Abstract
A highly sensitive method for the determination of ultra-trace levels of palladium in water samples by cloud point extraction and graphite furnace atomic absorption spectrometry is developed. The procedure is based on complexation of palladium with a laboratory-prepared novel chelating agent, 2-(5-bromo-4-methyl-2-pyridylazo)-5-dimethylaminoaniline (5-Br-4-CH3-PADMA) and subsequent micelle-mediated extraction of the product using the non-ionic surfactant octylphenoxypolyethoxyethanol (Triton X-114) as an extracting agent. Analytical parameters affecting the separation and detection process, such as pH, concentration of the chelating agent and surfactant, equilibration temperature, and time are investigated. The optimized conditions are as follows: pH 6.0 HAc–NaAc buffer solution, 1 × 10−5 mol L−1 5-Br-4-CH3-PADMA, and 0.1% (w/v) Triton X-114. Under the optimized conditions, the calibration graph is linear in the range of 0.1–12 ng/mL, the detection limit is 0.05 ng/mL for palladium, and the relative standard deviation is 2.9% (c = 1.0 ng/mL, n = 10). The enrichment factor, defined as the ratio of the aqueous solution volume to that of the surfactant-rich phase volume after dilution with HNO3–methanol solution, is 200. The proposed method is applied to the determination of palladium in water samples with satisfactory results.
Keywords
A novel o-amino pyridylazo reagent, 2-(5-bromo-4-methyl-2-pyridylazo)-5-dimethylaminoaniline (5-Br-4-CH3-PADMA) is used as the ligand for the determination of ultra-trace levels of palladium in water samples by cloud point extraction and graphite furnace atomic absorption spectrometry. The method offers many advantages such as simplicity, ease of operation, high preconcentration factor, and low limit of detection.
Introduction
Due to its unique physical and chemical properties, palladium has been widely used as a catalyst in the chemical industry, as microcontactors in the electronics industry, as hard alloys in the dentistry, and as an active component in the three-way catalysts in automotive catalytic converters. Since the introduction of catalytic converters into modern automobiles, more than 50% of the world production of palladium is consumed in the production of autocatalysts. 1 As a result, more and more palladium is being released into the environment during the operation of converters.1–3 It has been reported that anthropogenic palladium is mobile and bio-accumulated by aquatic organisms, generally, to a larger extent than Pt and Rh 2 , which can cause asthma, allergies, rhinoconjunctivitis, and other serious health problems. 4 Therefore, an efficient and reliable analytical technique to assess and study the impact of palladium on human health and the ecosystem is required. However, the determination of trace palladium in environmental and biological samples is very difficult owing to its extremely low concentration and the complexity of the matrix. 1 A highly sensitive analytical technique combined with preconcentration and a separation step is often required. 2
Different kinds of sensitive analytical techniques, such as neutron activation analysis, 5 graphite furnace atomic absorption spectrometry (GFAAS),6,7 inductively coupled plasma-atomic emission spectrometry (ICP-AES),8–10 and inductively coupled plasma-mass spectrometry (ICP-MS),11–13 have been developed for the determination of palladium at low concentrations. Among these methods, GFAAS has been employed here because it is a well-established and cost-effective technique with excellent sensitivity, requiring only small volumes of sample, and the instrumentation is reality available.
