 1H, OH), 6.96 (d, J = 1.2 Hz, 1H, ArH), 6.77 (d, J = 1.1 Hz, 2H, 2×ArH), 5.06 (ddt, J = 6.5, 7.9, 10.5 Hz, 1H, CH), 3.83 (s, 3H, OCH3), 3.48 (dd, J = 10.5, 16.9 Hz, 1H, CH2), 3.22 (dd, J = 8.1, 16.9 Hz, 1H, CH2), 3.03 (dd, J = 6.2, 13.9 Hz, 1H, CH2), 2.88 (dd, overlapping, 1H, CH2). 13C NMR (75 MHz, acetone-d6): δ 156.1, 149.1, 148.2, 146.2, 137.2, 129.3, 128.2 (2), 124.6 (2), 122.8, 115.7, 113.8, 84.1, 56.2, 41.0, 39.1. HRMS (APCI): m/z [M + H]+ calcd for C17H17N2O5: 329.1132; found: 329.1141.
2-Methoxy-4-((3-(pyridin-2-yl)-4,5-dihydroisoxazol-5-yl)methyl)phenol (1f)
Yellow oil (45.5 mg, 64%). IR (UATR): 3397, 2937, 1581, 1514, 1470, 1431, 1363, 1269, 1236, 1208, 1153 cm−1. 1H NMR (300 MHz, acetone-d6): δ 8.58 (ddd, J = 1.0, 1.8, 4.9 Hz, 1H, ArH), 7.94 (dt, J = 1.1, 8.0 Hz, 1H, ArH), 7.81 (td, J = 1.8, 7.7 Hz, 1H, ArH), 7.45 (s, 1H, OH), 7.37 (ddd, J = 1.2, 4.9, 7.5 Hz, 1H, ArH), 6.95 (s, 1H, ArH), 6.77 (s, 2H, 2×ArH), 4.99 (ddt, J = 6.5, 8.1, 10.4 Hz, 1H, CH), 3.83 (s, 3H, OCH3), 3.45 (dd, J = 10.4, 17.6 Hz, 1H, CH2), 3.22 (dd, J = 8.1, 17.6 Hz, 1H, CH2), 3.00 (dd, J = 6.3, 13.9 Hz, 1H, CH2), 2.86 (dd, J = 6.7, 13.9 Hz, 1H, CH2). 13C NMR (75 MHz, acetone-d6): δ 159.1, 150.7, 150.2, 148.2, 146.1, 137.2, 129.5, 124.9, 122.8, 121.8, 115.7, 113.8, 83.6, 56.2, 41.1, 39.5. HRMS (ESI+): m/z [M + H]+ calcd for C16H17N2O3: 285.1250; found: 285.1247.
Ethyl (R/S)-5-((R)-acetoxy(4-acetoxyphenyl)methyl)-4,5-dihydroisoxazole-3-carboxylate (2a)
Colourless oil (89.8 mg, 51% (major/minor, 1.04:1.00)). IR (UATR): 2985, 2938, 1742, 1719, 1594, 1508, 1371, 1194, 1124 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.59–7.41 (m, 4H, 4×ArH), 7.24–7.07 (m, 4H, 4×ArH), 5.99 (d, J = 4.4 Hz, 1H, CH, minor), 5.90 (d, J = 6.5 Hz, 1H, CH, major), 5.32–5.05 (m, 2H, 2×CH), 4.26 (qd, J = 1.3, 7.1 Hz, 4H, 2×CH2), 3.33–3.16 (m, overlapping, 3H, 2×CH2), 3.01 (dd, J = 7.8, 17.8 Hz, 1H, CH2), 2.26 (s, 3H, OAc, minor), 2.25 (s, 3H, OAc, major), 2.09 (s, 3H, OAc, minor), 2.07 (s, 3H, OAc, major), 1.28 (t, J = 7.1 Hz, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 170.1, 169.9, 169.5, 161.0, 160.9, 152.5 (2), 152.1, 151.9, 135.0, 134.6, 129.6, 128.9, 122.8 (2), 85.4, 85.0, 76.0, 74.9, 62.2 (2), 36.7, 35.5, 21.0, 20.9, 20.8, 14.3. HRMS (ESI+): m/z [M + Na]+ calcd for C17H19NO7Na: 372.1054; found: 372.1051.
