Abstract
Catalysis using supported gold nanoparticles has attracted significant research interest due to their unique properties and potential that is directly related to their particle size. An efficient one-pot, three-component procedure is developed for the preparation of pyrimido[4,5-b][1,6]naphthyridin-4(1H)-one derivatives (
Keywords

Introduction
1,6-Naphthyridines have received considerable attention because of their wide range of biological activities,1–3 including antitumor, anti-inflammatory, antimicrobial, 4 and anticonvulsant. Both pyrimidine and 1,6-naphthyridine scaffolds have been shown to be important structural motifs in chemistry; therefore, the preparation of pyrimidonaphthyridine derivatives, in which these scaffolds are merged, might provide compounds that exhibit simultaneously the biological properties of each moiety.4–6
Gangjee and co-workers have described the construction of the pyrimidonaphthyridine skeleton via a multistep reaction. 7 Previously, it was reported that pyrimido[4,5-b][1,6]naphthyridine moieties could be synthesized efficiently under microwave conditions. 1 Hence, the continued development of diverse pyrimidonaphthyridine compounds is still in strong demand. Marjani and his team were able to synthesize pyrimidonaphthyridine derivatives in the presence of AgNPs under mild conditions. 8 Some previously published protocols using acid,9–11 basic, 1 or metal catalysts, 12 or catalyst-free protocols under microwave 13 or thermal conditions for the formation of analogs of the target moiety. Unfortunately, all previous methods produce very low yields; therefore, it was our aim to develop new conditions based on using a nanocatalyst hoping to improve the yields. The use of a nanocatalytic system would allow the rapid, and selective chemical transformations coupled with the ease of catalyst separation and recovery. 14 Using a nano-sized catalyst (high surface area), the contact between the reactants and catalyst is increased dramatically (this phenomenon is close to homogeneous catalysis).15,16 The insolubility of the catalyst in the reaction solvent leads to a heterogeneous process, and hence, the catalyst can be separated easily from the reaction mixture (this phenomenon is close to heterogeneous catalysis).17–20 It is known that catalytic properties of metallic NPs are size- and shape-dependent.21–25 It has been reported that gold nanoparticles are stabilized and well dispersed on various supports (metal oxides,26–31 carbon materials,32–40 metal–organic frameworks,41–43 zeolites, 44 modified aluminum,32,44–47 ionic liquids, 48 etc.) and have the ability to catalyze several reactions successfully.