A variety of procedures have been developed for the separation and preconcentration of trace palladium ions, including liquid−liquid extraction (LLE), 14 dispersive liquid−liquid microextraction (DLLME),15–17 ion-exchange,18,19 solid-phase extraction,20–22 and cloud point extraction (CPE).23,24 Among them, CPE, based on surfactant-mediated phase separation, has emerged in the last decade as a very attractive and efficient technique for the separation and preconcentration. CPE has the advantages of simple operation, convenience, low-cost, high preconcentration factors, and environmental safety, and is considered to be a green extraction technique. 25
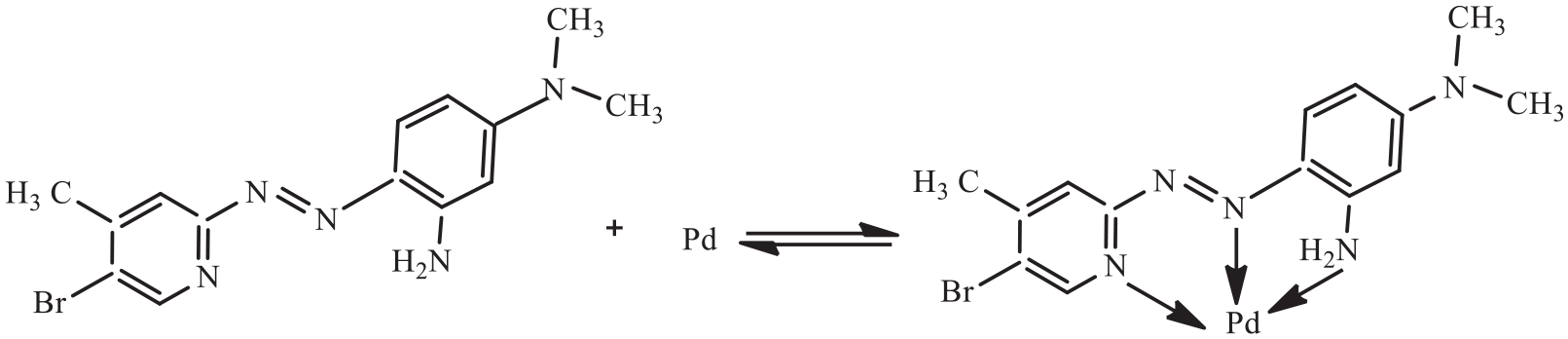
The aim of this work is to combine CPE, as a highly efficient technique for separation and preconcentration, with GFAAS, as a highly sensitive detection method, and to develop a simple and high efficiency method for the determination of trace palladium in water samples. In the proposed method, a laboratory-prepared novel o-amino pyridylazo reagent, 2-(5-bromo-4-methyl-2-pyridylazo)-5-dimethylaminoaniline (5-Br-4-CH3-PADMA) is used as the ligand as it was found to be a good chromogenic reagent for spectrophotometric determination of palladium. 26 The structures of the reagent and its palladium(II) complex are shown in Scheme 1. Triton X-114 was selected as the extractant because it has a convenient cloud point (in the range of 23–25 °C). The main factors affecting CPE and the sensitivity of the method were investigated in detail. The developed method has been applied to the determination of ultra-trace palladium in water samples with satisfactory results.
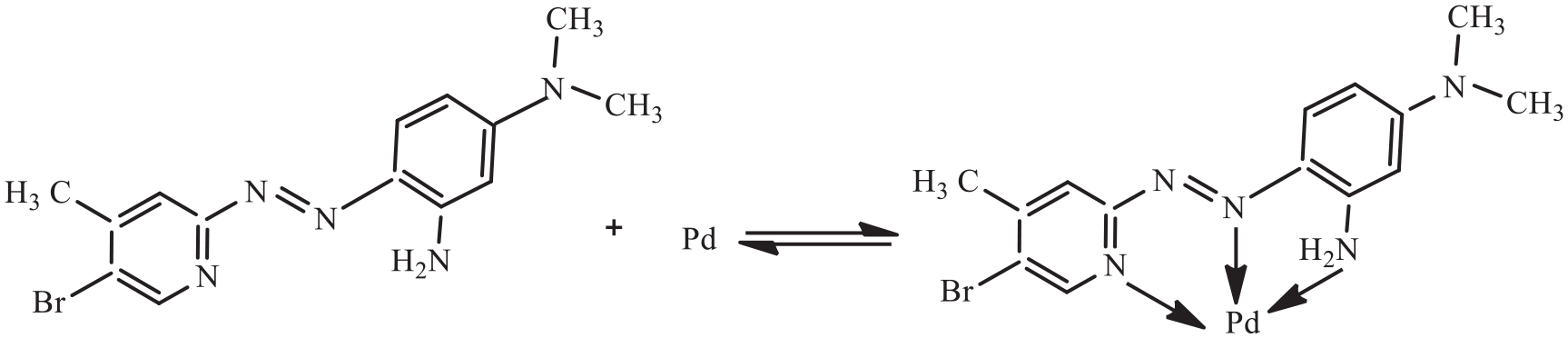
Chemical structure of 5-Br-4-CH3-PADMA and its palladium chelate.
