Abstract
An efficient approach for the synthesis of pyrazoline/pyrazole-tethered pyridinyl methanones is described via a one-pot, stepwise, sequential methodology using chalcones and pyridine-4-carbohydrazide as substrates through a Michael addition followed by cyclization. The reaction proceeds via a catalyst-, solvent-, work-up-, and column-chromatography-free method under melt conditions to provide the pyrazolines in short reaction times with high atom efficiency.
An efficient construction of pyrazoline-based pyridinyl frameworks through Michael addition mediated domino cyclization of chalcones with hydrazides. The two-step procedure generates four new C–C, C=C, C–N, and C=N bonds with regioselectivity and high atom efficiency in excellent yields. Interestingly, the accomplishment of this reaction under melt condition, the catalyst-, solvent-free synthesis in short time provided excellent yields is highly impressive.
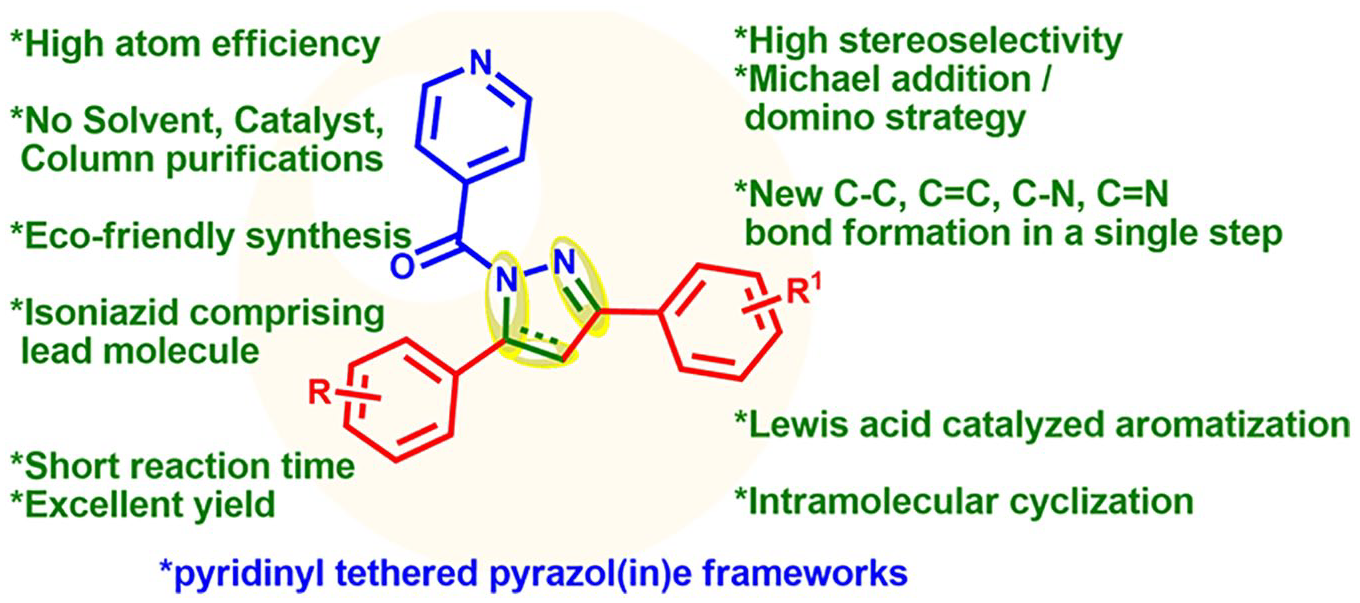
Introduction
The synthesis of nitrogen-containing heterocyclic molecules through catalyst-, solvent-, and column-chromatography-free conditions via melt-mediated reactions 1 in the solid state is very useful.2,3 Chalcones and pyridine-4-carbohydrazide (known as isoniazid) possess significant medicinal properties4,5 and isoniazid is a valuable drug for tuberculosis (TB). 