Experimental
Unless otherwise noted, all reactions were carried out in sealed oven-dried Schlenk tubes under air. Reagents and solvents were obtained from commercial suppliers and used without purification. Flash column chromatography was performed using 200–300 mesh silica gel. Visualization on thin-layer chromatography (TLC) was achieved by the use of UV light (254 nm). A FULI GC-9790II equipped with a flame ionization detector (FID) detector was used to analysis the reaction mixture. 1H nuclear magnetic resonance (NMR) and 13C NMR spectra were recorded on a Bruker AV-II 500-MHz NMR spectrometer (
1
H: 500 MHz, 13C: 125.76 MHz) in CDCl3 or DMSO-d6. The coupling constants J are given in Hz. Chemical shifts for 1H NMR are referred to internal Me4Si (0 ppm). GC-MS was recorded on a Shimadzu GCMS-QP2010 plus equipped with an electron ionization (EI) ion source. Substrates 1c,
35
1d,
36
1f,
37
1g,
38
1h–m,
39
1n,
37
1o,
39
and 1p
36
were synthesized according to the known methods.
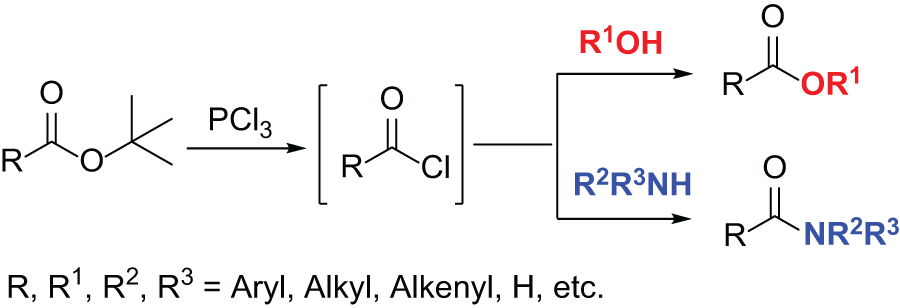
Typical procedure for the preparation of the target molecules: Under air, tert-butyl ester 1 (1.3 or 0.3 mmol), PCl3 (1.0 equiv.), and CH3CN (0.6 mL) were added to a 25-mL sealed Schlenk tube equipped with a magnetic stir bar. The mixture was stirred at 80 °C for 3 h. Next, the corresponding alcohol (5.0 equiv.) or amine (1.0 or 3.0 equiv.) was added to the reaction and the mixture was stirred at the indicated temperature for the indicated amount of time. The mixture was then quenched with aqueous NaHCO3 solution and extracted with EtOAc (×3). The combined organic layer was dried over MgSO4 and filtered. After evaporation of the solvent under reduced pressure, the residue was purified by column chromatography on silica gel to give the analytically pure product 2 or 3.
Methyl benzoate (2a):
40
Colorless oil; yield: 86% (152.1 mg). Petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz, CDCl3): δ 8.07–8.05 (m, 2H), 7.59–7.56 (m, 1H), 7.47–7.44 (m, 2H), 3.93 (s, 3H). 13C NMR (125.76 MHz CDCl3): δ 167.2, 133.0, 130.1, 129.6, 128.4, 52.2. GC-MS (EI, 70 eV): m/z = 136 (M+).
Methyl 4-methylbenzoate (2b):
40
Colorless oil; yield: 72% (140.5 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.84 (d, J = 8.0 Hz, 2H), 7.14 (d, J = 8.0 Hz, 2H), 3.80 (s, 3H), 2.31 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 167.2, 143.6, 129.6, 129.1, 127.4, 51.9, 21.6. GC-MS (EI, 70 eV): m/z = 150 (M+).
Methyl 2,4,6-trimethylbenzoate (2c):
41
Colorless oil; yield: 83% (192.1 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 6.66 (s, 2H), 3.70 (s, 3H), 2.11 (s, 6H), 2.09 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 170.6, 139.3, 135.2, 130.9, 128.4, 51.7, 21.1, 19.8. GC-MS (EI, 70 eV): m/z = 178 (M+).
Methyl 4-methoxybenzoate (2d):
40
White solid; yield: 81% (174.8 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.93–7.90 (m, 2H), 6.86–6.83 (m, 2H), 3.81 (s, 3H), 3.78 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 166.9, 163.3, 131.6, 122.6, 113.6, 55.4, 51.9. GC-MS (EI, 70 eV): m/z = 166 (M+).