Results and discussion
Effect of pH
The separation and preconcentration of metal ions by CPE involves prior formation of a hydrophobic chelate to be extracted into a small volume of surfactant-rich phase. As shown in Scheme 1, the chelating agent 5-Br-CH3-PADMA and its palladium chelate contain nitrogen atoms which can be protonated. Therefore, the pH of the solution plays an important role in the formation and hydrophobicity of the chelate, as well as subsequent extraction. The effect of pH on the CPE efficiency of palladium was investigated within the pH range of 3.5–8.0 using HAc–NaAc buffer solution (Figure 1). As can be seen, the optimum pH is in the range of 5.5–8.0. At lower pH values, the hydrophobicity of the chelate decreases, probably due to protonation, and hence, the extraction efficiency of palladium is low. Thus, pH 6.0 was chosen for subsequent experiments.

Effect of pH on the absorbance.
Effect of the 5-Br-CH3-PADMA concentration
The new chelating agent 5-Br-CH3-PADMA was employed to form a hydrophobic chelate with palladium. The effect of the 5-Br-CH3-PADMA concentration on the determination of palladium was studied in the range of 2.0 × 10−6–1.6 × 10−5 mol L−1 of 5-Br-CH3-PADMA, and the results are shown in Figure 2. As can be seen, the absorbance increased rapidly on increasing the concentration of 5-Br-CH3-PADMA from 2.0 × 10−6 to 8.0 × 10−6 mol L−1, and thereafter retained almost constant. A 5-Br-CH3-PADMA concentration of 1.0 × 10−5 mol L−1 was used for subsequent experiments.
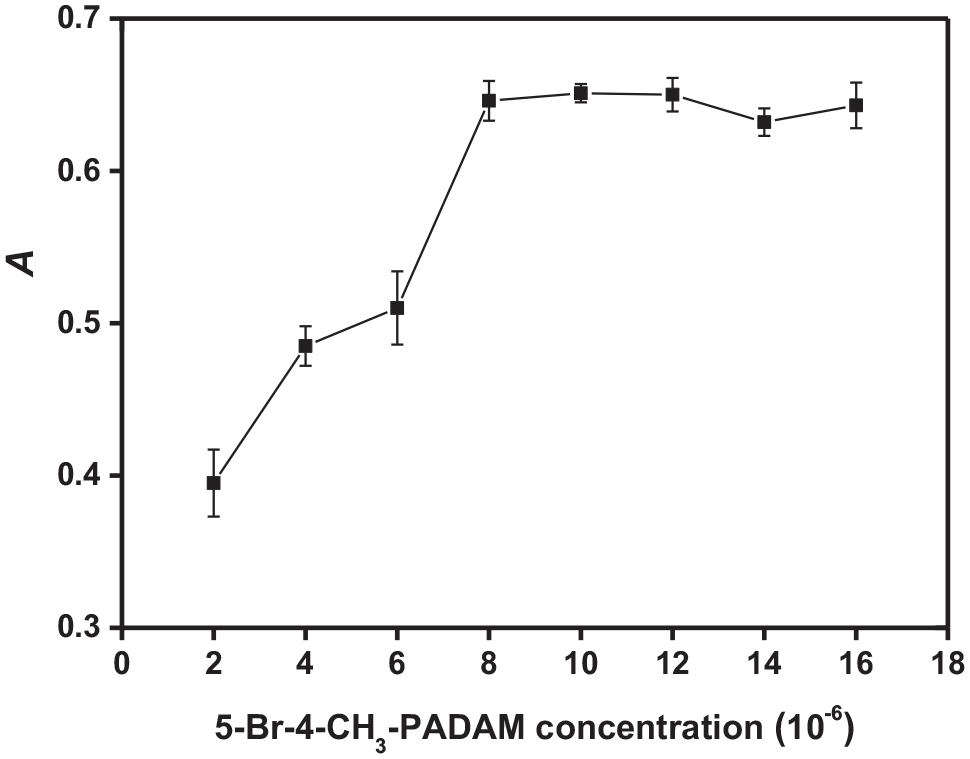
Effect of the concentration of 5-Br-4-CH3-PADMA on the absorbance.