4-((R)-Acetoxy((R/S)-3-phenyl-4,5-dihydroisoxazol-5-yl)methyl)phenyl acetate (2b)
Colourless oil (35.8 mg, 40% (major/minor, 1.02:1.00)). IR (UATR): 3059, 2934, 1746, 1607, 1508, 1447, 1370, 1195, 1167 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.72–7.62 (m, 4H, 4×ArH), 7.60–7.49 (m, 4H, 4×ArH), 7.46–7.39 (m, 6H, 6×ArH), 7.22–7.11 (m, 4H, 4×ArH), 5.98 (d, J = 4.8 Hz, 1H, CH, major), 5.91 (d, J = 6.8 Hz, 1H, CH, minor), 5.22–5.04 (m, 2H, 2×CH), 3.57–3.31 (m, overlapping, 3H, 2×CH2), 3.19 (dd, J = 7.6, 17.2 Hz, 1H, CH2), 2.26 (s, 3H, OAc, minor), 2.25 (s, 3H, OAc, major), 2.06 (s, 3H, OAc, minor), 2.05 (s, 3H, OAc, major). 13C NMR (75 MHz, acetone-d6): δ 170.1, 170.0, 169.5, 157.1, 157.0, 152.0, 151.8, 135.5, 135.3, 130.7 (2), 129.6, 129.5 (2), 129.0, 127.4, 122.7, 122.6, 83.4, 83.0, 76.4, 75.4, 37.7, 36.7, 20.9 (3). HRMS (ESI+): m/z [M + Na]+ calcd for C20H19NO5Na: 376.1155; found: 376.1150.
4-((R)-Acetoxy((R/S)-3-(4-chlorophenyl)-4,5-dihydroisoxazol-5-yl)methyl)phenyl acetate (2c)
Light yellow oil (39.4 mg, 40% (major/minor, 1.16:1.00)). IR (UATR): 2936, 1743, 1598, 1508, 1495, 1432, 1403, 1369, 1194, 1167 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.71–7.64 (m, 4H, 4×ArH), 7.58–7.50 (m, 4H, 4×ArH), 7.45 (dq, J = 2.5, 9.1 Hz, 4H, 4×ArH), 7.19–7.10 (m, 4H, 4×ArH), 5.98 (d, J = 4.7 Hz, 1H, CH, minor), 5.91 (d, J = 6.8 Hz, 1H, CH, major), 5.22–5.04 (m, 2H, 2×CH), 3.50–3.33 (m, overlapping, 3H, 2×CH2), 3.19 (dd, J = 7.6, 17.2 Hz, 1H, CH2), 2.26 (s, 3H, OAc, minor), 2.25 (s, 3H, OAc, major), 2.06 (s, 3H, OAc, minor), 2.05 (s, 3H, OAc, major). 13C NMR (75 MHz, acetone-d6): δ 170.1, 170.0, 169.5, 156.3 (2), 152.0, 151.8, 136.0, 135.3, 135.1, 129.6 (2), 129.4, 129.0, 128.9, 122.7, 122.6, 83.7, 83.3, 76.3, 75.3, 37.5, 36.5, 20.9 (3). HRMS (ESI+): m/z [M + Na]+ calcd for C20H18NO5ClNa: 410.0766; found: 410.0759.
