Abstract
Aflatoxins are extremely harmful carcinogens to humans and animals. In recent years, attention has been directed toward the application of molecular imprinting polymers for the separation and detection of aflatoxin. In this review, polymerization methods for the preparation of molecular imprinting polymers for aflatoxin detection, such as lump-bulk polymerization, spherical molecular imprinting polymer synthesis, surface-imprinted polymerization, and electropolymerization, are described. The applications of molecular imprinting polymers in solid-phase extraction, biosensors, and the surface-enhanced Raman detection of aflatoxin are also reviewed in this paper.
Keywords

Introduction
Aflatoxins (AF) are difuranocoumarin metabolites produced by

Examples of aflatoxins.
The rapid, accurate, and effective separation of AF from complex samples is highly desired in the food detection fields. In recent years, a number of articles concerning the detection of toxins have also been published. Well-known methods for the detection of AF include thin-layer chromatography (TLC), 4 enzyme-linked immunosorbent assays (ELISA), 5 high-performance liquid chromatography (HPLC), 6 high-performance liquid chromatography-mass spectrometry (HPLC-MS), 7 and electrochemical sensors. 8 However, pretreatment of the target analytes is required to eliminate the background interference before detection. An immunoaffinity column is often used for the pretreatment of samples. However, the procedure is operationally complex, consisting of extraction, centrifugation, filtration, and immunoaffinity column analysis.9–12 The immunoaffinity column is relatively expensive and unstable, and adsorption is easily affected by organic solvents, sample pH, and the system temperature. Due to economic and practical considerations, an increasing number of more economical methods for AF enrichment and detection have emerged in recent years. Among these methods, molecular imprinting polymers (MIPs) have attracted extensive attention. MIPs can selectively identify a target analyte because of its three-dimensional (3D) binding cavities which match the structure of the target analyte (generally called the template). In this overview, the application of AF-imprinted polymers for the detection of AF is summarized. This review paper includes (1) an introduction on the molecular imprinting technique (MIT), (2) a discussion of AF MIPs, (3) methods for the preparation of AF MIPs, and (4) the application of MIPs for the detection of AF.
MITs
MIT utilizes MIPs with memory functions, which has been termed the “lock and key theory.” The MIT concept was first proposed by Mosbach in 1993. 13 MIPs have three main characteristics: structure-effect predetermination, specific identification, and broad practicality. The application of MIPs mainly includes chromatographic separation, solid-phase extraction (SPE), biomimetic sensing, and membrane separation. 14 MIPs are prepared by the co-polymerization of a functional monomer and crosslinking agent in the periphery of template molecules. Before co-polymerization, the template molecule and functional monomer are fully mixed and interact with each other via non-covalent or covalent bonds. Subsequently, crosslinking agents and a radical initiator are added to initiate the polymerization process. After polymerization, the template molecules are fixed in the 3D cavities of the resulting polymer. Finally, the template molecule is removed via a physical or chemical method, thereby leaving behind specific 3D cavities complementary in size and shape to the template molecule in the MIPs (Figure 2). Thus, MIPs can be used to adsorb molecules that are structurally similar to the template molecule.

The preparation principle of MIPs.
AF MIPs
Due to the high toxicity and high cost of AF, the use of structural analogs as dummy templates for the preparation of MIPs is highly desired. 15 Reported dummy template molecules include 5,7-dimethoxycoumarin (DMC), 2 quercetin, 16 7-acetoxy-4-methylcoumarin, 17 and 6-phenyl-4-methyl-2-chromanone (Figure 3). 18 DMC is a common template molecule for the preparation of AF MIPs.

Examples of dummy template molecules for aflatoxin MIPs.
In addition to template molecules, the functional monomer is also a prerequisite for the preparation of MIPs. Reported functional monomers include methacrylic acid (MAA), acrylamide, 2-vinyl pyridine, and so on. Among them, MAA is used frequently for MIP preparation. The carbon–carbon double bond in MAA is used for polymerization, and the carboxyl groups interact with DMC by hydrogen bonding (Figure 4). 19 Ethylene glycol dimethacrylate (EDMA) is a common crosslinking agent for polymerization which leads to the formation of a 3D network structure in the presence of a radical initiator; molecular imprinted cavities are formed after removing the template molecule.

