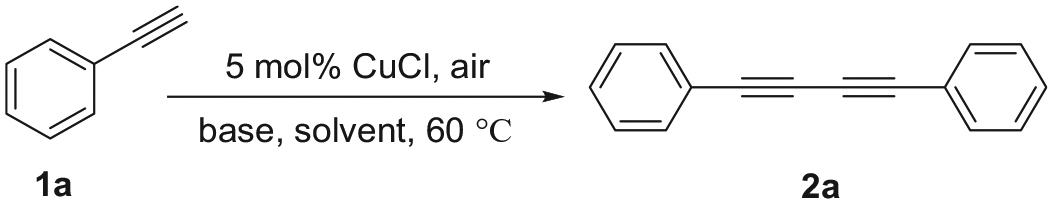
Abstract
A series of symmetrical 1,4-disubstituted buta-1,3-diynes is prepared with excellent yields (up to 95%) through homocoupling of terminal alkynes catalyzed by a copper salt under solvent-free conditions. This method provides an environmentally friendly process to prepare 1,3-diynes in short reaction times under mild conditions. Furthermore, the method is suitable for a wide substrate scope and has excellent functional group compatibility. The reaction can also be scaled up to gram level.
We developed a facile and green method for homocoupling of terminal alkyne. CuCl was found to catalyze the coupling reaction smoothly in the presence of n-propylamine without solvent. In the coupling method, aromatic terminal alkyne, aliphatic terminal alkyne, and heterocyclic terminal alkyne were all applicable (18%–95% yield). The great attractiveness of the current method lies in the simple experimental procedure, mild conditions, easy accessibility of the catalyst, air as environmental friendly oxidant, and a short reaction time. These features make this method an attractive protocol for the synthesis of complex 1,3-diynes, and the reaction can also be scaled up to gram level.

Introduction
Compounds containing polyacetylene structures are widely distributed in nature. They have a broad range of applications and have attracted significant interest from scientists. They can be used in the construction of linear π-conjugated alkyne oligomers and polymers, 1 the manufacture of molecular electronic devices, 2 optical materials, 3 supramolecular switches, 4 and so on. They can also possess strong biological activity, such as antifungal, 5 anti-HIV, 6 antibacterial, 7 and anticancer activities.8,9 In addition, they are widely used to synthesize natural products and their analogs, 10 and various heterocyclic compounds. 11 Hence, 1,3-diynes have a significant research value. The coupling of terminal alkynes represents an important method for the efficient preparation of 1,3-diynes.
Glaser12,13 synthesized 1,4-diphenylbuta-1,3-diyne from phenylacetylene in air with CuCl as the catalyst, ammonia as the base, and ethanol as the solvent. However, the reaction occurred slowly because of the heterogeneous phases and the mixture was liable to explode under dry conditions, which has limited its wide application. A breakthrough in the coupling reaction of alkynes was made by Eglinton and Galbraith in 1956. They14,15 found that the presence of excess Cu(OAc)2 in a solution of methanol and pyridine could effectively promote the coupling reaction of terminal alkynes. This method has been used widely for the construction of macrocyclic alkynes. Hay 16 made an important improvement by adding oxygen into the reaction to raise the yield and shorten the reaction time.
In recent years, great progress has been made in improving this method. Transition-metal catalysts, such as Pd,17–20 Au,21,22 Ag, 23 Ni,24,25 Co,26–28 and Ti, 29 have been employed in oxidative couplings of terminal alkynes instead of the palladium/copper bi-catalyst system. 30 However the Cu-catalyst is still the most attractive. Significant progress was made by Yin et al. 31 in 2011. They developed a facile method for the homocoupling reaction of terminal alkynes with CuCl as catalyst and DMSO as solvent at 90 °C. However, the system required a high reaction temperature and high boiling point solvent. Moodapelly et al. 32 used CuI to synthesize 1,3-dynes from 1,1-dibromoalkenes in the presence of DBU•H2O. However, there are still some disadvantages in the reported methods, such as the use of expensive and toxic palladium catalysts, the requirements of complex ligands with external oxidants, and the harsh reaction conditions.
Chen et al. 33 reported an effective method for the coupling of alkynes using Cu(II) as catalyst and Et3N as base and solvent in air at 60 °C. In their work, CuI did not catalyze the reaction. Furthermore, only a 16% yield of the coupling product was obtained when CuCl was used as catalyst. Herein, we report an environmentally friendly, economic, and efficient method for the rapid homocoupling of terminal alkynes using Cu(I) in air under mild and solvent-free conditions. The general applicability of this protocol has been highlighted by the synthesis of 19 symmetrical diynes. Furthermore, the process can also be carried out successfully at room temperature.
Results and discussion
The reaction conditions for the homocoupling of phenylacetylene (
Base and solvent screening for the homocoupling of

