Abstract
Chromic acid oxidation of
Keywords
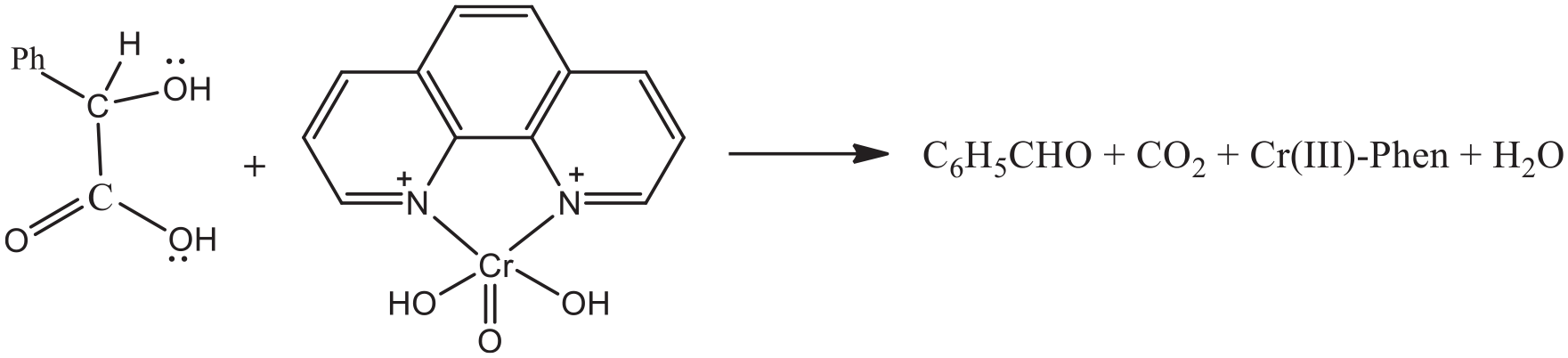
Introduction
Chromium(VI) acts as a powerful oxidizing agent in different types of redox reactions and is converted into chromium(III). Different mechanistic routes have been suggested for the reduction of chromium(VI) to chromium(III) from analysis of kinetic data and other experimental findings. The mode of reduction largely depends on the nature of the reductant and the experimental conditions.1–4 Intermediates like Cr(V) and Cr(IV) have been identified and characterized in many cases. To explore all these aspects, Cr(VI) has been extensively studied in redox kinetics. Here, it is important to note that Cr(VI) is hazardous because of its carcinogenic and mutagenic activity.5–8 This is why studies on the kinetics and mechanism of Cr(VI) oxidation of biologically relevant reducing agents are of interest to both biochemists and inorganic chemists.1,3 During the reduction of Cr(VI) to Cr(III), the intermediate oxidation states of chromium may interact with biologically active molecules and induce toxicity.
3
Thus, in terms of chromate toxicity, it is reasonable to assume that the reducing agent may have an important role. The present research paper deals with three-electron (3e) transfer Cr(VI) oxidation of
Results and discussion
Dependence on [Cr(VI)]T
Under the experimental conditions, [
Plot of log[Cr(VI)]T versus time (t): [Cr(VI)]T = 6 × 10−4 mol dm−3, [
Dependence on [Phen]T
The plots of kobs versus [Phen]T are linear (r > 0.99) with positive intercepts indicating the contribution of the relatively slower uncatalysed path (Figure 2). The pseudo first-order rate constants (kobs(u)) directly measured in the absence of Phen under the same conditions nicely agree with those obtained from the intercepts of the plots of kobs(T) versus [PA]T. In Figure 2, plots (a), (b) and (c) refer to the kinetic studies of the [1,10-phen] concentration variation at different temperatures (20 °C, 30 °C and 40 °C), respectively.