Several reports have demonstrated the wide applications of zeolites as catalysts and adsorbents.49–53 These microporous materials are three-dimensional and crystalline hydrated aluminosilicates, 54 and are highly rigid under dehydration. 55 The important structural, physical, and chemical properties of zeolites, with tailored channels and cavities on the molecular scale, make them versatile and valuable for such broad applications as adsorbents and catalysts in industrial, agricultural, and environmental applications. 56 Moreover, zeolite-nanogold possesses high thermal stability and plays the dual role of stabilizing the nanoparticles against sintering and their distinct pore structure can facilitate shape-selective catalysis. Zeolite nanoshell encapsulating gold nanoparticles has successfully been employed for cyclohexane oxidation, and these catalysts show better conversion with increased reusability. 44
All these properties encouraged us to study the loading of zeolite with nanogold (Figure 1). For use as a catalyst in a simple route to synthesize pyrimido[4,5-b][1,6]naphthyridine via a one-pot reaction between 6-amino-2-thioxo-2,3-dihydropyrimidin-4(1H)-ones (
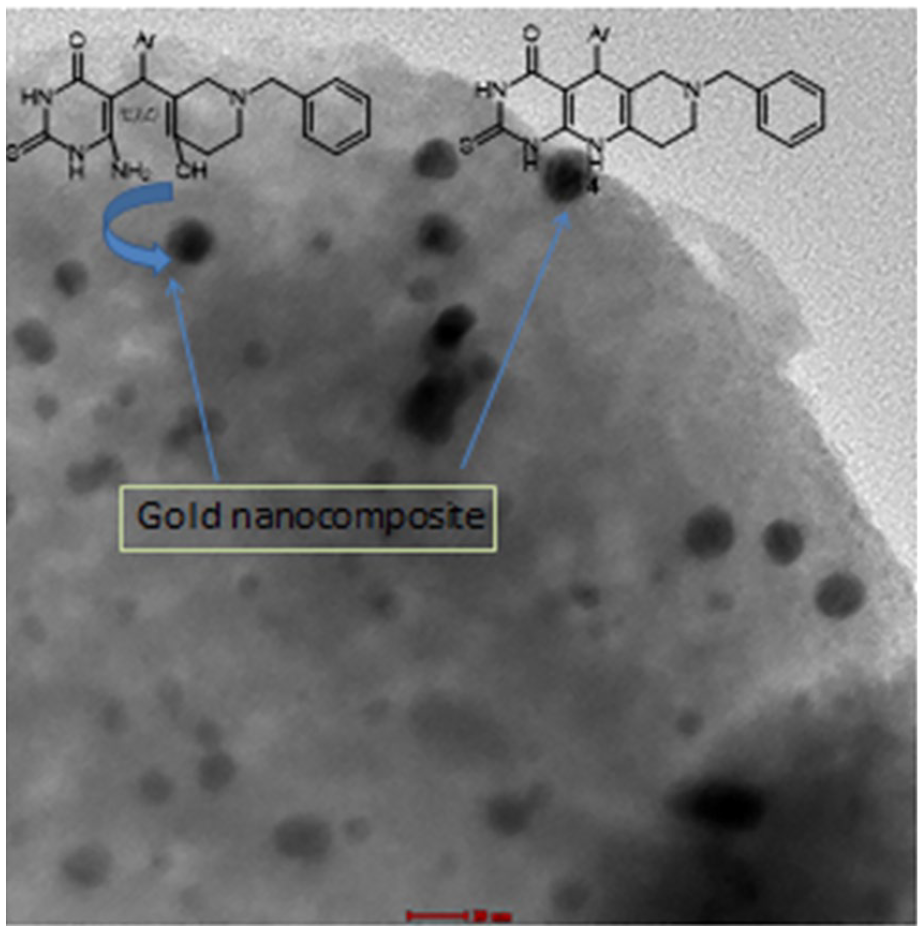
TEM micrograph of zeolite-doped AuNp used as a catalyst for the preparation of pyrimido[4,5-b][1,6]naphthyridines.
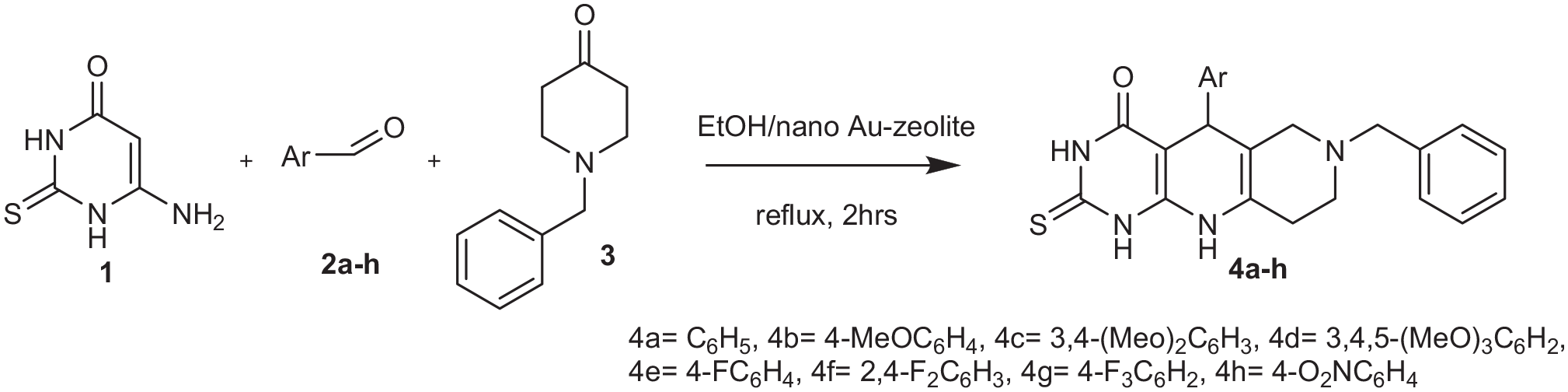
Synthesis of pyrimido-naphthyridinone (
Results and discussion
Physicochemical characterization of the nanocomposite
Low-angle XRD patterns corresponding to the prepared zeolite–Au nanocomposite are shown in Figure 2. The sum of the reflection intensities at 2θ of 15.5°, 24.0°, 28.0°, 32.0°, and 63.0° corresponding to the (450), (309), (207), and (202) planes of a cubic crystal system is also shown in Figure 2 for the prepared nanogold indicating the formation of a cubic crystal of zeolite-nano Au. The intensities of the peaks are relatively high being an indication of high crystallinity. The formed zeolite is a mixture of sodium aluminum silicate and sodium aluminum oxide silicate, which is confirmed from standard data for zeolites.