Methyl 4-fluorobenzoate (2e):
42
Colorless oil; yield: 85% (170.2 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 8.02–8.00 (m, 2H), 7.08–7.04 (m, 2H), 3.87 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 165.5, 165.2 (d, JC-F = 252.2 Hz), 131.6 (d, JC-F = 9.3 Hz), 125.9 (d, JC-F = 2.6 Hz), 114.9 (d, JC-F = 21.9 Hz), 51.6. GC-MS (EI, 70 eV): m/z = 154 (M+).
Methyl 4-chlorobenzoate (2f):
40
White solid; yield: 94% (207.7 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.90 (d, J = 8.5 Hz, 2H), 7.34 (d, J = 8.5 Hz, 2H), 3.84 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 165.8, 138.9, 130.5, 128.2, 128.1, 51.8. GC-MS (EI, 70 eV): m/z = 170 (M+).
Methyl 4-(trifluoromethyl)benzoate (2g):
40
Light yellow oil; yield: 93% (246.6 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 8.07 (d, J = 8.0 Hz, 2H), 7.63 (d, J = 8.5 Hz, 2H), 3.88 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 165.4, 133.9 (q, JC-F = 32.7 Hz), 132.9, 129.5, 124.9 (q, JC-F = 3.1 Hz), 123.1 (q, JC-F = 272.8 Hz), 52.0. GC-MS (EI, 70 eV): m/z = 204 (M+).
Methyl 4-acetylbenzoate (2h):
43
White solid; yield: 64% (148.1 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 8.06 (d, J = 8.5 Hz, 2H), 7.94 (d, J = 8.5 Hz, 2H), 3.88 (s, 3H), 2.58 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 197.1, 165.8, 139.7, 133.4, 129.4, 127.7, 52.0, 26.4. GC-MS (EI, 70 eV): m/z = 178 (M+).
Methyl 4-vinylbenzoate (2i):
44
White solid; yield: 63% (132.7 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.92 (d, J = 8.5 Hz, 2H), 7.38 (d, J = 8.5 Hz, 2H), 6.70–6.64 (m, 1H), 5.79 (d, J = 17.5 Hz, 1H), 5.30 (d, J = 11.0 Hz, 1H), 3.83 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 166.4, 141.4, 135.5, 129.4, 128.8, 125.6, 116.0, 51.6. GC-MS (EI, 70 eV): m/z = 162 (M+).
Methyl 2-naphthoate (2j):
40
Light yellow solid; yield: 90% (217.6 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 8.54 (s, 1H), 7.89 (dd, J = 1.5 Hz, 8.5 Hz, 1H), 7.89 (d, J = 8.0 Hz, 1H), 7.81 (d, J = 8.5 Hz, 2H), 7.54–7.46 (m, 2H), 3.91 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 167.3, 135.5, 132.5, 131.1, 129.4, 128.3, 128.2, 127.8, 127.4, 126.7, 125.2, 52.3. GC-MS (EI, 70 eV): m/z = 186 (M+).
Methyl 2-(naphthalen-2-yl)acetate (2k):
45
White solid; yield: 70% (182.0 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.74–7.01 (m, 3H), 7.65 (s, 1H), 7.41–7.32 (m, 3H), 3.71 (s, 2H), 3.62 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 172.1, 133.5, 132.5, 131.5, 128.3, 128.0, 127.7, 127.7, 127.4, 126.2, 125.9, 52.2, 41.4. GC-MS (EI, 70 eV): m/z = 200 (M+).
Methyl 2-(p-tolyl)acetate (2l):
46
Colorless oil; yield: 96% (204.7 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.07–7.02 (m, 4H), 3.57 (s, 3H), 3.48 (s, 2H), 2.22 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 171.8, 136.3, 130.5, 128.9, 128.7, 51.6, 40.3, 20.6. GC-MS (EI, 70 eV): m/z = 164 (M+).
Methyl 2-phenylpropanoate (2m):
47
Light yellow oil; yield: 95% (202.5 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.39–7.34 (m, 4H), 7.31–7.28 (m, 1H), 3.78 (q, J = 7.0 Hz, 1H), 3.69 (s, 3H), 1.55 (d, J = 7.0 Hz, 3H). 13C NMR (125.76 MHz, CDCl3): δ 174.5, 140.1, 128.2, 127.0, 126.7, 51.6, 45.0, 18.2. GC-MS (EI, 70 eV): m/z = 164 (M+).