Effect of the Triton X-114 concentration
To date, surfactants such as SDS, CTAB, PONPE 7.5, OP-7, OP-10, Triton X-100, and Triton X-114 have been used for CPE metal analysis.24,25 Triton X-114 was employed for the formation of the surfactant-rich phase due to its low cloud point temperature (CPT) (25 °C), commercial availability in a highly purified homogeneous form, the high density of the surfactant-rich phase which facilitates phase separation by centrifugation, and the low toxicity and low cost. The effect of the surfactant concentration on the absorbance in the range of 0.04%−0.2% (w/v) was investigated. As can be seen from Figure 3, the absorbance significantly increases when the Triton X-114 concentration increased from 0.02% to 0.08% (w/v). The optimum Triton X-114 concentration ranges from 0.08% to 0.14% (w/v). However, further increasing the concentration of Triton X-114 leads to a gradual decrease in the absorbance. A possible explanation might due to the increment in the volume of the surfactant-rich phase. 24 Therefore, 0.1% of Triton X-114 was selected for subsequent experiments.
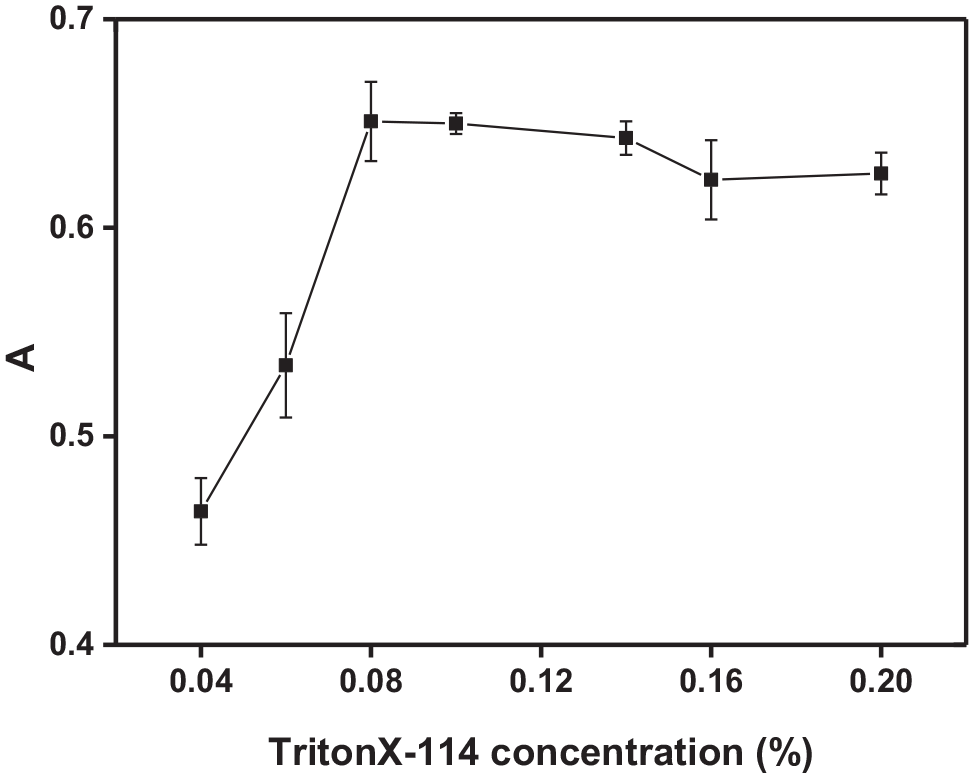
Effect of Triton X-114 on the absorbance.
Effect of the equilibration temperature and time
An optimum equilibration temperature and incubation time are necessary to achieve easy phase separation and an efficient as possible preconcentration. The maximum analyte preconcentration factor is expected when CPE is performed at equilibration temperatures well above the CPT of the surfactant. When the temperature increases, dehydration occurs and results in a decreased volume of the surfactant-rich phase, hereby increasing the phase volume ratio. The effect of the equilibration temperature and time was investigated in the range of 40−90 °C and for 5–30 min, respectively. The results are shown in Figures 4 and 5. As can be seen, the maximum absorbance and good phase separation were achieved when the equilibrium temperature was over 50 °C. Hence, an equilibration temperature of 60 °C was used throughout. It was also found that when the temperature was fixed at 60 °C, the absorbance became almost constant after 10 min. Therefore, an equilibration time of 10 min was employed in further studies.