XRD pattern of the zeolite–gold nanocomposite.
The method used to prepare the nanocomposite in this work produced a uniform dispersion of small particles, around 4–6 nm gold nanoparticles on zeolite, as shown in the TEM image in Figure 3. The formation of small nanoparticles may help the incorporation of gold nanoparticles within the zeolite framework as also indicated by XRD measurements.

TEM micrograph of the zeolite-doped nanogold.
Chemistry
The uracil nucleobase has different tautomeric forms in equilibrium, which is strongly dependent on the interaction of these molecules with their environment. Knowledge regarding this tautomerization in different environments can provide insight into the influence of solvent/catalyst effects on molecular stability. In this work, we have reported the one-pot, three-component condensation reaction of 6-amino-2-thiouracil (
From Table 1, it is very clear that the percentage of the product
Optimization of the reaction conditions for

Isolated yield based on thiouracil.
The formation of the pyrimidonaphthyridine product (
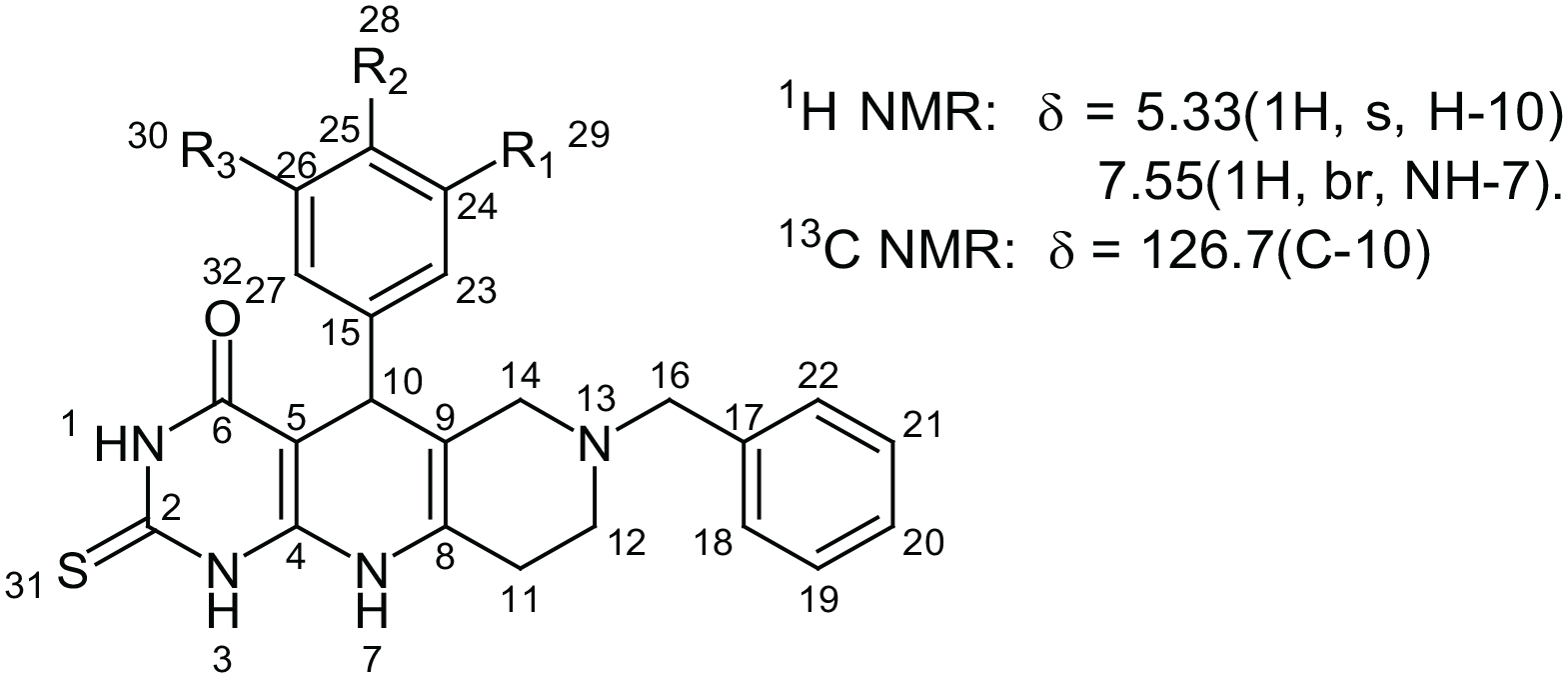
NMR of synthesized compound
Yields of compounds.