Reactions were performed with phenylacetylene (
Yield by GC.
I2 (5 mol%) was added.
The molar ratio of base to phenylacetylene.
The effect of the temperature and reaction time on the homocoupling reaction was also investigated under solvent-free conditions. The results are shown in Table 2, revealing that the highest yield (88%) was obtained at 60 °C over 3 h under solvent-free conditions (Table 2, entry 4). Overall, the temperature had little impact on the reaction yield (Table 2, entries 1–5). In addition, it was found that the presence of air was essential (Table 2, entry 10). CuI could be used (Table 2, entry 11).
Optimization of the copper-catalyzed homocoupling of phenylacetylene. a

Reactions were performed with phenylacetylene (
Yield by GC.
The reaction was carried out under a nitrogen atmosphere.
CuI was used instead of CuCl.
The homocoupling reaction of a variety of substrates was investigated under the optimum reaction conditions in order to access the substrate scope, the limitations, and the functional group compatibility (Table 3). As shown in Table 3, the yields of the coupling reactions of aromatic terminal alkynes were much higher than those of aliphatic terminal alkynes. Electron-donating and electron-withdrawing substituents on the phenyl of phenylacetylenes were well tolerated under the identified conditions to afford the desired products in 70%–95% yields. However, the homocouplings of 4-chlorophenylacetylene (
Substrate scope of the homocoupling of terminal alkynes. a

Homocoupling was performed with substrates
Isolated yield.
The reaction was carried out in THF (5 mL).
A mixture of terminal alkyne, 5 mol% of CuCl, and an equivalent of n-propylamine was stirred at 60 °C in air under solvent-free conditions to afford 1,3-diynes in good yields (up to 95%). When the reaction was conducted on a scale of 3 g of phenylacetylene, a high yield of 88% was maintained, thus showing that the method was able to scale-up.
Proposed mechanism
Based on the literature12,32,34,35 and the results from our investigations, a plausible mechanism for the homocoupling of terminal alkynes is depicted in Figure 1. Cu(I) or Cu(0) is first oxidized to Cu(II), which then reacted with the terminal alkyne to form a copper(II)-alkyne complex through π-bonding. The alkynyl-copper(II) complex is formed by deprotonation of the terminal alkyne with n-propylamine. The 1,3-diyne was formed and removed from metal, and Cu(II) is reduced to Cu(0) for the next catalytic cycle. To verify the proposed mechanism, a model reaction was carried out under a nitrogen atmosphere, and no desired product was obtained. However, bubbling of oxygen into the mixture could considerably accelerate the reaction. In addition, when 1.5 equiv. of CuCl2 were used instead of CuCl in the coupling of phenylacetylene under an inert atmosphere,