Interaction of DMC with MAA by hydrogen bonding.
To explore the use of effective functional monomers, Sergeyeva et al. 20 established a virtual library of 24 functional monomers, which can interact with AFs B1 and B2 via non-covalent bonding. An algorithm was used to analyze the possible interactions (ion and hydrogen bonds, van der Waals, dipole–dipole interactions, and spatial interactions) between the functional monomer and template molecule. Molecular dynamic research was carried out in order to obtain the correct molar ratio of the template molecule and functional monomer. It was found that acrylamide also interacted well with AF B1, indicating that computational simulation is an effective method for the discovery of potent functional monomers.
Methods for the preparation of AF MIPs
The majority of AF MIPs can be prepared by lump-bulk polymerization, spherical MIP synthesis, surface-imprinted polymerization, and electro-polymerization.
Lump-bulk polymerization
Bulk polymerization21–24 is a traditional method for the preparation of MIPs. The template molecule, functional monomer, crosslinking agent, and initiator are mixed in a non-polar solvent in a certain proportion. The polymerization reaction is initiated by photo or thermal irradiation. After polymerization, the resulting block polymer must be ground, crushed, and sieved so that the template molecule can be extracted from the polymer. For example, Arak et al. 25 employed the bulk polymerization method in order to prepare an AF-MIP using DMC as the template, MAA as the monomer, and EDMA as a cross-linker. The amelioration of AF in ducklings was examined by feeding the ducks the synthesized MIPs to target the AF B1 (AFB1) analog. DMC was used as a smart and novel toxin binder and compared with other commercial toxin-binding agents. The sorbent presented strong binding ability, selectivity, and stability under in vitro conditions.
Bulk polymerization has the advantages of being operationally simple and inexpensive. A disadvantage is that the polymerization process involves tedious steps such as pounding, grinding, and screening, which are time-consuming and laborious. These procedures also damage the spatial structure and recognition holes of the polymer and weaken the specific adsorption of the template molecules by the polymer. However, the use of a high concentration of crosslinking agent leads to the template molecule being embedded too deeply and so requiring copious amounts of eluent to remove. The synthesized MIPs do not possess a fixed shape.
In order to overcome the disadvantages of the bulk polymerization method, an in situ polymerization method had also been developed. The in situ polymerization21,26 involves the use of a rod-shaped MIP separation medium prepared by direct polymerization in an empty column or capillary. In this method, the mixed solution for pre-polymerization is directly injected into a column or capillary to prepare a continuous rod-shaped polymer. It has the characteristics of continuity and homogeneity and can provide high separation efficiency. Sergeyeva et al. 20 developed an in situ synthesis of a nanostructured molecular–imprinted membrane for the selective and sensitive recognition of AF B1. Computational modeling methods were employed to optimize the imprinted membrane. Ethyl 2-oxochlorocyclopentanoate was used as a template molecule. The surface binding sites of the imprinted membrane can selectively recognize AF B1. The detection limit of the fluorescent sensor system based on this MIP membrane is 14 ng mL−1, and the detection range is 14–500 ng mL−1.
Spherical MIP synthesis
Although the MIPs prepared by lump-bulk polymerization provide adequate selectivity, they still possess a number of shortcomings, including a high diffusion barrier, low-rate mass transfer, and poor site accessibility. A spherical MIP has a regular spherical shape and a particle size within a certain range. In order to improve the efficiency of MIPs for the detection of AF, a variety of effective synthetic methods for the preparation of spherical MIPs have been developed such as suspension polymerization, emulsion polymerization, and precipitation polymerization.