Comparative value to uracil.
Molecular docking
Genetic alteration of one or more components of the INK4 CDK4,6/cyclin D-retinoblastoma pathway is found in more than half of all human cancers. Therefore, CDK4 is an attractive target for the development of a novel anticancer agent. Docking studies of compounds

Compound
In order to increase the activity of
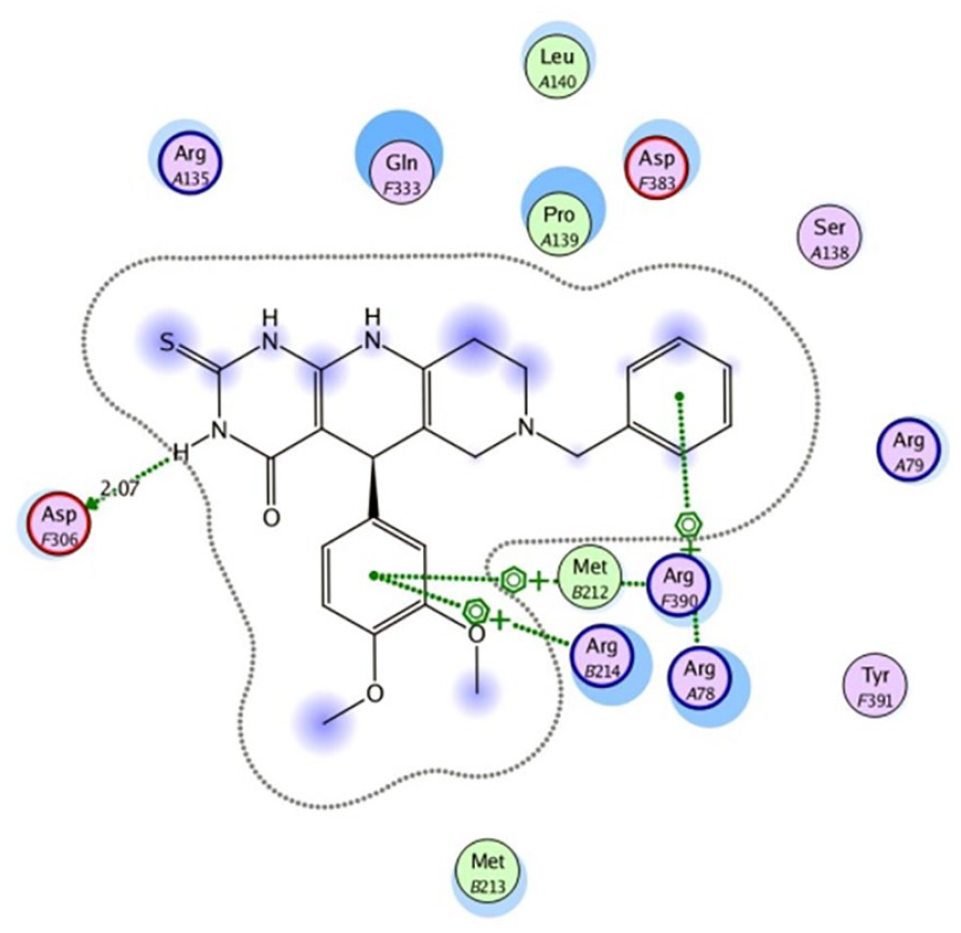
Compound
Cytotoxicity assay
Previously, it has been reported that the type of cancer cell plays a crucial role in antitumor activity of tested compounds. Upon screening our compounds against proliferation of Huh7, an in vitro model of human liver cancer cells, our results showed that the tested compounds showed different anti-proliferative activities against Huh7, with IC50 values ranging from 22.5 to 87 µM. Compounds

Cytotoxic effects of the tested compounds
Conclusion
Novel of pyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (
Experimental
Synthesis of nano Au-zeolite
The growth and ripening of gold nanoparticles were assembled on the surface and cavities of zeolite networks. Zeolite powder (Sigma-Aldrich, USA) was activated by annealing for 5 h at 200 °C in vacuum oven to get rid of humidity and activate the zeolite networks. 50 mL (0.01 M) gold chloride and HAuCl4 (Sigma-Aldrich) solution were mixed with 1 g activated zeolite under stirring at 60 °C for 3 h forming yellowish solution. Heat up the solution till boiling and then add 1 mL of 1% trisodium citrate (Sigma-Aldrich) solution, then left to stir for 5 min till characteristic pink color of gold nanoparticles formed. Centrifuge the solution at 10K r/min for 30 min for precipitating Au–zeolite nanocomposite. The obtained nanocomposite was dried in vacuum oven at 70° for 8 h and stored in desiccator for further characterization. 57
Characterization of nano Au-zeolite
The particle size and the morphology of the prepared catalyst were characterized by high TEM (Philips, The Netherlands). The measuring mode of the sample in the TEM instrument depends on its suspension in water followed by ultrasonication for 600 s in ultra 8050-H Clifton. It was then applied in the TEM instrument on 100 mesh copper grade coated with carbon. Powder XRD patterns were recorded with a PANalytical X’Pert PRO diffractometer using a Cu Kα radiation source for the investigation of the crystalline structure and phase.