4-((R)-Acetoxy((R/S)-3-(4-methoxyphenyl)-4,5-dihydroisoxazol-5-yl)methyl)phenyl acetate (2d)
Colourless oil (54.5 mg, 57% (major/minor, 1.15:1.00)). IR (UATR): 2938, 1747, 1608, 1515, 1369, 1306, 1252, 1230, 1196 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.68–7.55 (m, 4H, 4×ArH), 7.57–7.49 (m, 4H, 4×ArH), 7.21–7.09 (m, 4H, 4×ArH), 7.02–6.89 (m, 4H, 4×ArH), 5.96 (d, J = 4.8 Hz, 1H, CH, minor), 5.89 (d, J = 6.8 Hz, 1H, CH, major), 5.16–4.96 (m, 2H, 2×CH), 3.83 (s, 3H, OCH3), 3.83 (s, 3H, OCH3), 3.55–3.29 (m, overlapping, 3H, 2×CH2), 3.14 (dd, J = 7.6, 17.1 Hz, 1H, CH2), 2.26 (s, 3H, OAc, major), 2.25 (s, 3H, OAc, minor), 2.06 (s, 3H, OAc, minor), 2.05 (s, 3H, OAc, major). 13C NMR (75 MHz, acetone-d6): δ 170.1, 170.0, 169.5, 162.0, 156.5 (2), 151.9, 151.7, 135.5, 135.4, 132.6, 130.2, 129.6, 129.0, 123.0, 122.7, 122.6, 114.9 (2), 83.1, 82.6, 76.5, 75.4, 55.7, 37.9, 37.0, 20.9 (3). HRMS (ESI+): m/z [M + Na]+ calcd for C21H21NO6Na: 406.1261; found: 406.1253.
4-((R)-Acetoxy((R/S)-3-(4-nitrophenyl)-4,5-dihydroisoxazol-5-yl)methyl)phenyl acetate (2e)
Yellow oil (55.0 mg, 55% (major/minor, 1.04:1.00)). IR (UATR): 3079, 2938, 1744, 1608, 1579, 1518, 1435, 1369, 1345, 1194, 1167 cm−1. 1H NMR (300 MHz, acetone-d6): δ 8.27 (dq, J = 2.3, 9.3 Hz, 4H, 4×ArH), 7.99–7.87 (m, 4H, 4×ArH), 7.61–7.49 (m, 4H, 4×ArH), 7.23–7.10 (m, 4H, 4×ArH), 6.02 (d, J = 4.5 Hz, 1H, CH, minor), 5.95 (d, J = 6.7 Hz, 1H, CH, major), 5.32–5.14 (m, 2H, 2×CH), 3.60–3.47 (m, overlapping, 3H, 2×CH2), 3.31 (dd, J = 7.6, 17.3 Hz, 1H, CH2), 2.26 (s, 3H, OAc, major), 2.25 (s, 3H, OAc, minor), 2.07 (s, 3H, OAc, minor), 2.06 (s, 3H, OAc, major). 13C NMR (75 MHz, acetone-d6): δ 170.1, 170.0, 169.5, 156.3, 156.2, 152.0, 151.8, 149.3 (2), 136.6 (2), 135.2, 134.9, 129.6, 128.9, 128.5, 124.6 (2), 122.7 (2), 84.5, 84.0, 76.2, 75.2, 37.2, 36.1, 20.9 (2), 20.8. HRMS (ESI+): m/z [M + Na]+ calcd for C20H18N2O7Na: 421.1006; found: 421.0997.