Suspension polymerization
Suspension polymerization27–29 is the polymerization of monomers suspended in water in small droplets. It proceeds in two immiscible phases: a continuous phase and a dispersed phase. The dispersed phase involves functional monomers, initiators, porogens, and template molecules. Polyvinyl alcohols are often added to the continuous phase as a suspending agent to enhance its stability. A small droplet is equivalent to a small unit from the bulk polymerization, so that the shape and particle size of the polymer microspheres can be well controlled. The porosity of the surface of the particles can be effectively controlled by adjusting the amount of porogen. In order to prevent the particles from sticking to each other, a suspending agent must be added to the system in order to form a protective film on the surface of the particles.
Song et al.
18
reported a hydrophilic MIP prepared by suspension polymerization. Glycidyl methacrylate (GMA) is employed to regulate the hydrophilicity of the polymer, and polyvinyl alcohol is used as a suspending agent (Figure 5). After polymerization, epoxy bonds are successfully formed on the surfaces of the polymer nanospheres. Functional monomers, including MAA, acrylonitrile, acrylic acid,

Suspension polymerization process.
Emulsion polymerization
In order to obtain smaller MIP particles, emulsion polymerization has been developed. Emulsion polymerization is also an effective strategy for the preparation of spherical MIPs.30–32 Water is often used as the medium leading to a high polymerization rate, providing a relatively high molecular weight with a broad particle size distribution ranging from 50 to 1000 nm. The binding sites on the surface of the imprinted microspheres are distributed evenly, and the reuse rate of MIPs is high. Moreover, the viscosity of the system is low, which facilitates the heat transfer.
Wei et al.
33
developed AF MIP microspheres using emulsion polymerization. The MIPs are prepared using Span80 and cetyltrimethylammonium bromide as surfactants,
Precipitation polymerization
In precipitation polymerization, the monomer and initiator can be dissolved in the reaction medium, but the polymer itself is insoluble in the reaction medium; the polymer will precipitate out of the reaction system after a solid granular product is formed. Precipitation polymerization is an ideal method for the routine production of imprinted polymer microparticles with desirable characteristics. There is no suspending agent or dispersant in the precipitation polymerization process, which overcomes the disadvantage of the high viscosity of the polymerization system. The obtained polymers are of high purity compared with the MIPs prepared by suspension polymerization and emulsion polymerization. Rui et al. 17 prepared highly selective surface MIPs (FDU-12@MIPs) via precipitation polymerization based on molecular surface imprinting techniques. FDU-12-MPS as the carrier was dispersed in ethanol by ultrasonic vibration, and then 7-acetoxy-4-methylcoumarin as a dummy template, MAA as the monomer, EDMA as the crosslinking agent, AIBN as an initiator, and ethanol as the solvent were dissolved in the above-mentioned solution for pre-polymerization at room temperature with magnetic stirring. The reaction was carried out at 85 °C over 5 h. Subsequently, the precipitated FDU-12@MIP was separated and purified by rinsing with a mixture of methanol/acetic acid (4:1, v/v) in order to remove the template using a Soxhlet extractor. The polymer beads synthesized by this method are protected from aggregation during polymerization by their cross-linked surfaces and are completely surfactant free.
Bulk polymerization, emulsion polymerization, suspension polymerization, and precipitation polymerization are traditional approaches for the preparation of MIPs, the main features of which are summarized in Table 1.
Features of traditional approaches for MIPs.

Surface polymerization on carriers
The above-mentioned traditional approaches for MIPs still suffer from some drawbacks. For example, the imprinted materials are usually thick, and as a result, the removal of template molecules from the MIPs requires large amounts of solvents, and the adsorption capacity and stability of the MIPs still need to be improved. To overcome these drawbacks, a surface-imprinting strategy has been developed in which the molecularly imprinted layer can be grafted or modified on to the surface of the carriers such as silica gel, magnetic spheres, carbon materials, or quantum dots (QDs). Thus, the recognition sites are exposed on the surface of the MIP, resulting in an increase in the mass-transfer rate and efficiency. Compared with the traditional MITs, the surface MIPs possess more binding sites and provide improved specific recognition and adsorption capacity. Moreover, surface MIPs are able to facilitate the removal and recombination of template molecules.