6 Pyrazoles exhibit a range of potent activities such as antibacterial, antifungal, 7 antidiabetic, 8 and anti-inflammatory 9 behavior, and are also active against many mycobacteria.10,11 Examples of biologically active chalcones, pyridine-4-carbohydrazides, and pyrazoline/pyrazole motifs12–15 are shown in Figure 1. α,β-Unsaturated carbonyl compounds, such as chalcones, are particularly useful for their medicinal applications due to their easy synthesis and wide-ranging pharmacological applications against many human diseases such as TB, 16 cancer, and HIV.17,18 Significantly, the design of new drugs containing diverse pharmacophores in a single molecular scaffold may lead to new hybrid compounds with interesting biological profiles. By implementing this strategy, several research groups have developed numerous hybrid molecules by coupling chalcones with various bioactive molecules.19–22 Based on this approach, we have constructed pyrazolyl-tethered (pyridin-4-yl)methanones that have both pyridinyl and pyrazolyl motifs in a single molecular framework that may have biological significance. 23
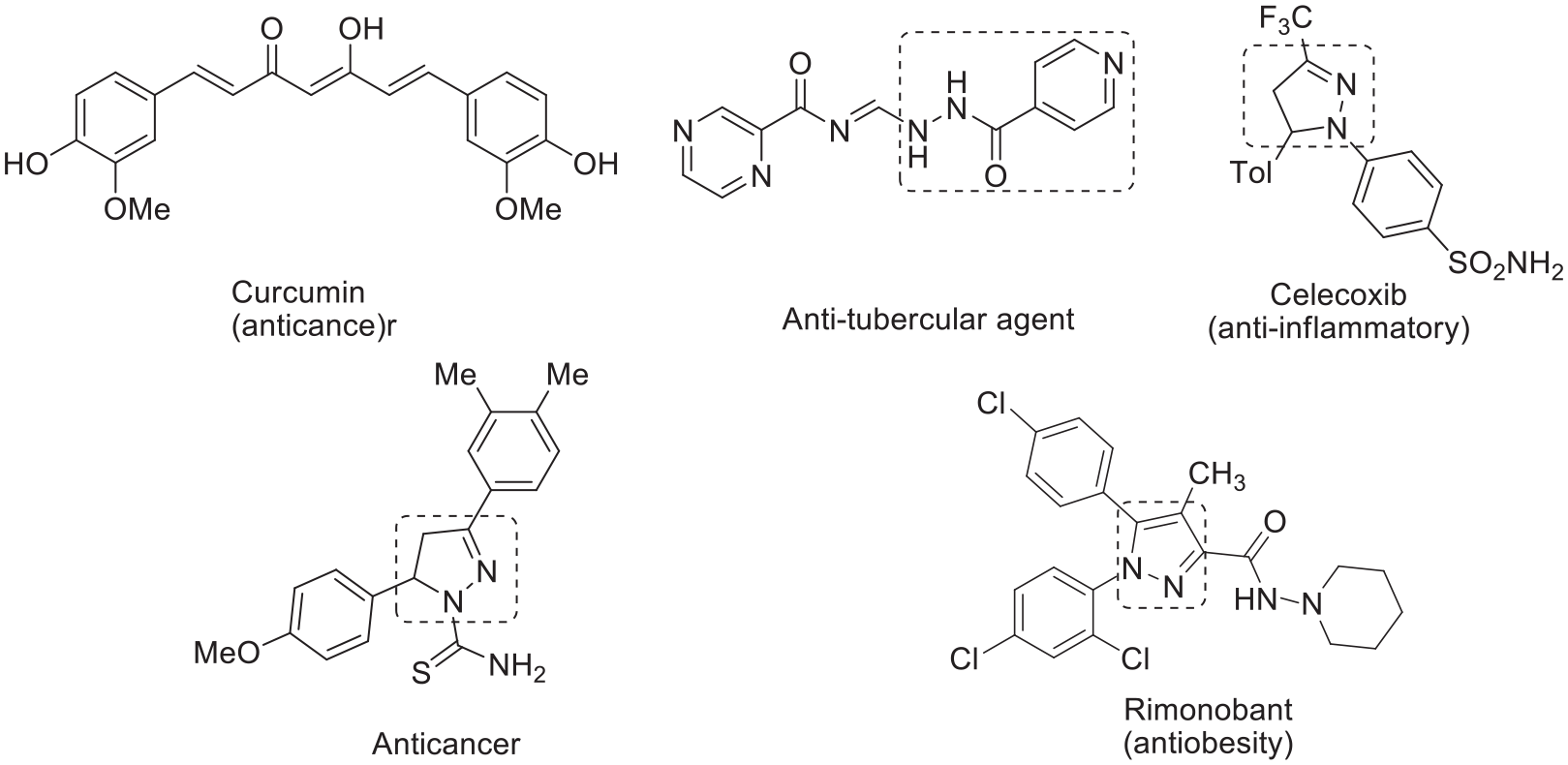
Representative examples of chalcone-, isoniazid-, pyrazoline- and pyrazole unit comprising potential molecules.