Molecular docking
All molecular modeling calculations and docking studies were performed using Molecular Operating Environment (MOE), version 2009.10, Chemical Computing Group. The program was used via the Windows XP operating system installed on an Intel Pentium IV PC with a 2.9 MHz processor and 512 RAM. The prepared compounds were built using the MOE builder interface and subjected to energy minimization using MOPAC. The produced model was subjected to a systematic conformational search, where all items were set as default with a root mean square (RMS) gradient of 0.01 kcal/mol and an RMS distance of 0.1 Å.
Biological assay
Cell culture
The Huh7, in vitro model of human liver cancer cell line, was obtained from ATCC (USA). Cells were cultured in DMEM media (Gibco, USA) supplemented with 10% fetal bovine serum (Gibco), antibiotics (2% penicillin-streptomycin (100 IU/mL)), and 0.5% fungi zone (Gibco). The cells were maintained in monolayer culture at 37 °C under a humidified atmosphere of 5% CO2. The cells were sub-cultured by trypsinization (0.025% trypsin and 0.0025% EDTA; Gibco), and maintained in the Tissue Culture Laboratory at the Virology & Immunology Unit, Cancer Biology Department, National Cancer Institute, Cairo University, Egypt, with cryogenic banking of low-passage cells to maintain uniformity of cell properties through the study. Cell numbers and viabilities were monitored by standard Trypan blue dye exclusion procedures.58,59
Treatment of cells and colorimetric MTT assay
For investigation of the cellular toxicity of all the synthesized compounds against proliferation of Huh7 cells, 8 × 103 cells/well were plated in a 96 tissue culture plate with 10% DMEM. After 24 h, five different twofold dilutions of compounds

where Atest is the absorbance of the test sample and Acontrol is the absorbance of the control sample. The results were the average of three wells and 100% viability was determined from the negative control, that is, untreated cells.

Cell morphology of Huh7: (a) untreated cell control, (b) Huh7 treated with a high concentration of
For each compound concentration, five wells were used (five replicate wells were prepared for each individual dose). The average was calculated. Data are expressed as the percentage of relative viability compared with the untreated cells. The cytotoxicity dose was calculated as a dose induced ≈100% relative on viability.
Chemistry
Melting points were measured on a Gallenkamp electrothermal melting point apparatus and are uncorrected. Infrared (IR) spectra were recorded as KBr disks using a Shimadzu FTIR Prestige 21 spectrophotometer. 1H and 13C NMR spectra were recorded in DMSO-d6 at 300 MHz on a Varian Mercury NMR spectrometer using TMS as the internal standard. Chemical shifts (δ) are reported in parts per million (ppm), and J values are given in hertz. The mass spectra were recorded on a GCeMS-QP1000 EX mass spectrometer at 70 ev. Elemental analyses were carried out at the Micro-analytical Centre of Cairo University, Giza, Egypt.