Proposed mechanism for the homocoupling of terminal alkynes.
Conclusion
In summary, we have developed a simple and green method for the homocoupling of terminal alkynes. The reaction proved to be tolerant of a variety of functional groups. CuCl can smoothly catalyze the coupling reaction in air in the presence of n-propylamine under solvent-free conditions. The advantages of the current method consist of the simple experimental procedure, the mild conditions, the easy accessibility of the catalyst, air is used as an environmental friendly oxidant, and the reaction times are short. These features make the method an attractive protocol for the synthesis of 1,3-diynes, while the reaction could also be scaled up to gram level.
Experimental
All reactions were carried out using oven-dried glassware and magnetic stirring under air unless otherwise stated. Reaction temperatures are reported as the temperature of the bath surrounding the vessel. Analytical thin-layer chromatography was performed on silica gel aluminum plates with F-254 indicator produced by the Yantai Chemical Technology Research Institute, and the samples were visualized by UV light (254 nm) or chemical staining with KMnO4 solution. Column chromatography was performed using 200–300 mesh silica gel produced by the Qingdao Hailang Silica Gel Desiccant Co., Ltd. Melting points were determined via the YRT-3 Microscopic Melting Point Apparatus with a capillary method and are uncorrected. NMR spectra were recorded on a Bruker Avance III 400 MHz spectrometer operating at 400 MHz for 1H NMR and 101 MHz for 13C NMR in deuterated solvent. The chemical shifts (δ) are quoted in ppm relative to TMS and coupling constants (J) are quoted in Hz. The following abbreviations were used to show the multiplicities: s: singlet, d: doublet, t: triplet, q: quart, dd: doublet of doublets, tt: triplet of triplets, qt: quart of triplets, tq: triplet of quartlets, and m: multiplet. All coupling constants are determined by analysis using MestReNova v 6.1.0 software. ESI–MS spectra were recorded on a Bruker Esquire 3000 instrument. High-resolution mass spectra (HRMS) were obtained on a MicrOTOF-Q II mass spectrometer with an ESI source (Waters, Manchester, UK).
General procedure 1: synthesis of symmetrical substituted buta-1,3-diynes (2a–s)
The terminal alkyne
Procedure for the preparation of diynes 2l and 2o
4-Chlorophenylacetylene (
1,4-Diphenylbuta-1,3-diyne (
1,4-Di-p-tolylbuta-1,3-diyne (
1,4-Bis(4-ethylphenyl)buta-1,3-diyne (
1,4-Bis(4-pentylphenyl) butane-1,3-diyne (
1,4-Bis(4-methoxyphenyl)buta-1,3-diyne (
1,4-Bis(4-(tert-butyl)phenyl)buta-1,3-diyne (
1,4-Bis(2-fluorophenyl)buta-1,3-diyne (
1,4-Bis(4-fluorophenyl)buta-1,3-diyne (
1,4-Bis(3,5-difluorophenyl)buta-1,3-diyne (
1,4-Bis(2-chlorophenyl)buta-1,3-diyne (
1,4-Bis(3-chlorophenyl)buta-1,3-diyne (
1,4-Bis(4-chlorophenyl)buta-1,3-diyne (
1,4-Bis(2-bromophenyl)buta-1,3-diyne (
1,4-Bis(3-bromophenyl)buta-1,3-diyne (
1,4-Bis(4-bromophenyl)buta-1,3-diyne (
1,4-Bis(3-nitrophenyl)buta-1,3-diyne (
1,4-Bis(2-thienyl)-1,3-butadiyne (
1,4-Di(naphthalen-2-yl)buta-1,3-diyne (
1,6-Diphenyl-2,4-hexadiyne (
Supplemental Material
sj-pdf-1-chl-10.1177_17475198211032580 – Supplemental material for Homocoupling of terminal alkynes catalyzed by CuCl under solvent-free conditions
Supplemental material, sj-pdf-1-chl-10.1177_17475198211032580 for Homocoupling of terminal alkynes catalyzed by CuCl under solvent-free conditions by Yan Xiao, Jiyu Gao, Peng Chen, Guangliang Chen, Zicheng Li and Wencai Huang in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the State Key Laboratory of Biotherapy, West China Hospital, Sichuan University for recording 1H NMR, 13C NMR, and HRMS. The authors also thank the Engineering Teaching Experiment Center, School of Chemical Engineering for recording the melting points.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.