4-((R)-Acetoxy((R/S)-3-(pyridin-2-yl)-4,5-dihydroisoxazol-5-yl)methyl)phenyl acetate (2f)
Colourless oil (41.7 mg, 47% (major/minor, 1.04:1.00)). IR (UATR): 3056, 2931, 1745, 1584, 1508, 1472, 1440, 1367, 1193, 1167 cm−1. 1H NMR (300 MHz, acetone-d6): δ 8.59 (dddd, J = 1.0, 1.8, 4.9, 8.4 Hz, 2H, 2×ArH), 7.92 (ddt, J = 1.2, 6.6, 7.9 Hz, 2H, 2×ArH), 7.86–7.78 (m, 2H, 2×ArH), 7.61–7.48 (m, 4H, 4×ArH), 7.39 (dddd, J = 1.3, 2.3, 4.9, 7.4 Hz, 2H, 2×CH), 7.25–7.10 (m, 4H, 4×ArH), 6.01 (d, J = 4.7 Hz, 1H, CH, minor), 5.93 (d, J = 6.8 Hz, 1H, CH, major), 5.29–5.06 (m, 2H, 2×CH), 3.58–3.37 (m, overlapping, 3H, 2×CH2), 3.25 (dd, J = 7.7, 17.9 Hz, 1H, CH2), 2.26 (s, 3H, OAc, minor), 2.25 (s, 3H, OAc, major), 2.07 (s, 3H, OAc, minor), 2.06 (s, 3H, OAc, major). 13C NMR (75 MHz, acetone-d6): δ 170.1, 170.0, 169.5, 159.0, 151.9, 151.8, 150.3, 150.1, 137.3 (2), 135.4, 135.1, 129.6, 129.0, 125.2 (2), 122.7, 122.6, 121.9 (2), 83.9, 83.5, 76.4, 75.3, 37.5, 36.5, 20.9 (2), 20.8. HRMS (ESI+): m/z [M + Na]+ calcd for C19H18N2O5Na: 377.1108; found: 377.1102.
Ethyl (R/S)-5-((R)-2-hydroxy-4-((1R,2R,4aS,8aS)-2-hydroxy-2,5,5,8a-tetramethyldecahydronaphthalen-1-yl)butan-2-yl)-4,5-dihydroisoxazole-3-carboxylate (3a)
White solid (68.4 mg, 65% (major/minor, 1.05:1.00)); m.p. 58–59 °C. IR (UATR): 3375, 2927, 2869, 1720, 1592, 1464, 1387, 1256, 1128 cm−1. 1H NMR (300 MHz, acetone-d6): δ 4.74–4.67 (m, 1H, CH, major), 4.66–4.60 (m, 1H, CH, minor), 4.27 (qd, J = 1.0, 7.1 Hz, 4H, 2×OCH2), 4.05 (br s, 1H, OH, minor), 3.90 (br s, 1H, OH, major), 3.52 (br s, 1H, OH, minor), 3.43 (br s, 1H, OH, major), 3.33–3.05 (m, 4H, 2×CH2), 1.90–1.16 (m, overlapping, 28H, 14×CH2), 1.29 (t, J = 7.1 Hz, 6H, 2×CH3), 1.22 (s, 3H, CH3, minor), 1.15 (s, 3H, CH3, major), 1.14 (s, 3H, CH3, major), 1.11 (s, 3H, CH3, minor), 1.07–0.90 (m, 4H, 4×CH), 0.87 (s, 6H, 2×CH3), 0.83 (s, 6H, 2×CH3), 0.81 (s, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 161.4, 152.5, 152.4, 90.6, 90.2, 89.7, 74.2, 74.1, 73.6, 73.5, 62.7 (2), 61.9 (2), 57.0, 45.0, 44.9, 42.8, 42.7, 42.4, 41.9, 40.5, 40.4, 39.9 (2), 34.6, 33.8, 33.7, 24.5, 24.4, 22.6, 22.0, 21.8, 21.2 (2), 21.1, 19.6, 19.1, 15.8 (2), 14.3. HRMS (ESI+): m/z [M + Na]+ calcd for C24H41NO5Na: 446.2877; found: 446.2866.