Thus, we envisaged synthesizing the pyrazole-based compounds by a Michael addition initiated domino cyclization. In continuation of our preliminary research studies24–27 and inspired by the efficient synthetic protocol accomplished by the Bakthadoss Research Group involving a solid-state melt reaction (SSMR),2,3 we have utilized the aforementioned SSMR protocol for the synthesis of pyrazole-containing molecules.
Results and discussion
Thus, we combined the substituted chalcone
Chalcones
Synthesis of pyrazolines

All products were fully characterized by spectroscopic analysis.
Isolated yields of pure products.

Synthesis of
We then decided to broaden the ring size, so we reacted o-phenylenediamine (OPDA) (
In order to extend the applicability of the reaction, the pyrazolines
Synthesis of pyrazole derivatives

All products were fully characterized by spectroscopic analysis.
Isolated yields of pure products.
The structures of the compounds

ORTEP diagrams of compounds
Conclusion
We have successfully constructed pyrazolines and pyrazoles in a one-pot, stepwise approach with high regioselectivity. Some of the notable features are as follows: (1) the reaction creates new C–C, C=C, C–N, and C=N bonds in a unique fashion through a domino process that includes a Michael addition followed by a cyclization; (2) the reactions proceed under eco-friendly, solvent-free reaction conditions via a work-up-, catalyst-, chromatography-free method that provides excellent yields in short reaction times; (3) this protocol provides the opportunity for the synthesis of libraries of tri-substituted pyrazol(in)es with high atom efficiency (>96%); and (4) the structures of the newly synthesized molecules have been characterized by IR, NMR, and mass spectrometry, and by single-crystal X-ray analyses. These pyrazoline/pyrazole products are currently undergoing biological screening.
Experimental
Materials and methods
All reagents and chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA) and Merck (Bangalore, Karnataka, India).
Physical and chemical characterization
A Perkin Elmer fourier transform infrared spectroscopy (FTIR) (4000-400 cm−1) instrument was used to record the IR spectra as KBr pellets. 1H and 13C NMR spectra were obtained at 500 and 125 MHz (BRUKER AV-ΙΙΙ, fourier-transform nuclear magnetic resonance (FT-NMR) spectrometer) using CDCl3 as the solvent and tetramethylsilane (TMS) as an internal standard. The DEPT135 spectrum was recorded in a standard manner (θ = 135 pulse program). Mass spectra were recorded on a Thermo Scientific Orbitrap Elite mass spectrometer. The XRD studies were conducted on a Bruker Kappa APE XII diffractometer. Thin-layer chromatography (TLC) was performed using pre-coated silica gel sheets, and the spots were observed by ultraviolet (UV) and iodine vapor absorption.
Synthesis of chalcones 3a
The aldehyde (1 mmol) and substituted acetophenone (1 mmol) were dissolved in ethanol (20 mL) in the presence of 40% NaOH. The reaction mixture was stirred at room temperature for 4 h and neutralized with 1N HCl. The resulting precipitate was filtered, and the crude sample was washed with hot ethanol and dried.24,25
Synthesis of isoniazid-containing pyrazoline 5a : Typical procedure
A mixture of substituted chalcone
Synthesis of isoniazid-containing pyrazole 8a
A solution of the pyrazoline
(3-(4-ethoxyphenyl)-5-(4-(methylthio)phenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5a )
Mp = 145–146 °C; Rf = 0.40 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3042, 2974, 2924, 1649; 1H NMR (500 MHz, CDCl3) δ 8.76 (d, J = 6.1 Hz, 2H), 7.86 (d, J = 6.0 Hz, 2H), 7.65 (d, J = 8.8 Hz, 2H), 7.28 (d, J = 9.8 Hz, 2H), 7.26 (d, J = 2.2 Hz, 2H), 6.97 – 6.92 (m, 2H), 5.75 (dd, J1,2 = 11.6, 4.7 Hz, 1H), 4.10 (q, J = 7.0 Hz, 2H), 3.80 (dd, J1,2 = 17.6, 11.6 Hz, 1H), 3.23 (dd, J1,2 = 14.0, 3.6 Hz, 1H), 2.48 (s, 3H), 1.46 (t, J = 7.0 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ 161.17, 156.74, 149.67, 141.81, 138.26, 138.06, 128.52, 127.19, 126.35, 123.75, 122.53, 117.82, 114.76, 63.71, 60.76, 41.68, 15.94, 14.69; HRMS (ESI): m/z [M]+ calculated for C24H23N3O2S: 417.1511; found: 417.1540.