[Cr(VI)]T = 6 × 10−4 mol dm−3,
Dependence on [S]T, that is, [d,l -mandelic acid ]T
From the plot of kobs versus [

[Cr(VI)]T = 6 × 10−4 mol dm−3, [1,10-phen] = 12 × 10−4 mol dm−3, [H2SO4] = 0.25 mol dm−3, [
Kinetic parameters and some representative rate constants for the Cr(VI) oxidation of

ΔH≠ (kJ mol−1) 7.766.
ΔS≠ (J K−1 mol−1) −239.18 (Islam and Das 17 ).
Subscript (u) for uncatalysed path; (c) for [1,10-phen] catalysed path; (w) for the value in the absence of surfactant; (CPC) for the value in the presence of CPC.
kobs(u)(w) = rate constant of the uncatalysed reaction in aqueous medium.
kobs(c)(w) = rate constant of the 1,10-phenanthroline-catalysed reaction in aqueous medium.
kcat(w) = slope plot of kobs(c)(w) versus [1,10-phen] of the 1,10-phenanthroline-catalysed reaction in aqueous medium.
keff(w) = (kobs(c)(w)–kobs(u)(w))/kobs(u)(w), calculated at [1,10-phen] = 18 × 10−4 mol dm−3; [Cr(VI)] = 6 × 10−4 mol dm−3; [
[Cr(VI)] = 6 × 10−4 mol dm−3; [
[Cr(VI)] = 6 × 10−4 mol dm−3; [
[Cr(VI)] = 6 × 10−4 mol dm−3; [
[Cr(VI)] = 6 × 10−4 mol dm−3; [
Dependence on [H+]
The acid dependence patterns for the uncatalysed and catalysed paths are the same first-order dependence (Figure 4). From the experimental fit, the observations are as follows


Thus, the observed rate laws are as follows



[Cr(VI)]T = 6 × 10−4 mol dm−3, [1,10-phen] = 12 × 10−4 mol dm−3[
Test for acrylonitrile polymerization
Under the experimental conditions, polymerization of acrylonitrile occurred under a nitrogen atmosphere. This indicates the generation of free radicals during the reaction.
Evaluation of the activation parameters
From the studies of the effect of temperature on the rate constant (k), the activation parameters, ΔH≠ (enthalpy of activation) and ΔS≠ (entropy of activation), have been evaluated (Figure 5) by using the Eyring equation

where kB is the Boltzmann constant (1.38 × 10−23 J K−1), h is Planck’s constant (6.62 × 10−34 J s−1) and R is the molar gas constant (8.31 J K−1 mol−1). Free energy of activation (ΔG≠) and its errors can also be calculated from the following equations