Methyl cinnamate (2n):
42
Light yellow solid; yield: 95% (200.1 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.63 (d, J = 16.0 Hz, 1H), 7.47–7.45 (m, 2H), 7.33–7.30 (m, 3H), 6.38 (d, J = 16.0 Hz, 1H), 3.74 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 167.5, 144.9, 134.4, 130.3, 128.9, 128.1, 117.8, 51.7. GC-MS (EI, 70 eV): m/z = 162 (M+).
Methyl 3-phenylpropanoate (2o):
48
Light yellow oil; yield: 96% (204.7 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.19–7.16 (m, 2H), 7.11–7.08 (m, 3H), 3.55 (s, 3H), 2.85 (t, J = 7.5 Hz, 2H), 2.52 (t, J = 8.0 Hz, 2H). 13C NMR (125.76 MHz, CDCl3): δ 172.9, 140.2, 128.1, 127.8, 125.8, 51.2, 35.3, 30.5. GC-MS (EI, 70 eV): m/z = 164 (M+).
Methyl hexanoate (2p):
40
Colorless oil; yield: 94% (158.9 mg). Eluent: petroleum ether. 1H NMR (500 MHz CDCl3): δ 3.60 (s, 3H), 2.23 (t, J = 7.5 Hz, 2H), 1.59–1.53 (m, 2H), 1.27–1.22 (m, 4H), 0.83 (t, J = 7.0 Hz, 3H). 13C NMR (125.76 MHz, CDCl3): δ 174.3, 51.4, 34.1, 31.3, 24.6, 22.3, 13.9. GC-MS (EI, 70 eV): m/z = 130 (M+).
Ethyl benzoate (2q):
47
Light yellow oil, yield 96% (187.2 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.99–7.97 (m, 2H), 7.50–7.46 (m, 1H), 7.38–7.35 (m, 2H), 4.31 (q, J = 7.5 Hz, 2H), 1.33 (t, J = 7.0 Hz, 3H); 13C NMR (125.76 MHz, CDCl3): δ 166.7, 132.8, 130.5, 129.5, 128.3, 61.0, 14.4. GC-MS (EI, 70 eV): m/z = 150 (M+).
Isopropyl benzoate (2r):
47
Light yellow oil; yield: 84% (179.1 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.94–7.95 (m, 2H), 7.47–7.44 (m, 1H), 7.36–7.33 (m, 2H), 5.21–5.14 (m, 1H), 1.29 (d, J = 6.5 Hz, 6H). 13C NMR (125.76 MHz, CDCl3): δ 166.1, 132.7, 130.9, 129.5, 128.3, 68.4, 22.0. GC-MS (EI, 70 eV): m/z = 164 (M+).
Cyclohexyl benzoate (2s):
49
Light yellow oil; yield: 87% (230.7 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 7.98–7.96 (m, 2H), 7.46–7.43 (m, 1H), 7.35–7.32 (m, 2H), 4.97–4.92 (m, 1H), 1.87–1.84 (m, 2H), 1.71–1.69 (m, 2H), 1.52–1.47 (m, 3H), 1.40–1.24 (m, 3H). 13C NMR (125.76 MHz, CDCl3): δ 166.0, 132.7, 131.0, 126.5, 128.3, 73.0, 31.7, 25.5, 23.7. GC-MS (EI, 70 eV): m/z = 204 (M+).
Phenyl benzoate (2t):
47
White solid; yield: 81% (208.5 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 8.15–8.13 (m, 2H), 7.58–7.55 (m, 1H), 7.46–7.42 (m, 2H), 7.38–7.34 (m, 2H), 7.22–7.17 (m, 1H), 7.15–7.13 (m, 2H). 13C NMR (125.76 MHz, CDCl3): δ 165.2, 151.0, 133.6, 130.2, 129.6, 129.5, 128.6, 125.9, 121.8. GC-MS (EI, 70 eV): m/z = 198 (M+).
Benzyl benzoate (2u):
40
Colorless oil; yield: 52% (143.3 mg). Eluent: petroleum ether/EtOAc = 10/1. 1H NMR (500 MHz CDCl3): δ 8.17–8.15 (m, 2H), 7.62–7.59 (m, 1H), 7.53–7.44 (m, 6H), 7.42–7.39 (m, 1H), 5.44 (s, 2H). 13C NMR (125.76 MHz, CDCl3): δ 166.5, 136.1, 133.1, 130.2, 129.8, 128.7, 128.5, 128.3, 128.3, 66.8. GC-MS (EI, 70 eV): m/z = 212 (M+).