(5-(4-(methylthio)phenyl)-3-(3-nitrophenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5b )
Mp = 153–154 °C; Rf = 0.27 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3043, 2919, 1663, 1532; 1H NMR (500 MHz, CDCl3) δ 8.81 (d, J = 6.0 Hz, 2H), 8.44 (t, J = 1.8 Hz, 1H), 8.32 (ddd, J1,2,3 = 8.2, 2.1, 0.8 Hz, 1H), 8.10 (d, J = 7.9 Hz, 1H), 7.89 (d, J = 5.7 Hz, 2H), 7.66 (t, J = 8.0 Hz, 1H), 7.28 (d, J = 9.6 Hz, 4H), 5.84 (dd, J1,2 = 11.8, 5.0 Hz, 1H), 3.91 (dd, J1,2 = 17.9, 11.8 Hz, 1H), 3.33 (dd, J1,2 = 17.9, 5.1 Hz, 1H), 2.48 (s, 3H); 13C NMR (126 MHz, CDCl3) δ 164.08, 154.03, 148.62, 142.54, 138.99, 137.18, 132.53, 132.21, 130.14, 130.06, 127.19, 126.25, 125.26, 124.02, 121.71, 61.32, 41.59, 15.71; HRMS (ESI): m/z [M]+1 calculated for C22H18N4O3S: 418.1100; found: 419.1187.
(3-(4-fluorophenyl)-5-(4-(methylthio)phenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5c )
Mp = 156–157 °C; Rf = 0.44 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3034, 2993, 2946, 2921, 1644, 1328; 1H NMR (500 MHz, CDCl3) δ 8.77 (d, J = 5.8 Hz, 2H), 7.86 (d, J = 5.8 Hz, 2H), 7.73 – 7.68 (m, 2H), 7.29 – 7.23 (m, 4H), 7.16 – 7.11 (m, 2H), 5.77 (dd, J1,2 = 11.7, 4.9 Hz, 1H), 3.82 (dd, J1,2 = 17.8, 11.7 Hz, 1H), 3.24 (dd, J1,2 = 17.8, 4.9 Hz, 1H), 2.47 (s, 3H); 13C NMR (126 MHz, CDCl3) δ 164.28 ( 1 JC–F = 253.2 Hz), 163.98, 163.28, 155.11, 149.07, 142.30, 138.59, 137.75, 128.93 ( 3 JC–F = 8.8 Hz),127.16, 127.05 ( 4 JC–F = 2.52 Hz), 126.31, 123.89, 116.12 ( 2 JC–F = 22.6 Hz), 116.03, 60.99, 41.72; HRMS (ESI): m/z [M]+ calculated for C22H18FN3OS: 391.1155; found: 391.1180.
(3-(3,4-dimethoxyphenyl)-5-(4-(methylthio)phenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5d )
Mp = 140–141 °C; Rf = 0.18 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3063, 2922, 1733, 1647; 1H NMR (500 MHz, CDCl3) δ 8.80 (d, J = 4.8 Hz, 2H), 8.01 (d, J = 5.5 Hz, 2H), 7.28 (dd, J1,2 = 7.1, 3.9 Hz, 5H), 7.20 (dd, J1,2 = 8.3, 1.9 Hz, 1H), 6.91 (dd, J1,2 = 8.4, 2.0 Hz, 1H), 5.76 (dd, J1,2 = 11.6, 4.7 Hz, 1H), 3.95 (s, 3H), 3.92 (s, 3H), 3.84 (dd, J1,2 = 17.7, 11.6 Hz, 1H), 3.27 (dd, J1,2 = 17.7, 4.7 Hz, 1H), 2.48 (s, 3H); 13C NMR (101 MHz, CDCl3) δ 162.93, 157.74, 151.83, 149.34, 148.99, 142.66, 141.22, 137.69, 127.16, 126.36, 124.72, 123.35, 120.99, 110.84, 108.90, 60.94, 55.99, 55.95, 41.78, 15.76; HRMS (ESI): m/z [M]+ calculated for C24H23N3O3S: 433.1460; found: 433.1490.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(4-fluorophenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5e )
Mp = 143–144 °C; Rf = 0.46 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3065, 3043, 2945, 2922, 1749, 1646, 1332; 1H NMR (500 MHz, CDCl3) δ 8.75 (d, J = 5.4 Hz, 2H), 7.83 (d, J = 5.8 Hz, 2H), 7.69 (dd, J1,2 = 8.7, 5.3 Hz, 2H), 7.12 (t, J = 8.5 Hz, 2H), 6.83 (dd, J1,2 = 8.0, 1.1 Hz, 1H), 6.78 (d, J = 7.9 Hz, 2H), 5.93 (s, 2H), 5.72 (dd, J1,2 = 11.6, 4.8 Hz, 1H), 3.78 (dd, J1,2 = 17.8, 11.7 Hz, 1H), 3.21 (dd, J1,2 = 17.8, 4.8 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 164.24 ( 1 JC–F = 253.2 Hz), 164.19, 155.00, 149.49, 148.29, 147.38, 141.90, 135.05, 128.91 ( 3 JC–F = 8.8 Hz), 127.12 ( 4 JC–F = 2.5 Hz), 123.72, 119.36, 116.26, 116.08 ( 2 JC–F = 22.6 Hz), 108.65, 106.00, 101.26, 61.18, 41.85; HRMS (ESI): m/z [M]+1 calculated for C22H16FN3O3: 389.1176; found: 390.1251.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3-nitrophenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5f )