and


Plot of–log(kh/kBT) versus 1/T (T = 20 °C; 30 °C; 40 °C).
Mechanism and interpretation
Reaction mechanism for the Phen-assisted chromic acid oxidation of mandelic acid
The results obtained from the Phen-assisted reactions can be explained by considering the reaction mechanism outlined in Scheme 1. Here, Phen readily forms a reactive cyclic Cr(VI)-Phen complex
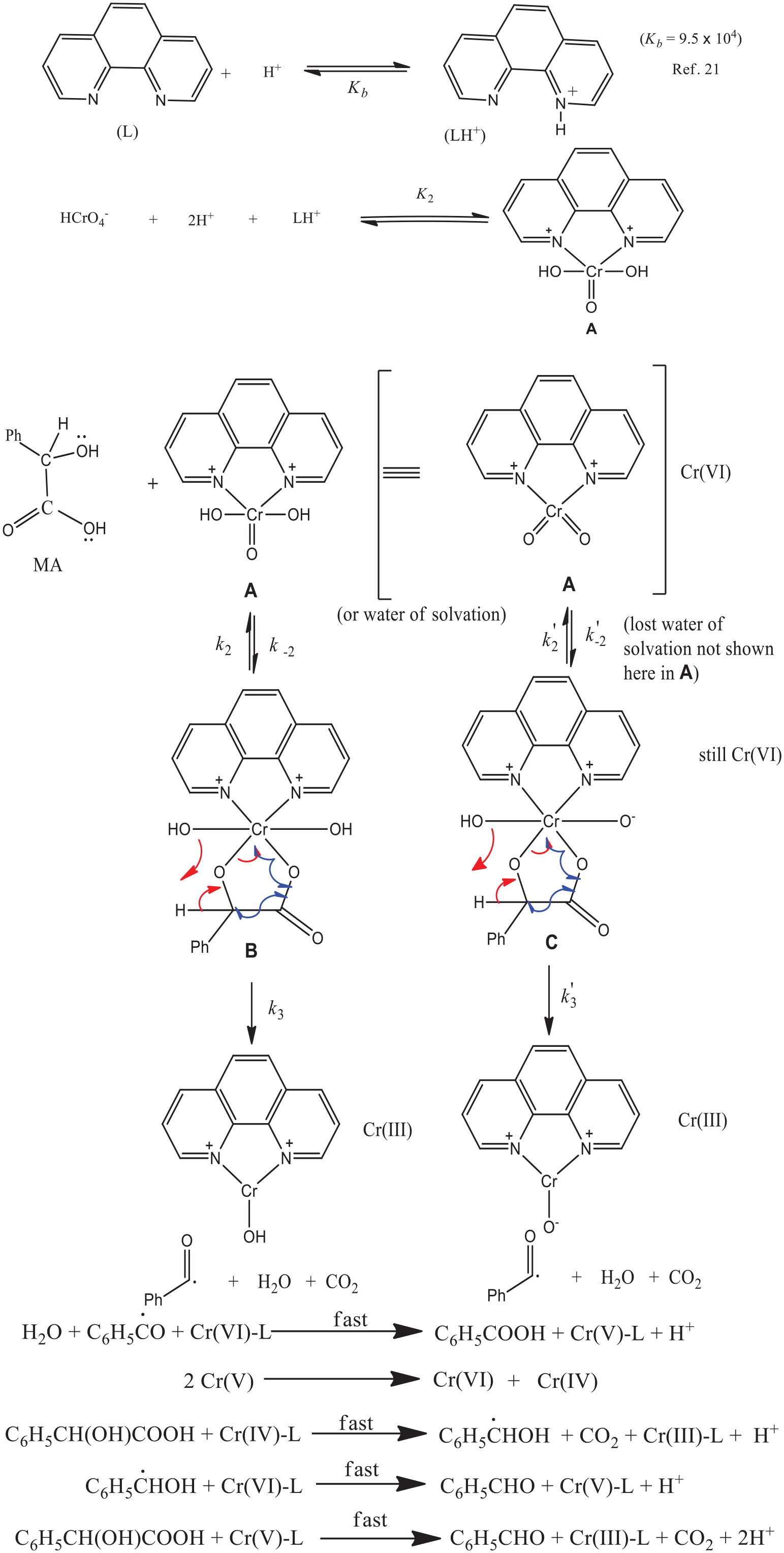
Oxidation of
The observed rate law is
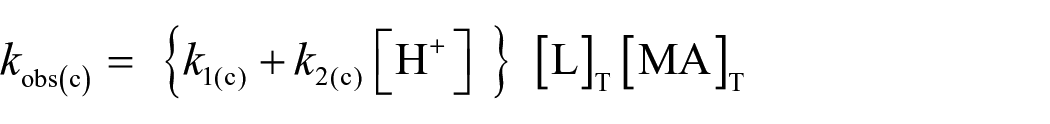
Effect of CPC
Catalysed path: For N-cetylpyridinum chloride (CPC), a representative cationic surfactant, the plot of kobs(c) versus [CPC]T (Figure 6) shows a steady decrease and eventually levels off at higher concentration of CPC. This observation is similar to that observed by Bunton and Cerichelli
23
in the oxidation of ferrocene by ferric salts of the presence of the cationic surfactant cetyltrimethyl ammonium bromide (CTAB). Similar observations have also been noted by Panigrahi and Sahu
24
in the oxidation of acetophenone by Ce(IV) in the presence of N-dodecylpyridinium chloride (NDPC), by Sarada and Reddy,
25
in the oxalic acid–catalysed oxidation of aromatic azo-compounds by Cr(VI) in the presence of SDS, and by us in the chromic acid oxidation of

1[Cr(VI)]T = 6 × 10−4 mol dm−3, [1,10-phen] = 12 × 10−4 mol dm−3, [H2SO4] = 0.25 mol dm−3, [
The neutral substrate can be partitioned in the Stern layer of the micellar phase due to favourable H-bonding and ion–dipole interactions.
1
In the Phen-catalysed pathway, the positively charged micellar head groups of CPC electrostatically restrict the positively charged Cr(VI)-Phen complex