General procedure for the synthesis of 7-benzyl-5-aryl-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-ones (4 )
6-Amino-2-thioxo-2,3-dihydropyrimidin-4(1H)-one (
7-Benzyl-5-phenyl-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4a )
Colorless powder (89%); m.p. 297–298 °C. IR (KBr) (νmax/cm−1): 3399, 3181, 1607, 1550. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.34 (t, J = 6 Hz, 2H, CH2), 2.51 (t, J = 6 Hz, 2H, CH2), 3.34 (s, 2H, CH2), 3.61 (s, 2H, CH2), 5.33 (s, 1H, CH), 6.37 (brs, 1H, NH), 6.78-7.96 (m, 10H, H-Ar), 11.62 (br, 1H, NH), 12.04 (br, 1H, NH). 13C NMR (75 MHz, DMSO-d6): δ (ppm) 21.7, 31.2, 41.1, 52.7, 61.1, 78.6, 90.7, 125.7, 126.9, 127.5, 128.3, 128.7, 129.0, 129.1, 129.7, 138.4, 153.9, 162.1, 175.0. MS (EI, 70 eV): m/z (%): 403 (M + M+, 25), 402 (M+, 100). Anal. calcd for C23H22N4OS (402.15): C, 68.63; H, 5.51; N, 13.92; found: C, 68.92; H, 5.60; N, 13.99.
7-Benzyl-5-(4-methoxyphenyl)-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4b )
Colorless powder (75%); m.p. 241–243 °C. IR (KBr) (νmax/cm−1): 3349, 3181, 1604, 1555. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.08 (t, J = 6 Hz, 2H, CH2), 2.50 (t, J = 6 Hz, 2H, CH2), 3.43 (s, 2H, CH2), 3.45 (s, 2H, CH2), 3.68 (s, 3H, OCH3), 5.27 (s, 1H, CH), 6.72 (brs, 1H, NH), 6.75-6.94 (m, 9H, H-Ar), 7.95 (s, 2H, NH), 11.50 (s, 1H, NH). MS (EI, 70 eV): m/z (%): 432 (M+, 100). Anal. calcd for C24H24N4O2S (432.16): C, 66.64; H, 5.59; N, 12.95; found: C, 66.62; H, 5.53; N, 12.99.
7-Benzyl-5-(4,5-dimethoxyphenyl)-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4c )
Colorless powder (89%); m.p. 218–220 °C. IR (KBr) (νmax/cm−1): 3351, 3181, 1623, 1545. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.07 (t, J = 6 Hz, 2H, CH2), 2.50 (t, J = 6 Hz, 2H, CH2), 3.23 (s, 2H, CH2), 3.66 (s, 2H, CH2), 3.74 (s, 3H, OCH3), 3.80 ( s, 3H, OCH3), 5.30 (s, 1H, CH), 6.57 (brs, 1H, NH), 6.59-7.40 (m, 8H, H-Ar), 8.51 (s, 2H, NH), 11.75 (s, 1H, NH). MS (EI, 70 eV): m/z (%): 463 (M + M+, 27), 462 (M+, 100). Anal. calcd for C25H26N4O3S (462.17): C, 64.91; H, 5.67; N, 12.11; found: C, 64.72; H, 5.63; N, 12.07.
7-Benzyl-2-thioxo-5-(3,4,5-trimethoxyphenyl)-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4d )
Yellow powder (90%); m.p. 228–230 °C. IR (KBr) (νmax/cm−1): 3387, 3182, 1623, 1592. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.35 (t, J = 6 Hz, 2H, CH2), 2.50 (t, J = 6 Hz, 2H, CH2), 3.61 (s, 2H, CH2), 3.65 (s, 2H, CH2), 3.67 (s, 3H, OCH3), 3.81 (s, 6H, 2OCH3), 5.33 (s, 1H, CH), 6.36 (brs, 1H, NH), 6.49–7.35 (m, 6H, H-Ar), 9.89 (brs, 1H, NH), 11.95 (brs, 2H, NH). 13C NMR (75 MHz, DMSO-d6): δ (ppm), 21.7, 33.0, 41.09, 52.7, 56.3, 61.1, 78.6, 90.9, 104.8, 107.2, 125.7, 126.9, 128.7, 129.1, 134.5, 136.0, 152.9, 154.2, 163.4, 173.2. MS (EI, 70 eV): m/z (%): 492 (M+, 100), 493 (M + M+, 28). Anal. calcd for C26H28N4O4S (492.59): C, 63.40; H, 5.73; N, 11.37; found: C, 63.34; H, 5.70; N, 11.39.