Mp = 132–133 °C; Rf = 0.29 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3055, 3028, 2928, 1770, 1644, 1527; 1H NMR (500 MHz, CDCl3) δ 8.81 (d, J = 5.5 Hz, 2H), 8.43 (s, 1H), 8.35 – 8.30 (m, 1H), 8.10 (d, J = 7.8 Hz, 1H), 7.89 (d, J = 5.0 Hz, 2H), 7.66 (t, J = 8.0 Hz, 1H), 6.87 – 6.76 (m, 3H), 5.97 (s, 2H), 5.80 (dd, J1,2 = 11.7, 4.9 Hz, 1H), 3.89 (dd, J1,2 = 17.9, 11.8 Hz, 1H), 3.32 (dd, J1,2 = 17.9, 5.0 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 164.10, 154.00, 148.62, 148.43, 147.62, 142.58, 134.40, 132.55, 132.19, 130.25, 130.05, 125.25, 124.02, 121.71, 119.41, 108.78, 105.91, 101.36, 61.53, 41.74; HRMS (ESI): m/z [M]+1 calculated for C22H16N4O5: 416.1121; found: 417.1198.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(2-chlorophenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5g )
Mp = 124–125 °C; Rf = 0.46 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3031, 2919, 1713, 1644; 1H NMR (500 MHz, CDCl3) δ 8.73 (d, J = 4.7 Hz, 2H), 7.88 (d, J = 5.8 Hz, 2H), 7.65 (d, J = 7.7 Hz, 1H), 7.44 (dd, J1,2 = 7.9, 1.2 Hz, 1H), 7.35 (td, J = 7.7, 1.8 Hz, 1H), 7.33 – 7.28 (m, 1H), 6.85 (d, J = 8.0 Hz, 1H), 6.81 – 6.77 (m, 2H), 5.93 (s, 2H), 5.71 (dd, J1,2 = 11.6, 4.6 Hz, 1H), 3.98 (dd, J1,2 = 18.3, 11.7 Hz, 1H), 3.39 (dd, J1,2 = 18.3, 4.7 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 164.18, 155.64, 148.76, 148.27, 147.40, 142.22, 134.82, 133.04, 131.33, 131.16, 130.44, 129.86, 127.12, 124.08, 119.44, 108.64, 106.07, 101.25, 61.29, 44.52; HRMS (ESI): m/z [M]+ calculated for C22H16ClN3O3: 405.0880; found: 405.0910.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(4-ethoxyphenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5h )
Mp = 129–130 °C; Rf = 0.30 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3065, 3045, 2977, 2918, 1647; 1H NMR (500 MHz, CDCl3) δ 8.77 (d, J = 5.9 Hz, 2H), 7.92 (d, J = 5.6 Hz, 2H), 7.64 (d, J = 8.8 Hz, 2H), 6.94 (d, J = 8.8 Hz, 2H), 6.85 (dd, J1,2 = 8.0, 1.6 Hz, 1H), 6.81 – 6.77 (m, 2H), 5.95 (s, 2H), 5.70 (dd, J1,2 = 11.5, 4.6 Hz, 1H), 4.09 (q, J = 7.0 Hz, 2H), 3.78 (dd, J1,2 = 17.7, 11.6 Hz, 1H), 3.22 (dd, J1,2 = 17.7, 4.7 Hz, 1H), 1.46 (t, J = 7.0 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ 163.55, 161.23, 156.09, 148.67, 148.26, 147.32, 142.85, 135.17, 128.54, 124.15, 123.10, 119.42, 114.77, 108.63, 106.06, 101.23, 63.71, 61.00, 41.85, 14.68; HRMS (ESI): m/z [M]+1 calculated for C24H21N3O4: 415.1532; found: 416.1612.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3-bromophenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5i )