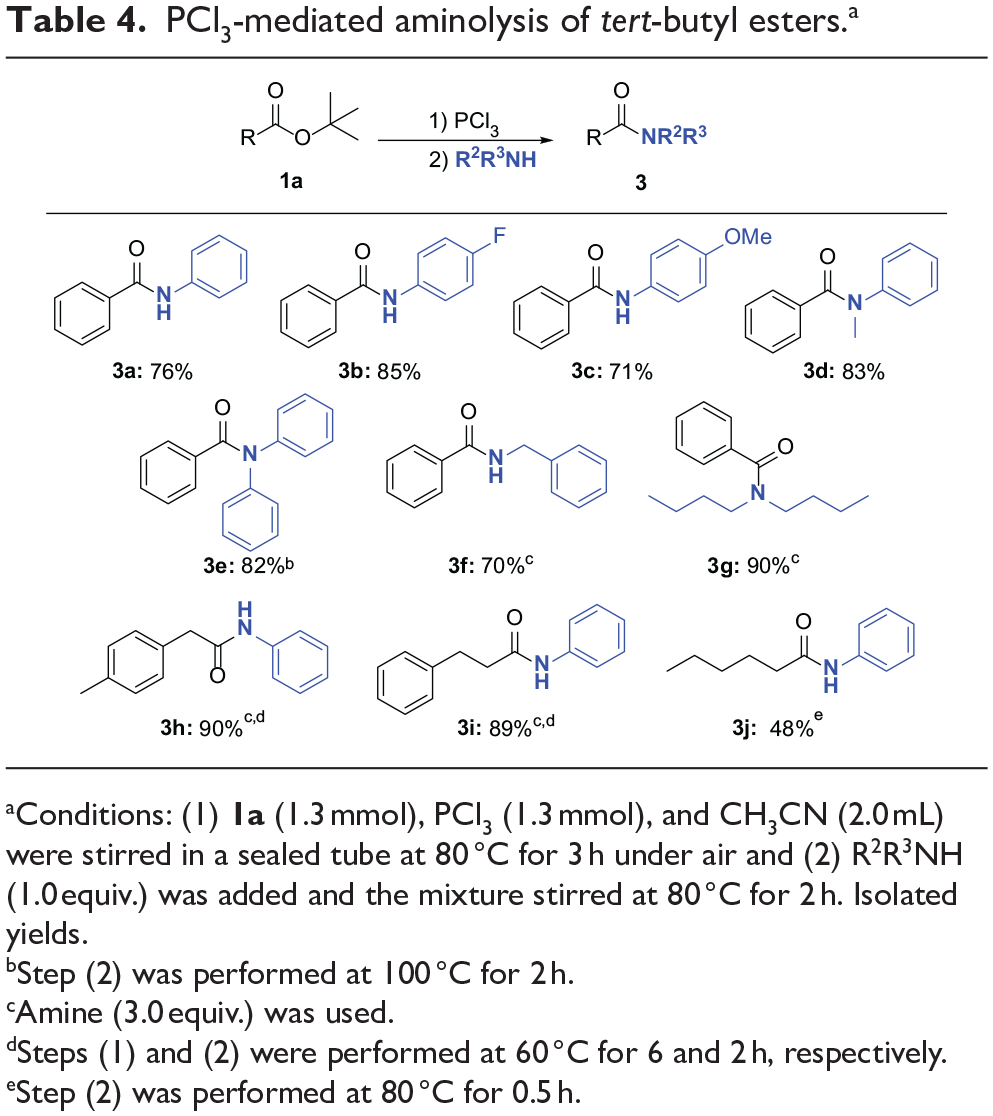
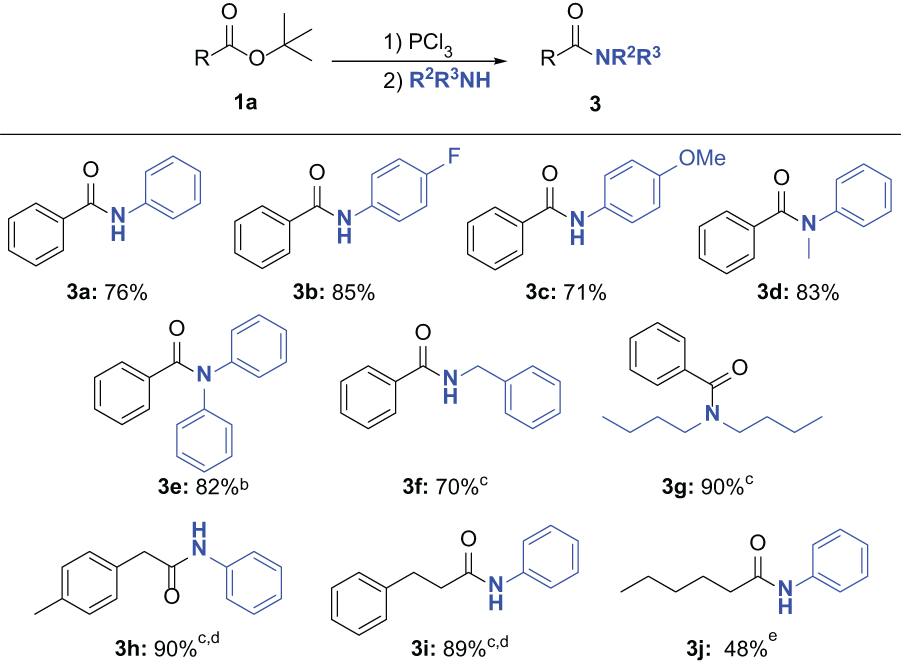
N-phenylbenzamide (3a):
50
White solid; yield: 76% (194.6 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.81–7.78 (m, 3H), 7.58–7.57 (m, 2H), 7.50–7.47 (m, 1H), 7.44–7.41 (m, 2H), 7.31 (t, J = 8.0 Hz, 2H), 7.09 (t, J = 7.5 Hz, 1H). 13C NMR (125.76 MHz, CDCl3): δ 165.8, 137.9, 135.0, 131.9, 129.1, 128.8, 127.0, 124.6, 120.2. GC-MS (EI, 70 eV): m/z = 197 (M+).
N-(4-Fluorophenyl)benzamide (3b):
51
White solid; yield: 85% (237.6 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz DMSO-d6) δ 10.30 (s, 1H), 7.97–7.94 (m, 2H), 7.82–7.78 (m, 2H), 7.61–7.57 (m, 1H), 7.55–7.51 (m, 2H), 7.22–7.17 (m, 2H). 13C NMR (125.76 MHz, DMSO-d6) δ 165.9, 158.8 (d, JC-F = 240.2 Hz) 136.0 (d, JC-F = 2.5 Hz), 135.3, 132.1, 128.9, 128.1, 122.7, 115.6 (d, JC-F = 22.6 Hz). GC-MS (EI, 70 eV): m/z = 215 (M+).
(4-Methoxyphenyl)benzamide (3c):
50