7-Benzyl-5-(4-fluorophenyl)-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4e )
Yellow powder (75%); m.p. 234–237 °C. IR (KBr) (νmax/cm−1): 3371, 3193, 3109, 1620, 1588. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.34 (t, J = 6 Hz, 2H, CH2), 2.67 (t, J = 6 Hz, 2H, CH2), 3.41 (s, 2H, CH2), 3.60 (s, 2H, CH2), 5.29 (s, 1H, CH), 6.96 (brs, 1H, NH), 6.99-7.35 (m, 9H, H-Ar), 7.95 (s, 2H, NH), 11.65 (s, 1H, NH). MS (EI, 70 eV): m/z (%): 420 (M+, 100), 421 (M + M+, 24). Anal. calcd for C23H21FN4OS (420.51): C, 65.70; H, 5.03; N, 13.32; found: C, 65.62; H, 5.08; N, 13.39.
7-Benzyl-5-(2,4-difluorophenyl)-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4f )
Yellow powder (90%); m.p. 230–233 °C. IR (KBr) (νmax/cm−1): 3428, 3325, 3111, 1624, 1593. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.34 (t, J = 6 Hz, 2H, CH2), 2.67 (t, J = 6 Hz, 2H, CH2), 3.07 (s, 2H, CH2), 3.60 (s, 2H, CH2), 5.32 (s, 1H, CH), 6.38 (brs, 1H, NH), 6.63–7.35 (m, 8H, H-Ar), 8.96 (s, 1H, NH), 11.61 (brs, 2H, NH). 13C NMR (75 MHz, DMSO-d6): δ (ppm) 21.7, 31.2, 41.1, 52.7, 61.1, 78.6, 90.7, 125.7, 126.9, 127.5, 128.3, 128.7, 129.1, 129.3, 129.7, 138.4, 153.9, 154.3, 162.1, 163.2, 175.0. MS (EI, 70 eV): m/z (%): 439 (M + M+, 25), 438 (M+, 100). Anal. calcd for C23H20F2N4OS (438.50): C, 63.00; H, 4.60; N, 12.78; found: C, 63.32; H, 4.83; N, 12.99.
7-Benzyl-2-thioxo-5-[4-(trifluoromethyl)phenyl]-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4g )
Colorless powder (87%); m.p. 288–290 °C. IR (KBr) (νmax/cm−1): 3500, 3320, 1605, 1560. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.50 (t, J = 6 Hz, 2H, CH2), 2.73 (t, J = 6 Hz, 2H, CH2), 3.34 (s, 2H, CH2), 3.60 (s, 2H, CH2), 5.40 (s, 1H, CH), 6.79 (brs, 1H, NH), 7.31-7.58 (m, 9H, H-Ar), 7.96 (s, 1H, NH), 11.89 (brs, 1H, NH), 12.11 (brs, 1H, NH). 13C NMR (75 MHz, DMSO-d6): δ (ppm) 21.7, 31.2, 41.1, 52.7, 61.1, 78.6, 90.7, 123.9, 125.7, 126.9, 127.5, 128.3, 128.7, 129.0, 129.1, 129.7, 138.4, 153.9, 162.1, 173.3. MS (EI, 70 eV): m/z (%): 470 (M+, 100). Anal. calcd for C24H21F3N4OS (470.51): C, 61.27; H, 4.50; N, 11.91; found: C, 61.92; H, 4.83; N, 11.99.
7-Benzyl-5-(4-nitrophenyl)-2-thioxo-2,3,5,6,7,8,9,10-octahydropyrimido[4,5-b][1,6]naphthyridin-4(1H)-one (4h )
Colorless powder (90%); m.p. <300 °C. IR (KBr) (νmax/cm−1): 3340, 3140, 1600, 1541. 1H NMR (300 MHz, DMSO-d6): δ (ppm) 2.49 (t, J = 6 Hz, 2H, CH2), 2.50 (t, J = 6 Hz, 2H, CH2), 3.56 (s, 2H, CH2), 3.60 (s, 2H, CH2), 5.42 (s, 2H, CH2), 6.77 (brs, 1H, NH), 7.35-8.14 (m, 9H, H-Ar), 8.17 (s, 1H, NH), 11.80 (brs, 1H, NH). MS (EI, 70 eV): m/z (%): 447 (M+, 100). Anal. calcd for C23H21N5O3S (447.51): C, 61.73; H, 4.73; N, 15.65; found: C, 61.75; H, 4.73; N, 15.69.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