Mp = 167–168 °C; Rf = 0.38 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3027, 3055, 2918, 1778, 1645; 1H NMR (500 MHz, CDCl3) δ 8.80 (d, J = 5.6 Hz, 2H), 7.90 (d, J = 5.6 Hz, 2H), 7.81 (s, 1H), 7.64 (d, J = 7.8 Hz, 1H), 7.60 (ddd, J1,2,3 = 7.9, 1.8, 0.8 Hz, 1H), 7.33 (t, J = 7.9 Hz, 1H), 6.86 – 6.76 (m, 3H), 5.96 (s, 2H), 5.74 (dd, J1,2 = 11.7, 4.8 Hz, 1H), 3.80 (dd, J1,2 = 17.9, 11.7 Hz, 1H), 3.23 (dd, J1,2 = 17.9, 4.9 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 163.88, 154.88, 148.51, 148.36, 147.50, 142.79, 134.68, 133.80, 132.75, 130.42, 129.78, 125.37, 124.11, 123.06, 119.40, 108.71, 105.97, 101.30, 61.26, 41.71; HRMS (ESI): m/z [M]+1 calculated for C22H16BrN3O3: 449.0375; found: 450.0454.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3-methoxyphenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5j )
Mp = 141–142 °C; Rf = 0.29 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3063, 2912, 1731, 1643; 1H NMR (500 MHz, CDCl3) δ 8.76 (d, J = 5.9 Hz, 2H), 7.86 (d, J = 6.0 Hz, 2H), 7.35 (t, J = 7.9 Hz, 1H), 7.29 – 7.23 (m, 2H), 7.01 (dd, J1,2 = 8.1, 2.5 Hz, 1H), 6.84 (dd, J1,2 = 8.0, 1.3 Hz, 1H), 6.81 – 6.77 (m, 2H), 5.94 (s, 2H), 5.71 (dd, J1,2 = 11.6, 4.7 Hz, 1H), 3.84 (s, 3H), 3.79 (dd, J1,2 = 17.8, 11.7 Hz, 1H), 3.22 (dd, J1,2 = 17.8, 4.8 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 164.13, 159.83, 156.03, 149.27, 148.27, 147.36, 142.05, 135.08, 132.11, 129.94, 123.88, 123.10, 119.46, 116.44, 112.18, 108.64, 106.05, 101.24, 61.14, 55.37, 41.87; HRMS (ESI): m/z [M]+ calculated for C23H19N3O4: 401.1376; found: 401.1405.
(3-(3-aminophenyl)-5-(benzo[d][1,3]dioxol-5-yl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5k )
Mp = 181–182 °C; Rf = 0.11 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3327, 3027, 2987, 2922, 1649; 1H NMR (500 MHz, CDCl3) δ 8.76 (d, J = 5.9 Hz, 2H), 7.85 (d, J = 5.5 Hz, 2H), 7.22 (t, J = 7.8 Hz, 1H), 7.08 – 7.01 (m, 2H), 6.84 (dd, J1,2 = 8.0, 1.5 Hz, 1H), 6.80 – 6.77 (m, 3H), 5.95 (s, 2H), 5.69 (dd, J1,2 = 11.6, 4.7 Hz, 1H), 3.76 (dd, J1,2 = 17.8, 11.6 Hz, 1H), 3.28 (s, 2H), 3.20 (dd, J1,2 = 17.8, 4.7 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 164.11, 156.38, 149.40, 148.25, 147.32, 146.79, 142.09, 135.16, 131.73, 129.77, 123.83, 119.39, 117.72, 117.44, 112.51, 108.62, 106.06, 101.23, 61.03, 41.88; HRMS (ESI): m/z [M]+ calculated for C22H18N4O3: 386.1379; found: 386.1408.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3,4-dimethoxyphenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5l )
Mp = 122–123 °C; Rf = 0.14 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3003, 2955, 2925, 1638, 1332; 1H NMR (500 MHz, CDCl3) δ 8.79 (d, J = 5.9 Hz, 2H), 7.99 (d, J = 6.0 Hz, 2H), 7.29 (s, 1H), 7.19 (dd, J = 8.3, 2.0 Hz, 1H), 6.91 (d, J = 8.4 Hz, 1H), 6.86 (dd, J1,2 = 8.0, 1.6 Hz, 1H), 6.81 (s, 1H), 6.80 – 6.78 (m, 1H), 5.96 (s, 1H), 5.72 (dd, J1,2 = 11.5, 4.6 Hz, 1H), 3.95 (s, 1H), 3.91 (s, 1H), 3.81 (dd, J1,2 = 17.7, 11.6 Hz, 1H), 3.25 (dd, J1,2 = 17.7, 4.7 Hz, 1H); 13C NMR (126 MHz, CDCl3) δ 163.02, 156.50, 151.80, 149.32, 148.32, 147.43, 147.35, 144.18, 134.90, 124.64, 123.40, 120.97, 119.47, 110.83, 108.89, 108.67, 106.03, 101.28, 77.28, 77.02, 76.77, 61.14, 56.04, 55.98, 41.92; m/z [M]+ calculated for C24H21N3O5: 431.1481; found: 431.1512.