Light yellow solid; yield: 71% (209.5 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.79 (d, J = 7.5 Hz, 2H), 7.73 (brs, 1H), 7.48–7.45 (m, 3H), 7.42–7.39 (m, 2H), 6.85–6.82 (m, 2H), 3.74 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 165.7, 156.7, 135.0, 131.7, 131.0, 128.8, 127.0, 122.1, 114.3, 55.5. GC-MS (EI, 70 eV): m/z = 227 (M+).
N-Methyl-N-phenylbenzamide (3d):
50
Light yellow oil; yield: 83% (227.7 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.22–7.21 (m, 2H), 7.17–7.13 (m, 3H), 7.09–7.04 (m, 3H), 6.95 (d, J = 7.5 Hz, 2H), 3.42 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 170.7, 144.9, 135.9, 129.6, 129.2, 128.7, 127.7, 126.9, 126.5, 38.4. GC-MS (EI, 70 eV): m/z = 211 (M+).
N,N-Diphenylbenzamide (3e):
50
White solid; yield: 82% (291.1 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.39–7.38 (m, 2H), 7.23–7.20 (m, 5H), 7.16–7.08 (m, 8H). 13C NMR (125.76 MHz, CDCl3): δ 170.8, 143.9, 136.1, 130.2, 129.2, 129.1, 127.9, 127.5, 126.4. GC-MS (EI, 70 eV): m/z = 273 (M+).