Song et al. employed mesoporous silica materials, such as FDU-12, 34 SBA15, 35 or the metal-organic framework (MOF) material, HKUST-1, 36 as carriers to support MIPs. 3-Methacryloxypropyltrimethoxysilane (MPS) was linked to the carrier, and its carbon–carbon double bonds are fully exposed on the surface of the carrier. In the presence of radical initiators, polymerization occurs at the surface of the carrier, and a layer of MIPs is formed on the surface. The carrier and the polymer can be combined firmly on the surface of the MIT, and the surface polymer is not easily displaced. The obtained surface MIPs possess a high specific surface area, fast mass transfer rate, fast elution rate, and good reusability. Importantly, surface MIPs can be used as packing materials for SPE columns for the pretreatment of AF and can be considered as promising alternatives for AF immunoaffinity columns. When the MIPs-SPE is combined with HPLC using a fluorescence detector, it provides a new low-cost, low-time-consuming, and highly sensitive detection method for the separation, analysis, and detection of AF in wheat.
Based on silica nanoparticles, Zhang et al.
37
developed imidazolium salt–imprinted molecules supported on silica (Scheme 1).

Preparation of a supported imidazolium salt. 37
QDs are semiconductor nanocrystals that have attracted significant attention as new types of fluorescent materials in recent years, possessing good size tunability, narrow emission spectra, broad excitation spectra, strong signal intensity, and high photostability and biocompatibility. 38 Due to the high selectivity of MIPs, the combination of MIPs with QDs provides a new tool for analyte recognition. Guo et al. 39 developed CdTe-coated MIP-QDs (MIP@CdTe) as a sensitive fluorescence sensor (Scheme 2). In this protocol, DMC is used as a pseudo-template molecule, 3-aminopropyltriethoxysilane as a functional monomer, ethyl orthosilicate as a crosslinking agent, and ammonia as a catalyst. The methoxy and lactone groups of DMC and the amino group (-NH2) of the aminopropyltriethoxysilane (APTES) self-assemble via hydrogen bonding. The optimal molar ratio of DMC to the functional monomer and crosslinking agent is 4:20:15. The relative fluorescence intensity of MIP@CdTe shows a linear relationship ranging from 80 to 400 ng g−1 with regards to AF B1 concentration, and the detection limit reaches 4 ng g−1. Importantly, MIP@CdTe was successfully applied to the quantitative determination of AF B1 in actual samples. The recovery rate of the standard addition ranges from 99.20% to 101.78% and is consistent with the HPLC-MS results.