(5-(2-(benzyloxy)phenyl)-3-(4-fluorophenyl)-4,5-dihydro-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (5m )
Mp = 148–149 °C; Rf = 0.45 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3057, 2983, 2919, 1725, 1607; 1H NMR (400 MHz, CDCl3) δ 8.64 (d, J = 5.9 Hz, 2H), 7.68 (dd, J1,2 = 4.5, 1.6 Hz, 2H), 7.51 – 7.45 (m, 2H), 7.29 – 7.26 (m, 2H), 7.24 – 7.16 (m, 5H), 7.02 – 6.87 (m, 4H), 5.92 (dd, J1,2 = 11.8, 5.5 Hz, 1H), 5.01 (s, 2H), 3.63 (dd, J1,2 = 17.6, 11.9 Hz, 1H), 3.13 (dd, J1,2 = 17.6, 5.6 Hz, 1H); 13C NMR (101 MHz, CDCl3) δ 163.18, 163.01 ( 1 JC–F = 252.5 Hz), 154.60, 154.29, 148.60, 140.90, 135.43, 128.15, 127.73 ( 3 JC–F = 8.0 Hz), 127.48, 127.32, 127.09, 126.67, 126.43, 126.37 ( 4 JC–F = 6.0 Hz), 122.72, 120.04, 114.79 ( 2 JC–F = 22.2 Hz), 111.22, 69.29, 57.34, 39.51; HRMS (ESI): m/z [M]+ calculated for C28H22FN3O2: 451.1696; found: 451.1422.
2-(2-(benzyloxy)phenyl)-1H-benzo[d]imidazole (6 )
Mp = 142–144 °C; Rf = 0.35 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 1346, 1680, 2998, 3174; 1H NMR (500 MHz, CDCl3): δ 5.20 (s, 2H), 7.04 – 8.61 (m, 13H), 10.63 (br s, 1H); 13C NMR (125 MHz, CDCl3): δ 71.37, 112.96, 118.31, 122.09, 122.56, 127.84, 128.87, 129.11, 130.23, 131.22, 135.99, 149.79, 156.13; HRMS (ESI): m/z [M]+1 calculated for C20H16N2O: 300.1263; found: 301.1408.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(4-fluorophenyl)-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (8a )
Mp = 112–113 °C; Rf = 0.28 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3034, 2922, 1734, 1635; 1H NMR (400 MHz, CDCl3) δ 8.86 – 6.77 (m, 12H), 5.93 (s, 2H); 13C NMR (101 MHz, CDCl3) δ 164.31 ( 1 JC–F = 254.5 Hz), 164.21, 155.14, 149.58, 148.40, 147.45, 145.51, 142.01, 135.16, 128.93 ( 3 JC–F = 8.8 Hz), 127.13 ( 4 JC–F = 3.7 Hz), 123.91, 119.49, 116.08 ( 1 JC–F = 21.4 Hz), 108.87, 106.19, 103.71, 101.49; HRMS (ESI): m/z [M]+ calculated for C22H14FN3O3: 387.1019; found: 387.1009.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3-nitrophenyl)-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (8b )
Mp = 126–127 °C; Rf = 0.24 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3212, 3116, 3036, 2922, 2854, 1649, 1595; 1H NMR (400 MHz, CDCl3) δ 8.93 – 6.93 (m, 12H), 5.97 (s, 2H); 13C NMR (101 MHz, CDCl3) δ 164.47, 154.66, 148.99, 148.51, 147.85, 144.93, 142.79, 132.68, 134.52, 132.30, 130.25, 125.46, 124.28, 123.47, 121.87, 119.64, 109.23, 106.65, 104.06, 101.96; HRMS (ESI): m/z [M]+ calculated for C22H14N4O5: 414.0964; found: 414.0904.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(2-chlorophenyl)-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (8c )