Benzylbenzamide (3f):
50
White solid, yield 70% (192.0 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.73–7.71 (m, 2H), 7.45–7.42 (m, 1H), 7.38–7.34 (m, 2H), 7.29–7.21 (m, 5H), 6.36 (brs, 1H), 4.58 (d, J = 5.5 Hz, 2H). 13C NMR (125.76 MHz, CDCl3): δ 167.4, 138.2, 134.4, 131.6, 128.8, 128.6, 128.0, 127.7, 127.0, 44.2. GC-MS (EI, 70 eV): m/z = 211 (M+).
N,N-Dibutylbenzamide (3g):
52
Light yellow oil; yield: 90% (272.6 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.30–7.25 (m, 5H), 3.41–3.10 (m, 4H), 1.57–1.32 (m, 6H), 1.04–0.70 (m, 8H). 13C NMR (125.76 MHz, CDCl3): δ 171.6, 137.4, 129.0, 128.3, 126.4, 48.7, 44.4, 30.8, 29.6, 20.3, 19.7, 13.9, 13.6. GC-MS (EI, 70 eV): m/z = 233 (M+).
N-Phenyl-2-(p-tolyl)acetamide (3h):
53
White solid; yield: 90% (263.3 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.34–7.33 (m, 2H), 7.20–7.17 (m, 3H), 7.14–7.10 (m, 4H), 7.01–6.98 (m, 1H), 3.61 (s, 2H), 2.29 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 169.51, 137.68, 137.43, 131.33, 129.95, 129.46, 128.93, 124.44, 119.87, 44.41, 21.15. GC-MS (EI, 70 eV): m/z = 225 (M+).
N,3-Diphenylpropanamide (3i):
53
White solid; yield: 89% (260.3 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.75–7.70 (m, 1H), 7.35–7.33 (m, 2H), 7.17–7.06 (m, 7H), 6.98–6.95 (m, 1H), 2.90 (t, J = 8.0 Hz, 2H), 2.52 (t, J = 8.0 Hz, 2H). 13C NMR (125.76 MHz, CDCl3): δ 171.02, 140.66, 137.89, 128.96, 128.65, 128.41, 126.38, 124.39, 120.30, 39.25, 31.64. GC-MS (EI, 70 eV): m/z = 225 (M+).
N-Phenylhexanamide (3j):
54