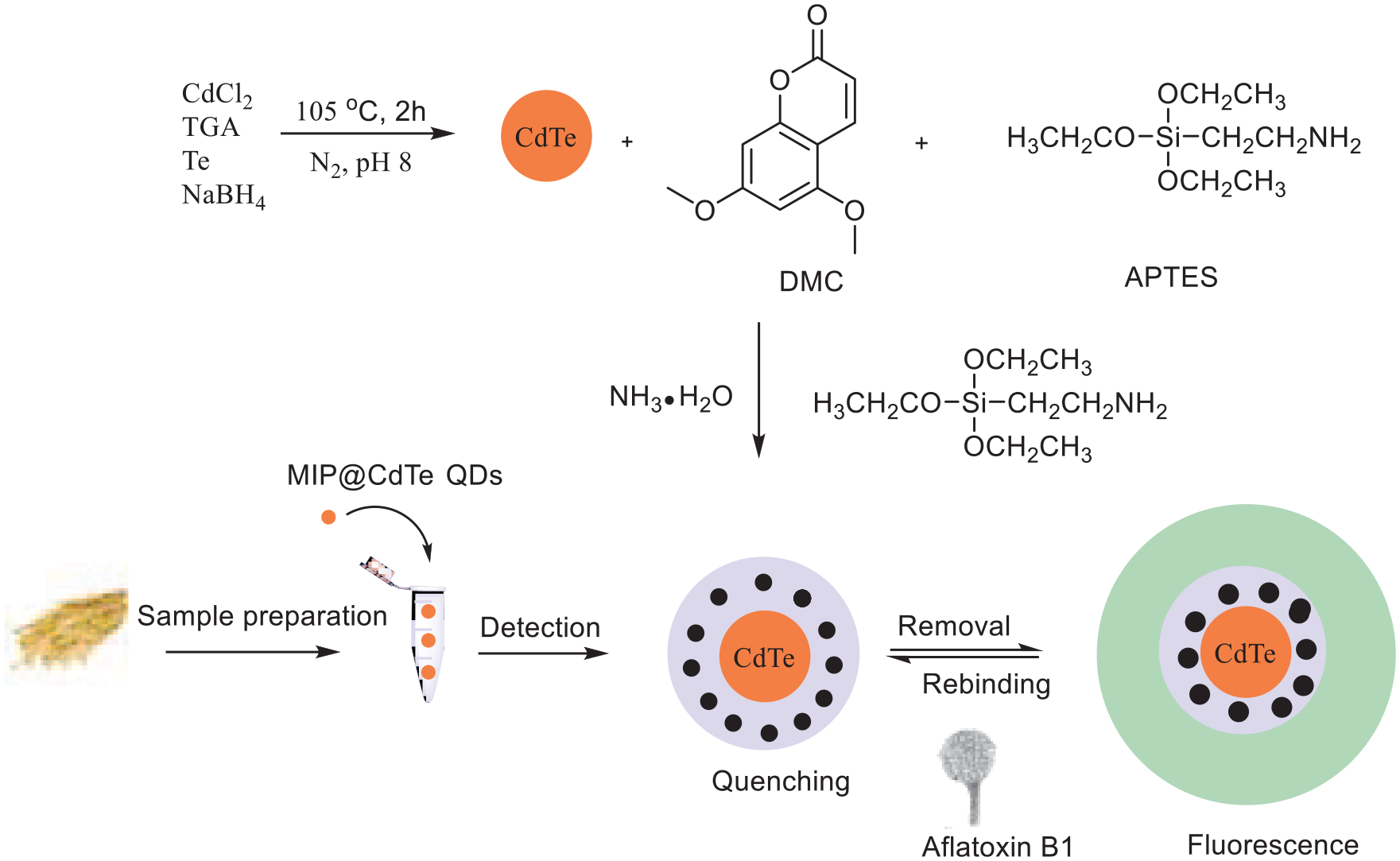
Preparation of MIP@CdTe. 39
Due to the potential toxicity of CdTe, a number of non-cadmium based QDs, such as ZnS, have been developed. Jayasinghe et al. 40 reported surface MIPs on Mn-doped ZnS QDs for the spectral analysis of AF. The MIP-QDs exhibit strong phosphorescence emission at room temperature, which can be quenched when the AF interacts with the recognition cavities of the MIP-QDs. Compared with non-imprinted QD materials, MIP-QDs have better adsorption capacity and selectivity toward AF. The room-temperature phosphorescence quenching of AF is linear in the range of 2–20 μg L−1, and the detection limit is 3.56 μg kg−1. This value is lower than the acceptable residual level (AF in feed) mandated by the European Commission. Thus, MIP-QDs can be used as a general tool for detecting AF in fish feed, with good sensitivity and selectivity. Anis et al. 41 also developed an MIP-QD fluorescent probe for the detection of AF (Scheme 3). The MIP-QD probes were prepared in three steps. First, PEG-Mn-doped ZnS QDs were prepared by the reaction of ZnSO4·7H2O with MnCl2, Na2S, and polyethylene glycol. Second, after preparation of PEG-Mn-doped ZnS, the self-assembly of the template molecule and monomer were carried out, in which DMC was used as a pseudo-template molecule and MAA was used as a functional monomer. Third, the PEG-Mn-doped ZnS particles were dispersed in ultrapure water and mixed with the pre-polymerized mixture (DMC-MAA), the crosslinker divinylbenzene (DVB), and initiator (AIBN) for polymerization. After polymerization, the synthetic material was washed with methanol, centrifuged, and dried to give the corresponding MIP-QD probe. The prepared composite material has high affinity and selectivity for a range of AFs (AF B1, AF B2, AF G1, and AF G2). The detection limit and quantification limit are 0.016 and 0.053 mg L−1, respectively. The recovery rate is 95%–122%, and the relative average deviation (RSD) is less than 7%. The optimized method was also applied to the sensing of all AFs in several non-dairy beverage samples.