(1R,2R,4aS,8aS)-1-((R)-3-Hydroxy-3-((R/S)-3-phenyl-4,5-dihydroisoxazol-5-yl)butyl)-2,5,5,8a-tetramethyldecahydronaphthalen-2-ol (3b)
White solid (63.0 mg, 59% (major/minor, 1.11:1.00)); m.p. 79–80 °C. IR (UATR): 3365, 2924, 2868, 1600, 1463, 1446, 1388, 1359 1264, 1134 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.73–7.68 (m, 4H, 4×ArH), 7.45–7.40 (m, 6H, 6×ArH), 4.66–4.59 (m, 1H, CH, major), 4.59–4.53 (m, 1H, CH, minor), 3.92 (br s, 1H, OH, minor), 3.70 (br s, 1H, OH, major), 3.59 (br s, 1H, OH, minor), 3.54–3.20 (m, 4H, 2×CH2), 3.39 (br s, overlapping, 1H, OH, major), 1.85–1.25 (m, 28H, 14×CH2), 1.22 (s, 3H, CH3, minor), 1.17 (s, 3H, CH3, major), 1.15 (s, 3H, CH3, minor), 1.14 (s, 3H, CH3, major), 1.03–0.88 (m, 4H, 4×CH), 0.86 (s, 6H, 2×CH3), 0.83 (s, 6H, 2×CH3), 0.80 (s, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 157.1 (2), 131.3, 131.2, 130.4 (2), 129.4 (2), 127.3, 88.3, 88.1, 74.2, 74.1, 73.7 (2), 62.8 (2), 57.0, 45.0, 44.9, 42.8, 42.7, 42.0, 40.5, 40.4, 39.9 (2), 35.8, 33.8, 33.7, 24.5 (2), 22.9, 22.0, 21.8, 21.2, 21.1, 19.7, 19.2, 19.1 (2), 15.9. HRMS (ESI+): m/z [M + Na]+ calcd for C27H41NO3Na: 450.2979; found: 450.2969.
(1R,2R,4aS,8aS)-1-((R)-3-((R/S)-3-(4-Chlorophenyl)-4,5-dihydroisoxazol-5-yl)-3-hydroxybutyl)-2,5,5,8a-tetramethyldecahydronaphthalen-2-ol (3c)
Light yellow oil (50.0 mg, 43% (major/minor, 1.14:1.00)). IR (UATR): 3367, 2924, 2868, 1598, 1495, 1462, 1403, 1388, 1263, 1133 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.72 (d, J = 8.5 Hz, 4H, 4×ArH), 7.48–7.44 (m, 4H, 4×ArH), 4.69–4.62 (m, 1H, CH, major), 4.61–4.55 (m, 1H, CH, minor), 3.97 (br s, 1H, OH, minor), 3.75 (br s, 1H, OH, major), 3.63 (br s, 1H, OH, minor), 3.52–3.27 (m, 4H, 2×CH2), 3.41 (br s, overlapping, 1H, OH, major), 1.99–1.20 (m, 28H, 14×CH2), 1.23 (s, 3H, CH3, minor), 1.16 (s, 3H, CH3, major), 1.15 (s, 3H, CH3, minor), 1.14 (s, 3H, CH3, major), 1.01–0.88 (m, 4H, 4×CH), 0.86 (s, 6H, 2×CH3), 0.83 (s, 6H, 2×CH3), 0.80 (s, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 156.3 (2), 135.7(2), 130.1, 130.0, 129.6 (2), 128.9, 128.8, 88.7, 88.4, 74.2, 74.1, 73.7 (2), 62.8, 62.7, 57.0, 45.0, 44.8, 42.8, 42.7 (2), 42.1, 40.4, 39.9 (2), 35.6, 33.8, 33.7, 24.6, 24.5, 22.9, 22.0, 21.8, 21.2, 21.1, 19.7, 19.2, 19.1, 15.9. HRMS (ESI+): m/z [M + Na]+ calcd for C27H40NO3ClNa: 484.2589; found: 484.2584.