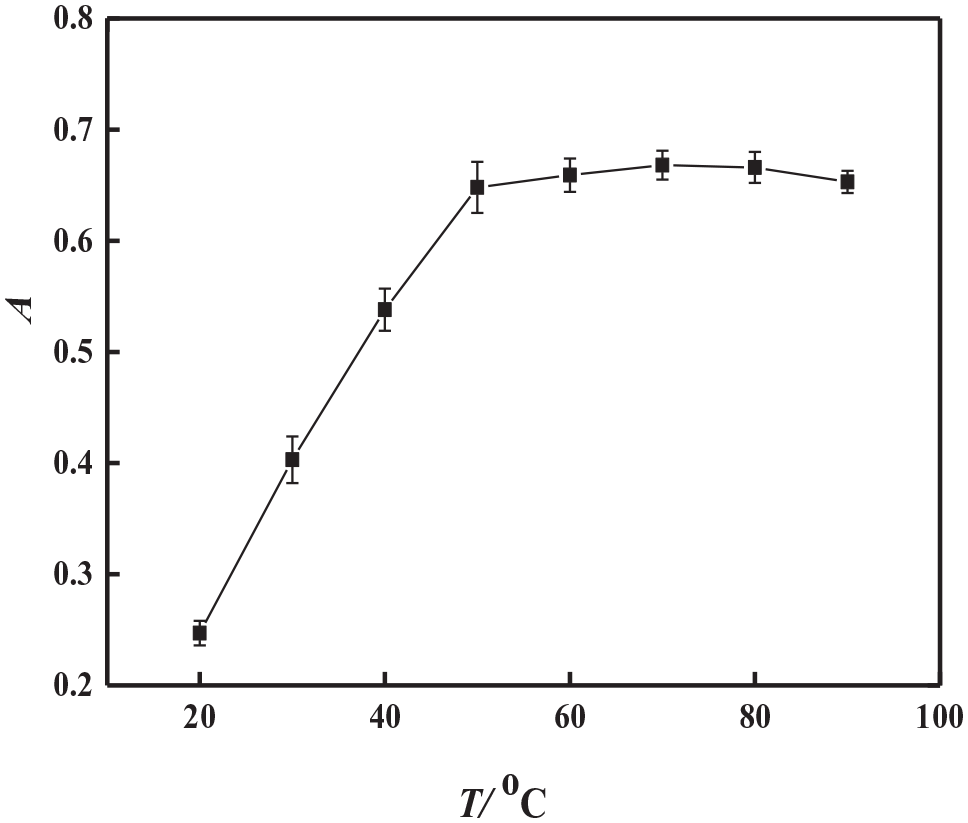
Effect of the equilibrium temperature on the absorbance.