Mp = 130–131 °C; Rf = 0.24 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3058, 3020, 2959, 2923, 1724, 1642; 1H NMR (400 MHz, CDCl3) δ 8.96 – 7.05 (m, 12H), 5.93 (s, 2H); 13C NMR (101 MHz, CDCl3) δ 164.37, 155.82, 148.90, 148.35, 147.48, 145.97, 142.30, 134.70, 133.20, 131.44, 131.22, 130.53, 129.92, 127.46, 124.30, 119.64, 108.79, 106.21, 104.04, 101.36; HRMS (ESI): m/z [M]+ calculated for C22H14ClN3O3: 403.0724; found: 403.2350.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(4-ethoxyphenyl)-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (8d )
Mp = 116–117 °C; Rf = 0.45 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3236, 3064, 3033, 2919, 2851, 1782, 1611; 1H NMR (400 MHz, CDCl3) δ 8.90 – 6.91 (m, 12H), 5.95 (s, 2H), 4.10 (q, J = 10.0 Hz, 2H), 1.46 (t, J = 5.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ 163.51, 161.23, 156.21, 148.92, 148.15, 147.49, 145.39, 142.69, 135.17, 128.79, 124.33, 123.29, 119.65, 115.02, 108.91, 106.34, 104.16, 101.42, 64.08, 15.17; HRMS (ESI): m/z [M]+ calculated for C24H19N3O4: 413.1376; found: 413.2657.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3-bromophenyl)-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (8e )
Mp = 135–136 °C; Rf = 0.41 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3063, 2957, 2922, 1684, 1611; 1H NMR (400 MHz, CDCl3) δ 8.90 – 6.87 (m, 12H), 5.96 (s, 2H); 13C NMR (101 MHz, CDCl3) δ 164.10, 155.07, 148.62, 148.43, 147.46, 144.92, 142.85, 134.90, 133.92, 132.94, 130.62, 129.57, 125.51, 124.22, 123.25, 119.58, 108.84, 106.23, 104.48, 101.47; HRMS (ESI): m/z [M]+ calculated for C22H14BrN3O3: 447.0219; found: 448.1430.
(5-(benzo[d][1,3]dioxol-5-yl)-3-(3-methoxyphenyl)-1H-pyrazol-1-yl)(pyridin-4-yl)methanone (8f )
Mp = 108–109 °C; Rf = 0.30 (hexane/EtOAc, 1:1); IR (KBr, cm−1): 3062, 2947, 2928, 1681, 1614; 1H NMR (400 MHz, CDCl3) δ 8.93 – 6.85 (m, 12H), 5.94 (s, 2H), 3.84 (s, 3H); 13C NMR (101 MHz, CDCl3) δ 164.21, 159.90, 156.13, 149.32, 148.36, 147.42, 145.26, 142.13, 135.13, 132.20, 130.00, 123.99, 119.56, 119.34, 116.54, 112.29, 108.74, 106.16, 104.27, 101.36, 55.37; HRMS (ESI): m/z [M]+ calculated for C23H17N3O4: 399.1219; found: 399.1418. The 1H and 13C NMR spectra, MS data for
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820977165 – Supplemental material for An expedient, one-pot, stepwise sequential approach for the regioselective synthesis of pyrazolines
Supplemental material, sj-pdf-1-chl-10.1177_1747519820977165 for An expedient, one-pot, stepwise sequential approach for the regioselective synthesis of pyrazolines by Suresh Ganesan, Muniraj Sarangapani and Mukesh Doble in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the Sophisticated Analytical Instrumentation Facility (SAIF), Indian Institute of Technology Madras (IITM), for analytical support. S.G. thanks the Council of Scientific and Industrial Research, New Delhi, for his Senior Research Fellowship (Project No. 08/508 (0001)/2013-EMR-I).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Council of Scientific and Industrial Research, New Delhi.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