(1R,2R,4aS,8aS)-1-((R)-3-Hydroxy-3-((R/S)-3-(4-methoxyphenyl)-4,5-dihydroisoxazol-5-yl)butyl)-2,5,5,8a-tetramethyldecahydronaphthalen-2-ol (3d)
Yellow oil (60.0 mg, 52% (major/minor, 1.71:1.00)). IR (UATR): 3367, 2924, 2868, 1609, 1517, 1462, 1359, 1307, 1253, 1176 cm−1. 1H NMR (300 MHz, acetone-d6): δ 7.69–7.59 (m, 4H, 4×ArH), 6.97 (dd, J = 1.9, 8.9 Hz, 4H, 4×ArH), 4.57 (dd, J = 9.3, 11.0 Hz, 1H, CH, major), 4.49 (dd, J = 8.7, 11.1 Hz, 1H, CH, minor), 3.95 (br s, 1H, OH, minor), 3.83 (s, 6H, 2×OCH3), 3.72 (br s, 1H, OH, major), 3.48–3.19 (m, 4H, 2×CH2), 1.88–1.18 (m, 28H, 14×CH2), 1.16 (s, 6H, 2×CH3), 1.14 (s, 6H, 2×CH3), 1.00–0.87 (m, 4H, 4×CH), 0.85 (s, 6H, 2×CH3), 0.83 (s, 6H, 2×CH3), 0.80 (s, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 161.7, 156.6, 156.5, 128.8 (2), 123.7, 123.6, 114.8 (2), 88.1, 87.8, 74.3, 74.1, 73.8, 73.6, 62.8, 62.7, 56.9, 55.6, 44.9, 44.7, 42.7, 42.6, 41.9, 40.4, 39.9 (2), 36.1, 33.8, 33.7 (2), 24.6, 24.5, 22.9, 22.0, 21.8, 21.2, 21.1, 19.6, 19.1, 15.9. HRMS (ESI+): m/z [M + Na]+ calcd for C28H43NO4Na: 480.3084; found: 480.3072.
(1R,2R,4aS,8aS)-1-((R)-3-Hydroxy-3-((R/S)-3-(4-nitrophenyl)-4,5-dihydroisoxazol-5-yl)butyl)-2,5,5,8a-tetramethyldecahydronaphthalen-2-ol (3e)
Off-white powder (86.2 mg, 73% (major/minor, 1.13:1.00)); m.p. 88–89 °C. IR (UATR): 3369, 2925, 2868, 1600, 1578, 1520, 1462, 1388, 1341, 1133, 1109 cm−1. 1H NMR (300 MHz, acetone-d6): δ 8.30 (dq, J = 2.4, 9.3 Hz, 4H, 4×ArH), 8.03–7.92 (m, 4H, 4×ArH), 4.79–4.73 (m, 1H, CH, major), 4.72–4.66 (m, 1H, CH, minor), 4.02 (br s, 1H, OH, minor), 3.82 (br s, 1H, OH, major), 3.57 (br s, 1H, OH, minor), 3.56–3.38 (m, 4H, 2×CH2), 3.37 (br s, 1H, OH, major), 1.96–1.19 (m, overlapping, 28H, 14×CH2), 1.25 (s, 3H, CH3, minor), 1.18 (s, 3H, CH3, major), 1.15 (s, 3H, CH3, minor), 1.14 (s, 3H, CH3, major), 0.98–0.87 (m, 4H, 4×CH), 0.86 (s, 3H, CH3, major), 0.85 (s, 3H, CH3, minor), 0.83 (s, 6H, 2×CH3), 0.80 (s, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 156.3, 156.2, 149.1, 137.3 (2), 128.3, 128.2, 124.7, 124.6, 89.5 (2), 89.2, 88.7, 74.2, 74.1, 73.8, 73.7, 62.8 (2), 57.0, 45.3, 45.0, 44.9, 42.8 (2), 42.6, 42.3, 42.1, 40.6, 40.5, 39.9 (2), 35.3, 33.8, 33.7, 24.6, 24.5, 22.9, 22.8, 22.1, 21.8, 21.2, 21.1, 19.8, 19.7, 19.2, 19.1, 15.9. HRMS (ESI+): m/z [M + Na]+ calcd for C27H40N2O5Na: 495.2829; found: 495.2813.