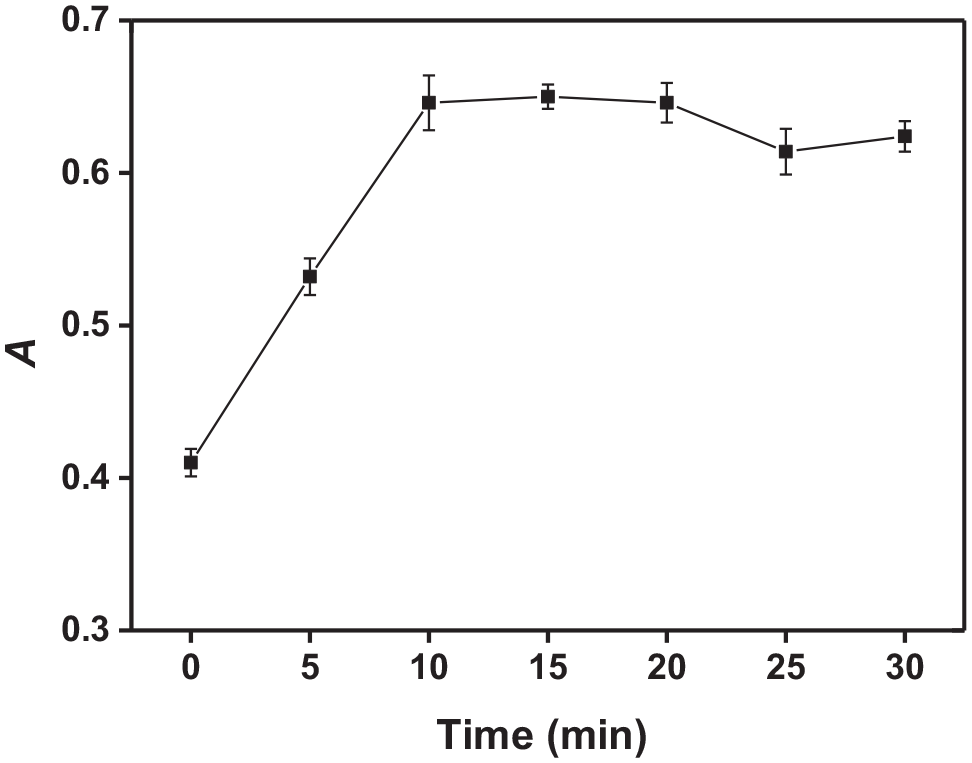
Effect of the equilibrium time on the absorbance.
Matrix interference studies
The effect of foreign species on the determination of 8 ng/mL palladium(II) by the proposed method was investigated. The tolerance limits of various species have been taken as the value that caused relative error of not more than ±5% in the absorbance. The results of the matrix interference study are summarized in Table 1.
Effect of foreign species on the preconcentration/determination of palladium.
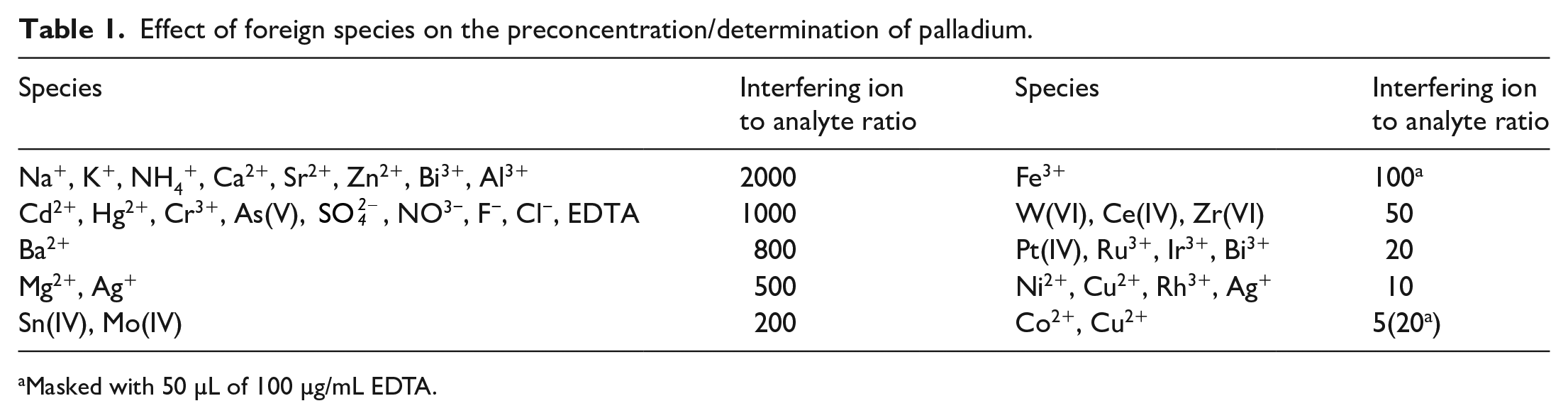
Masked with 50 µL of 100 µg/mL EDTA.