White solid; yield: 48% (119.2 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.53–7.51 (m, 2H), 7.38 (brs, 1H), 7.30 (t, J = 8.0 Hz, 2H), 7.11–7.08 (m, 1H), 2.35 (t, J = 7.5 Hz, 2H), 1.75–1.69 (m, 2H), 1.38–1.33 (m, 4H), 0.90 (t, J = 7.0 Hz, 3H). 13C NMR (125.76 MHz, CDCl3): δ 171.61, 138.00, 128.98, 124.18, 119.84, 37.80, 31.44, 25.37, 22.45, 13.96. GC-MS (EI, 70 eV): m/z = 191 (M+).
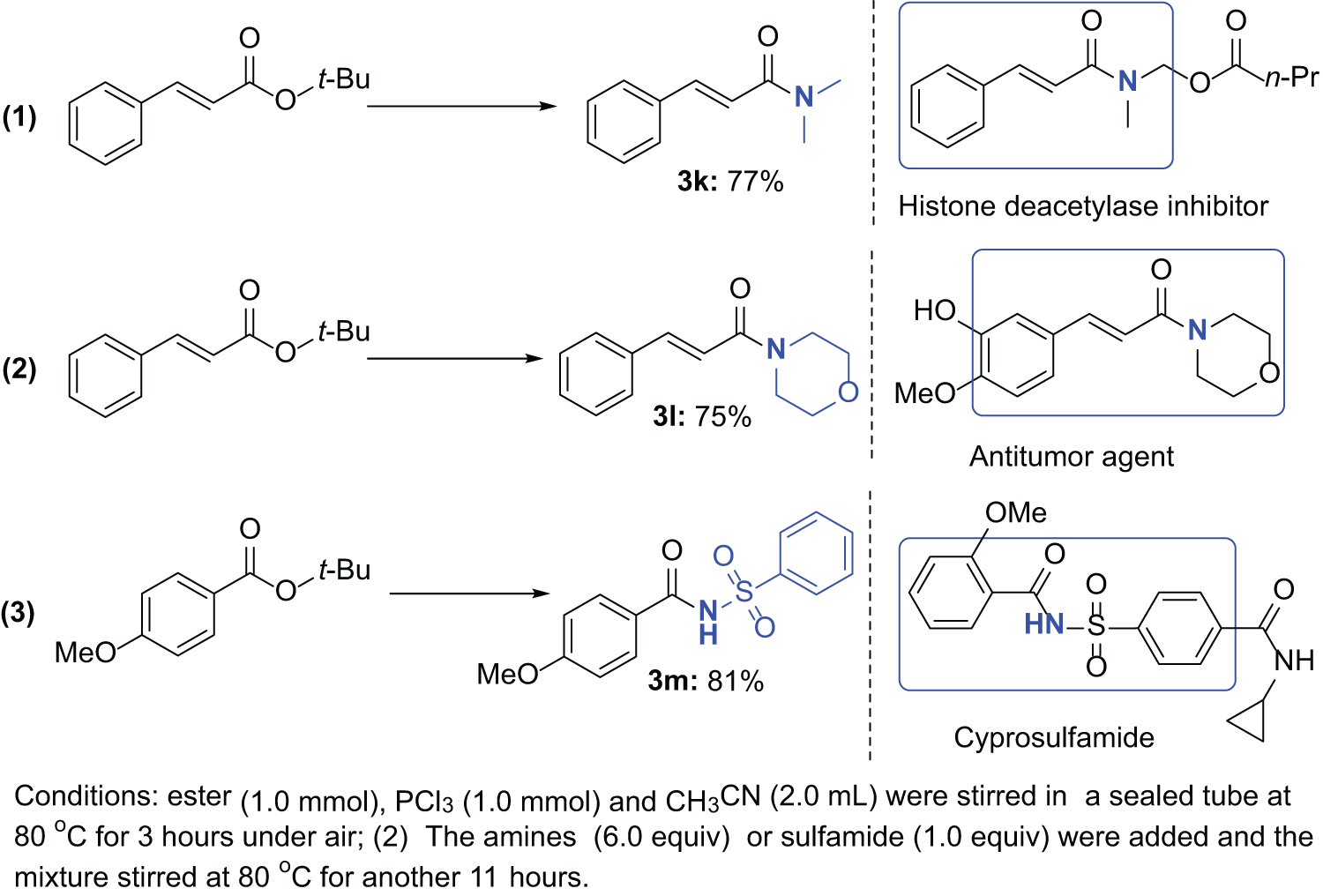
N,N-Dimethylcinnamamide (3k):
55
White solid; yield: 77% (134.8 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.69 (d, J = 15.5 Hz, 1H), 7.56–7.54 (m, 2H), 7.41–7.34 (m, 3H), 6.91 (d, J = 15.5 Hz, 1H), 3.18–3.09 (m, 6H). 13C NMR (125.76 MHz, CDCl3): δ 166.7, 142.4, 135.3, 129.6, 128.8, 127.8, 117.4, 37.5, 36.0. GC-MS (EI, 70 eV): m/z = 175 (M+).
(E)-1-Morpholino-3-phenylprop-2-en-1-one (3l):
56
White solid; yield: 75% (162.8 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 7.63 (d, J = 15.5 Hz, 1H), 7.47–7.45 (m, 2H), 7.33–7.28 (m, 3H), 6.78 (d, J = 15.5 Hz, 1H), 3.66–3.62 (m, 8H). 13C NMR (125.76 MHz, CDCl3): δ 165.6, 143.3, 135.1, 129.8, 128.9, 127.8, 116.5, 66.9, 46.2, 42.5. GC-MS (EI, 70 eV): m/z = 217 (M+).
4-Methoxy-N-(phenylsulfonyl)benzamide (3m):
57
White solid; yield: 81% (235.7 mg). Eluent: petroleum ether/EtOAc = 5/1. 1H NMR (500 MHz CDCl3): δ 9.47 (brs, 1H), 8.10–8.08 (m, 2H), 7.75–7.32 (m, 2H), 7.59–7.56 (m, 1H), 7.50–7.46 (m, 2H), 6.81–6.80 (m, 2H), 3.74 (s, 3H). 13C NMR (125.76 MHz, CDCl3): δ 164.0, 163.8, 138.6, 134.0, 130.2, 129.0, 128.6, 123.1, 114.2, 55.6. GC-MS (EI, 70 eV): m/z = 291 (M+).