(1R,2R,4aS,8aS)-1-((R)-3-Hydroxy-3-((R/S)-3-(pyridin-2-yl)-4,5-dihydroisoxazol-5-yl)butyl)-2,5,5,8a-tetramethyldecahydronaphthalen-2-ol (3f)
Light yellow oil (21.0 mg, 20% (major/minor, 1.53:1.00)). IR (UATR): 3375, 2925, 2868, 1708, 1583, 1566, 1471, 1441, 1388, 1365, 1254, 1151, 1133 cm−1. 1H NMR (300 MHz, acetone-d6): δ 8.66–8.56 (m, 2H, 2×ArH), 7.99–7.89 (m, 2H, 2×ArH), 7.92–7.76 (m, 2H, 2×ArH), 7.45–7.35 (m, 2H, 2×ArH), 4.73–4.66 (m, 1H, CH, major), 4.65–4.57 (m, 1H, CH, minor), 3.55–3.32 (m, 4H, 2×CH2), 1.94–1.15 (m, 28H, 14×CH2), 1.17 (s, 6H, 2×CH3), 1.14 (s, 6H, 2×CH3), 1.03–0.92 (m, 4H, 4×CH), 0.87 (s, 6H, 2×CH3), 0.84 (s, 6H, 2×CH3), 0.81 (s, 6H, 2×CH3). 13C NMR (75 MHz, acetone-d6): δ 159.3, 150.2 (2), 137.2, 124.9, 124.8, 121.7, 89.0, 88.9, 88.6, 82.8, 74.0, 73.8, 73.7 (2), 62.9, 62.8, 62.7 (2), 57.1, 57.0, 45.3, 45.1, 42.8 (2), 42.7, 42.2 (2), 40.6, 40.5 (2), 40.0, 39.9, 36.9, 35.8, 35.6, 33.8 (2), 33.7, 24.5, 22.9, 22.6, 22.2, 21.8, 21.2, 19.7, 19.2, 19.1, 15.9. HRMS (ESI+): m/z [M + Na]+ calcd for C26H40N2O3Na: 451.2931; found: 451.2927.
Antitubercular activity assay
The MIC values used to determine the activities of the compounds against M. tuberculosis were determined using a microplate Alamar Blue assay. 41 All the tested compounds were individually dissolved in dimethyl sulfoxide (DMSO) as stock solutions (10,000 µg/mL). Each solution (100 µL) of the tested compound in Middlebrook 7H9GC (concentration 400 µg/mL) was diluted twofold in 100 µL of Middlebrook 7H9GC in 96-well microplates and subsequently diluted serially. The prepared M. tuberculosis suspension (100 µL; at approximately 105–106 CFU/mL) was added to each microplate well (the highest concentration 100 µg/mL and the lowest concentration 0.1 µg/mL), and the microplates were incubated at 37 °C for 7 days. Both 20% Tween 80 (12.5 µL) and Alamar Blue (20 µL) were added to each microplate well, and the microplates were reincubated at 37 °C for 16–24 h. Mycobacterial growth was determined by a change in colour from blue to pink. If no colour change occurred, the MIC value was recorded as the lowest concentration. The M. tuberculosis strain H37Ra used in this study was obtained from a stock culture at Ramathibodi Hospital, Bangkok, Thailand. Rifampin was used as a standard drug for comparison.
Antibacterial activity assay
The MIC values were determined using the broth microdilution method in accordance with the guidelines of the European Society of Clinical Microbiology and Infectious Diseases. 31 All synthetic compounds were evaluated on both Gram-positive bacteria (B. cereus and S. aureus) and Gram-negative bacteria (E. coli and P. aeruginosa). The standard drugs chloramphenicol and tetracycline were used as reference compounds.
Cytotoxic activity assay
The cytotoxic activities of all synthetic compounds against four cancer cell lines were evaluated. The cytotoxic activities against the adhesive cell lines HuCCA-1, A549 and HepG2 were evaluated using the MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide] assay. 42 Cytotoxicity against the non-adhesive MOLT-3 cell line was evaluated using the XTT [2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide] assay.43,44 Doxorubicin and etoposide were used as standard drugs for comparison.
Expression and purification of InhA
Both the expression and purification of InhA were conducted using the previously reported method.