Analytical figures of merit
Using the proposed method and under the optimized experimental conditions, the calibration graph is linear over a range of 0.2–12.0 ng/mL Pd(II). The linear equation is Aint = 0.0590 + 0.0738 c (ng/mL), and the correlation coefficient is R = 0.9954. The detection limit (LOD), calculated as three times the standard deviation of 11 duplicate measurements of the blank solution, is 0.05 ng/mL. The relative standard deviation (RSD) for 10 samples of 1.0 ng/mL of Pd(II) subjected to the CPE procedure was 3.7%. The enrichment factor (EF), defined as the ratio of the aqueous solution volume (10 mL) to that of the surfactant-rich phase volume after dilution with HNO3–methanol solution (50 µL), was 200 (Table 2).
Comparison of published methods with the proposed method for CPE of palladium.

TMK: 4,4’-Bis(dimethylamino)thiobenzophenone; NTACA: 4-(2-naphthalenyl)thiozol-2-yl-azo chromotropic acid; PMBP: 1-phenyl-3-methyl-4-benzoyl-5-pyrazolone; DMG: dimethylglyoxime; PAN: 1-(2-pyridylazo)-2-naphthol; BIMPI: 2-((2-((1H-benzo[d]imidazole-2-yl)methoxy)phenoxy)methyl)-1H-benzo[d]imidazol; APDC: ammonium pyrolysine dithiocarbamate; BIES: bis((1H-benzo[d]imidazol-2-yl)ethyl)sulfane; 5-Br-PADAP: 2-(5-bromo-2-pyridylazo)-5-diethylaminophenol; BMPH; benzil mono-(2-pyridyl)hydrazone; DDTP: O,O-diethyldithiophosphate; DBHA: 1,8-diamino-4,5-bis(hydroxyamino)anthraquinone reagent; 2-MBT: 2-mercaptobenzothiazole; 3,5-diCl-PADMA: 2-(3,5-dichloro-2-pyridylazo)-5-dimethylaminoaniline; SP: spectrophotometry; FAAS: flame atomic absorption spectrometry; FJ-FAAS: flow injection flame atomic absorption spectrometry; ETAAS: electrothermal atomic absorption spectrometry; GFAAS: graphite furnace atomic absorption spectrometry; CZE-DAD: capillary zone electrophoresis coupled with a diode array detector; ICP-AES: inductively coupled plasma-atomic emission spectrometry; ICP-MS: inductively coupled plasma-mass spectrometry; TLS: thermal lens spectrometry.
Application of real water samples
In order to assess the reliability of the proposed method, the optimized procedure was applied for the determination of palladium in tap water, well water, reservoir water, and river water samples. For this purpose, 5.0 mL of each water sample was taken in a 10-mL graduated conical centrifuge tube, and 50 µL 100 μg/mL of EDTA solution was added. The palladium content was determined according to the proposed procedure. The results are given in Table 3. As shown in the table, the recoveries for the spiked samples are within the range of 95.8%~104.0%, and the RSD is below 4%.
Determination of the palladium content in environmental water samples.

LOD: detection limit; RSD: relative standard deviation.
All the water samples and their analytical results were provided by Xi’an Hydrographic Bureau, Xi’an, Shaanxi Province, P.R. China.
Mean value of six experiments ±standard deviation.
pH = 7.05, G = 1204 µS/cm, Chemical Oxygen Consumption (COD) = 28.3, ρ(
pH = 7.28, G = 1226 µS/cm, COD = 32.2, ρ(
Collected from the mouth of the North Luo River (Weinan, Shaanxi, China) to the Wei River. pH = 7.34, G = 1680 µS/cm, COD = 29.1, ρ(
Collected from the mouth of the Jing River (Xi’an, China) to the Wei River. pH = 7.15, G = 2220 µS/cm, COD = 39.8, ρ(
Conclusion
In this study, we have developed a novel method for the determination of ultra-trace palladium by CPE combined with GFAAS using a new synthesized reagent, 5-Br-CH3-PADMA, as a chelating reagent, and the non-ionic surfactant Triton X-114 as the extractant. This combination is favorable because CPE is a simple, convenient, inexpensive, and eco-friendly technique for preconcentration and separation of trace metals from aqueous solutions to a very small volume of surfactant-rich phase. In addition, GFAAS, a well-established technique with excellent sensitivity, is suitable for analysis of low volumes of the remaining phase after CPE. The developed method offers many advantages such as simplicity, ease of operation, high preconcentration factor, and low limit of detection. It provides satisfactory results when applied to the determination of palladium in real water samples.