Preparation of MIPs on Mn-doped ZnS QDs (MIP-QDs). 41
Magnetic particle–supported surface MIPs (MMIPs) have also been developed. Incorporation of magnetic components such as Fe3O4 nanoparticles into MIPs allows for the preparation of magnetic responsive MIPs (MMIPs), which show directional movement upon application of an external magnetic field. 42 Tan et al. 43 employed vinyl-functionalized superparamagnetic Fe3O4 in order to support the surface MIPs. The procedure for the preparation of the MIPs involves (a) coating magnetic nanoparticles with a silica shell (typical diameter is 200 nm), (b) modifying the surface with methacryloxy groups, and (c) using DMC as a pseudo-template molecule, and MAA and 4-vinylpyridine as functional monomers (Scheme 4). The separation of the samples becomes simpler and more convenient using magnetic MIPs. AF can be eluted from the MIPs, and the amounts can be quantitatively determined by ultra-high performance liquid chromatography tandem mass spectrometry (UHPLC-MS/MS). The detection limit for AFs B2 and G2 is 0.05 μg kg−1, and the detection limit for AFs B1 and G1 is 0.1 μg kg−1.

Preparation of magnetic surface MIPs. 43
Polymerization on an electrode surface
The sensitivity of electrochemical sensors can be enhanced by improving their recognition ability. 44 Significant progress has been made with regards to the synthesis of molecular imprinted sensors over the past 10 years. Several procedures have been used to prepare MIP films on transducer surfaces, including drop-coating or spin-coating of a solution on a prepared polymer, in situ chemical polymerization and electro-polymerization. In comparison with other procedures for MIP film preparation, electro-polymerization allows for the thickness and the density of the MIP films to be regulated via the polymerization conditions (e.g. applied voltage and cyclic scans). 45
Gold nanoparticles (AuNPs) exhibit unique electrochemical properties, high chemical stability, favorable biocompatibility, and can be easily functionalized, thus they have been widely employed for the fabrication of sensors.
38
Gu et al.
46
developed a quartz crystal microbalance (QCM) sensor based on an AuNP-doped molecularly imprinted and covalent organic framework composite (COFs-AuNPs) for the detection of AF B1. The MIP-modified layer on the electrode was electrochemically synthesized by the cyclic voltammetry (CV) method in a solution containing
MOFs, as a highly selective sensor-development platform, have attracted much attention in recent years due to their combined fluorescence and electrochemical detection abilities.47–49 Jiang et al.
49
developed an electrochemical sensor based on a molecularly imprinted MOF for the sensitive and selective detection of AF B1. The MOF was prepared by the electro-polymerization of

The fabrication of an MIP sensor for AF B1 detection. 49
Applications of AF MIPs
Application of MIPs in SPE
Sample preparation is the most time-consuming and laborious step in sample analysis. 50 The enrichment and purification of samples directly affect the precision and accuracy of the analytical results. Although conventional SPE has the advantages of high recovery rate, the need for only small amounts of organic solvent, environmental friendliness, no phase separation operation, and simple collection; however, the limited selectivity and sensitivity remain a problem. 51
MIPs, as a type of synthetic polymeric material, have proven to be excellent adsorbents for molecularly imprinted solid-phase extraction (MISPE). 42 MISPE has remarkable advantages such as possessing specific recognition ability, high predetermined selectivity, high stability, reusability, and is able to overcome background interference in detection.