45
A culture of E. coli BL21(DE3) single competent cells carrying an InhA gene-inserted pET15b plasmid that contained N-terminal 6xHis-tag was grown in a large-scale Lysogeny Broth (LB) medium (5 L) containing ampicillin (100 µg/mL). The culture was incubated at 37 °C until an optical density (OD600) value of 0.6 was observed. Proteins were subsequently induced by adding a solution of 50 µM isopropyl-β-
InhA inhibitory activity assay
The kinetic assay for InhA inhibitory activity was conducted using the previously reported method. 45 In brief, a stock solution of each inhibitor was prepared in DMSO. The final concentration of DMSO relative to the final volume (100 μL) for all kinetic reactions was 1%. The relative inhibitory activities of all the tested compounds against InhA were evaluated in 30 mM PIPES buffer (pH 6.8) containing 150 mM NaCl, 2 mM DTT, 1 mM EDTA, 1 mg/mL bovine serum albumin, 10% v/v glycerol, 50 μM NADH, 75 μM trans-2-dodecenoyl-CoA (DD-CoA), and 1 μM inhibitor. The prepared 96-well microplate for all kinetic reactions was shaken for 1 min before 20 nM (final concentration) InhA was added. NADH was added to the reaction mixture as a substrate and incubated at 25 °C for 5 min, followed by the addition of the test compound and further incubation at 25 °C for 20 min. A solution of 75 μM DD-CoA was added to the reaction, which was monitored over 10 min at 25 °C and measured by following the oxidation of NADH to NAD+ at λex = 340 nm and λem = 420 nm. The fluorescence intensity of each well was read and recorded every 30 s. The relative inhibitory activity of each compound was determined as the percent of relative inhibition of InhA with respect to the control reaction, as indicated in equation (1)

where Fcompound is the fluorescence intensity of the InhA inhibitory activity for each compound concentration, Fpositive control is the fluorescence intensity of the InhA inhibitory activity in the absence of any compound, and Fnegative control is the fluorescence intensity of the NADH oxidation reaction in the absence of the InhA enzyme.
Molecular modelling
Ligands were constructed using Scigress (version 2.8.1); the chemical structures were drawn and the energy minimised using the MM2 46 force field, and all were saved in 3D format. The crystal structure of M. tuberculosis enoyl-ACP reductase (InhA) was obtained from the Protein Data Bank 47 ID:2B35 48 with a resolution of 2.30 Å. Discovery studio (version 3.5) was used to prepare the crystal structure for docking. Specifically, in this process, hydrogen atoms were added, and co-crystallised ligands were removed. The NADH substrate was retained at the binding site because a study documented that InhA inhibitors were bound in an NADH-dependent manner. 49 The centre of the InhA binding site was defined at coordinates (x = 23.6.17, y = 20.702, z = 57.042) with 10 Å radii. The basic amino acids lysine and arginine were deemed to be protonated. Furthermore, aspartic and glutamic acids were assumed to be deprotonated. The ChemPLP 50 scoring function was implemented to dock the ligands to InhA using the Genetic Optimisation for Ligand Docking software package (GOLD, version 5.8.1). 51 GOLD has been reported to be one of the best docking software packages available, demonstrating the best accuracy in identifying ligand pose in protein–ligand interactions. 52 Molecular docking was conducted at 100% efficiency at 50 docking runs per ligand. In addition, the physicochemical properties of the ligands were calculated using Dragon (version 7.0). 53
Footnotes
Acknowledgements
The authors thank P. Intachote, S. Sengsai and B. Saimanee for their help in cytotoxic activity testing. The authors also appreciate P. Poolchanuan’s advice regarding antibacterial activities. In addition, HRMS spectra and IR data were kindly provided by the staff at Chulabhorn Research Institute.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Center of Excellence on Environmental Health and Toxicology (EHT), Science & Technology Postgraduate Education and Research Development Office, Ministry of Education. C.K. expresses thanks for the postdoctoral grant provided by the EHT.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