Experimental
Instrumentation
A TAS-990 atomic absorption spectrometer (Beijing Purkinje General Instrument Co. Ltd., Beijing, China), equipped with a deuterium arc background corrector, a GFH-990 graphite furnace atomizer system (side-heated), and an ASC-990 autosampler, was employed throughout. A palladium hollow cathode lamp (Beijing ShuGuang-Ming Electronic Lighting Instrument Co., Ltd., Beijing, China) was used as the radiation source. Argon (99.999%) was used as the purge and protective gas. All measurements were performed using integrated absorbance (peak area, Aint). Preliminary experiments indicated that the instrumental parameters and temperature program for the graphite atomizer suggested by the manufacturer could be efficient in this method. The operating parameters recommended by the manufacturer are listed in Table 4. All pH measurements were performed with a PB-10 digital pH meter (Sartoris Scientific Instrument Co. Ltd., Beijing, China) supplied with a combined glass electrode. An HH-2 thermostatic bath (Kewei Yongxing Instrument Co. Ltd., Beijing, China), maintained at the desired temperature, was used for CPT experiments. An 800-1 centrifuge (Pudong Physical Instruments Factory, Shanghai, China) was employed to accelerate phase separation.
Instrumental parameters and the temperature program for GFAAS.

Reagents and solutions
All chemicals used were of analytical grade unless otherwise mentioned. All solutions were prepared with ultrapure water (⩽0.055 μS/cm) obtained from a Simplicity 185 (Millipore Company, USA) water purification system. A 1000-mg L−1 Pd(II) standard stock solution was prepared by dissolving 0.1000 g of spectrographically pure palladium in 50 mL of aqua regia, adding 1 mL of 20% NaCl, heating almost to dryness, then adding 2 mL of concentrated HCl, evaporating to nearly dryness, dissolving the residue in 6 mol L−1 HCl, and diluting to 100 mL with 6 mol L−1 HCl. The working standard solutions were obtained by appropriate step-by-step dilution of this solution with water. A 1.0 × 10−3 mol L−1 5-Br-CH3-PADMA (laboratory-synthesized) 26 solution was prepared by dissolving 0.0334 g of 5-Br-4-CH3-PADMA in 1000 mL of ethanol. 1% (w/v) Triton X-114 (Sigma-Aldrich, Milwaukee, USA) solution was prepared by dissolving 1.0 g of Triton X-114 in 100 mL of water. A buffer solution of pH 6.0 was prepared by mixing 0.25 mol L−1 acetic acid (HAc) and 0.25 mol L−1 sodium acetate (NaAc) solutions at appropriate ratios and correcting using a pH meter. A 0.1-mol L−1 HNO3–methanol solution was prepared by mixing of 0.2 of mol L−1 HNO3 and methanol in equal volumes.
Procedure
An aliquot of working standard palladium or sample solution, 2.0 mL of acetate buffer solution (pH = 6.0), and 100 μL of 1.0 × 10−3 mol L−1 5-Br-CH3-PADMA solution were transferred into a 10-mL graduated conical centrifuge tube. For the formation of the palladium complex, the mixture was heated in a boiling water bath for 5 min. Afterwards, for cloud point extraction, 1.0 mL of 1% (w/v) Triton X-114 solution was added and made up to 10 mL with water. The resulting solution was shaken and kept in a thermostated bath at 60 °C for 10 min. Separation of the two phases was then accelerated by centrifugation at 3500 r/min for 5 min. After cooling in an ice-bath for 2 min, the surfactant-rich phase became viscous and was retained at the bottom of the tube. The aqueous phase was easily decanted by inverting the tube. The surfactant-rich phase in the tube was dissolved using 50 µL of 0.1 mol L−1 HNO3–methanol solution. The final solution was introduced by autosampler into the furnace for GFAAS determination of the palladium content.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (nos 21545014 and 21445004) and the Science and Technology Plan Project of Xi’an, Shaanxi Province, China (no. 2016CXWL09).