In 1994, Sellergren 52 reported pioneering work on the use of MIPs as fillers for SPE columns. This technology can quickly and accurately extract the target analyte from samples. Molecularly imprinted polymers acting as sorbents for SPE can be prepared with different physical forms according to specific requirements. The MIP can be made into a whole rod-shaped structure, then ground and sieved, and the polymer with the appropriate particle size can be selected for use in SPE. The spherical particles of undetermined particle size can also be synthesized using different synthetic strategies, such as precipitation polymerization, suspension polymerization, and emulsion polymerization. MIPs can also be made into membranes for the adsorption and extraction of related substances. 20
The samples that are loaded on the MISPE cartridge are mostly aqueous solutions rather than pure organic solvent extracts. 53 The residual water on the cartridge from the samples affects the specific binding ability of the MIP, greatly reducing its ability to recognize target analytes. Therefore, MIPs should be more stable and work well in polar solutions. The washing process is a key step in the SPE. In the washing process, the analytes remain on the column due to the specific selectivity, while other unwanted substances are washed out. Therefore, the combination of MIPs and analytes should be strong enough to prevent damage from the washing process. Finally, the analyte is eluted with an appropriate solvent.
Most MISPEs are combined with HPLC. Rui et al. 17 prepared a highly selective surface molecularly imprinted polymer (FDU-12@MIPs) which could be used as an SPE sorbent coupled with HPLC for the detection of AFs G2, G1, B2, and B1 in food samples. The specific operation is as follows: 400 mg of FDU-12@MIPs are packed into an SPE cartridge. The cartridge is activated and conditioned with a suitable solvent. A diluted 50-mL sample of the extract is transferred to an MISPE column. The cartridge is then washed with 10 mL of water to remove unwanted substances. Finally, the AFs are eluted with 3 mL of methanol into a cuvette. The eluent is slowly blown to near-dryness with nitrogen at 50 °C for HPLC analysis. Under the optimized conditions, recoveries from the spiked cereals are 82.6%–116.7%, with relative standard deviations ranging from 2.73 to 4.21. The detection limits for AFs G2, G1, B2, and B1 were estimated to be 0.05, 0.06, 0.06, and 0.05 μg kg−1, respectively.
The combination of MIPs and a capillary column can reduce the amount of filler and further reduce associated costs. Three main forms of MIPs are used in a capillary columns: block, film, and nanoparticles. The bulk is mostly formed by in situ polymerization. Liang et al. 54 prepared a carbon QDs-coated dummy molecularly imprinted polymer (CD-DMIP) monolithic column which was applied to determine AF B1 amounts with HPLC-FLD. The CD-DMIP monolithic capillary column is activated with 80 μL of methanol and 80 μL of water. The injected CD-DMIP monolithic capillary column is washed with 40 μL of toluene and eluted with 100 μL of methanol–acetic acid (9:1, v/v) at the same flow rate. The eluent is collected and evaporated under nitrogen to dryness and then reconstituted with 0.1 mL of methanol for further HPLC detection. Satisfactory linearity was obtained over 0.5–2000 ng mL−1, with a high correlation coefficient of 0.9999. The recoveries of AF B1 in peanut sample ranged from 79.5% to 91.2%. The limit of detection (S/N = 3) and limit of quantitation (S/N = 10) were 0.118 and 0.393 ng mL−1, respectively. This monolithic capillary column saves time with regards to filling the column and simplifies the column loading procedure.
Biosensors
The above methods (SPE-HPLC) for AF analysis usually require complicated sample pretreatment and expensive instruments and only allow the analysis of targets at relatively high concentrations. Biosensors have received considerable attention because of their promising potential in efficient and rapid AF analysis. 55
The application of MIPs in the sensor field is an important aspect of molecularly imprinted technology. The outstanding characteristic of MIPs is that they have high selectivity for specific analytes. When the MIPs are combined with template molecules, a physical or chemical signal is generated. The converter transforms this signal into a quantitative output signal, and the real-time determination of the molecule to be analyzed is realized by monitoring the output signal. Due to the excellent specificity and environmental tolerance of MIPs, they are promising materials for the development of a variety of sensor-sensitive membranes. In 1987, Tabushi et al. 56 employed MIPs as sensitive materials to determine vitamin K1 and K2 concentrations for the first time. Since then, MIPs have been widely applied in the field of electrochemical sensing.57–59 At present, the applications of AF MIPs for use as sensors include luminescence probes, electrochemical sensors, and QCMs.