Distribution of the reactive species between the aqueous and micellar phases.
Conclusion
In the Phen-assisted path, a Cr(VI)-Phen complex, a cationic species has been found to act as the active oxidant. In the Phen-assisted path, the Cr(VI)-Phen complex undergoes a nucleophilic attack by the
Experimental and methods
Materials and reagents
1,10-Phenanthroline (Qualigens) was used after repeated crystallization from methanol (m.p. 136 °C).
Procedure and kinetic measurements
T = 30 °C, [S]T >> [Cr(VI)]T and [Phen]T >> [Cr(VI)]T, acid and other necessary chemicals were separately thermostated (±0.1 °C). Progress of the reactions was monitored by following the rate of disappearance of Cr(VI) by using the titrimetric quenching technique as discussed earlier. 8 The pseudo first-order rate constants (kobs) were calculated as usual. Errors associated with the different rate constants and activation parameters were estimated. 28
Product analysis and stoichiometry
Product analysis was carried out by using the 2,4-dinitrophenylhydrazine (DNP) test. 29 The solution of the reaction product was treated with an excess of a saturated solution of DNP in dilute hydrochloric acid. The precipitated 2,4-dinitrophenylhydrazone was filtered off, dried and recrystallized from ethanol. The melting point of the DNP derivative (230 °C) was found to be lower than the melting point (239 °C) of the DNP derivative of authentic benzaldehyde. This slight lowering of the m.p. was due to the presence of the DNP derivative of phenylglyoxylic acid produced as a by-product in a small amount. Thus, the product analysis agreed with the reports of Dominic and Rocek. 9 Moreover, phenylglyoxylic acid was detected and confirmed by spot tests29,30 using a reagent solution prepared from a 0.3% solution of thiophene in benzene. The product solution was subjected to esterification by treatment with ethyl alcohol and concentrated sulfuric acid. A drop of the esterified solution was placed in a micro test tube and evaporated to dryness in a water bath. The residue was dissolved in three drops of concentrated sulfuric acid and treated with two drops of thiophene solution. A characteristic red colour appeared within 15 min. A similar observation was noted by using an authentic sample of phenylglyoxylic acid.
In the reaction mixture, benzoic acid was detected and confirmed by a spot test. 29 The product was extracted using n-hexane. One drop of the benzene solution was treated with a colourless saturated solution of Rhodamine B and an intense pink colour appeared. This colour intensified when the sample was shaken with an aqueous solution of uranyl acetate salts. The same identification was noted by using an authentic sample of benzoic acid.
Carbon dioxide was detected qualitatively 31 under the kinetic conditions by purging dinitrogen through the reaction solution and passing the effluent gas through a narrow tube containing Ca(OH)2. Thus, the stoichiometry of the reaction is

The reaction mixture solutions were scanned (in the range 350–700 nm) at regular time intervals by using a UV-Vis spectrophotometer (UV-VIS-NIR Scanning Spectrophotometer, UV-1800 PC, Shimadzu) to follow the gradual development of the reaction intermediate and the product spectrophotometrically (Figure 7). The plots indicate the gradual disappearance of the Cr(VI) species and the appearance of a Cr(III) species with an isosbestic point at λ = 525 nm.32,33 The observation of this single isosbestic point indicates the very low concentrations of probable intermediates such as Cr(V) and Cr(IV) under the present experimental conditions. In other words, a gradual decrease of Cr(VI) with a concomitant increase of Cr(III) occurs.

Absorption spectra of the reaction mixture in the presence of a catalyst at regular time intervals (7 min) from 0 to 92 min. Concentrations at the beginning of the reaction, [Cr(VI)]T = 6 × 10−4 mol dm−3, [Phen]T = 24 × 10−4 mol dm−3, [H2SO4] = 0.25 mol dm−3, [
Footnotes
Acknowledgements
We acknowledge the Department of Chemistry, Seth Anandram Jaipuria College, Kolkata, for laboratory facilities. Cooperation from Dinesh Chandra Ghosh (of my Department) is gratefully appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