Guo et al. 39 developed a novel and sensitive fluorescent sensor for the rapid and specific recognition of AF B1 in which MITs were combined with QD technology. Under the optimum conditions, the relative fluorescence intensity of the MIP@CdTe QDs showed adequate linearity with the AF B1 concentration at 80–400 ng g−1. The detection limit is 4 ng g−1, according to 3 s K−1. Finally, the method was successfully applied to the quantitative detection of AF B1 in real samples. The method developed for AF B1 detection lays the foundation for the rapid detection of trace amounts of other harmful substances in a complicated matrix.
The combination of electrochemical detection technology and molecular imprinting technology greatly improves the detection ability. Electro-deposition and electro-polymerization are two typical methods used for the construction or electrochemical sensors. Wang et al.
8
developed a sensitive and selective imprinted electrochemical sensor for the determination of AF B1 by modifying a glassy carbon electrode with functional multi-walled carbon nanotubes (MCNTs), Au/Pt bimetallic nanoparticles (Au/PtNPs), and imprinted films. A multi-walled carbon nanotube nanocomposite film is prepared using a controllable electro-deposition technology to graft Au/Pt metals onto uniformly porous poly-
Mass sensitive sensors, especially QCMs, have been developed rapidly. A QCM sensor consists of a thin quartz disk with electrodes on both sides. When the mass changes, a piezoelectric effect is generated and the disk vibrates. The molecularly imprinted film is coated on both sides of the disk, and the target is enriched on the disk. This results in a change in mass which is converted into a change in the oscillation frequency. This is then used to detect the frequency signal. Gu et al.
61
constructed a QCM-sensing platform for the determination of AF B1 by electropolymerizing an
Surface-enhanced Raman detection
Surface-enhanced Raman spectroscopy (SERS) technology is an emerging surface spectroscopic technique.62,63 Its use has attracted significant interest because of its multiple advantages, including its high sensitivity, rapid readout speed, low susceptibility to environmental factors, and the possibility of use for on-site or field detection. 64 However, a number of questions need to be addressed before proving their practical application, such as how to achieve a reliable SERS signal, how to reduce costs, and how to avoid interference from the complex sample matrix. 65 A combination of SERS and MIPs is expected to solve this problem. Zhang et al. 66 reported a surface-enhanced gold nanoparticle Raman analysis and detection method for AF B1. Gold nanoparticles were modified by an AF B1 imprinted polymer to prepare a new material in which nanogold was used as the core and MIPs as a shell. The cavities of the MIP shell can specifically capture AF B1; AF B1 remains close to the surface of the gold nanoparticles and is located in the effective range of the gold nanoparticle plasmon magnetic resonance field. Therefore, an SERS signal is generated allowing for the effective detection of AF B1. The recovery rate for AF B1 ranges from 89.84% to 101.12%, and the RSD is acceptable, ranging from 3.51% to 15.44%.
Outlook
In summary, MIPs for the detection of AF have been well studied. They can be used as SPE absorbents and sensor tools for online enrichment and detection of AF in food. However, their use faces several challenges. For example, AFs are highly toxic and very expensive, and they are not a suitable template molecule for the preparation of MIPs. Although several pseudo-template molecules have been developed, most of these molecules cannot be easily prepared and are expensive. The selectivity of the corresponding MIPs are not sufficient for practical detection. The design and development of inexpensive and effective pseudo-template molecules is still needed. Presently, the procedure for the removal of template molecules from the MIPs is complex and requires a large amount of organic solvent. Thus, further exploration of effective post-treatment technologies for MIPs is required. It is worth noting that a combination of MIT with nanotechnology and advanced spectral technology for the detection of AF is a promising research field.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the Project of Guangzhou Science and Technology (No. 201806010127).
