Abstract
Hydroxy-α-sanshool was synthesized in a 13% overall yield through eight steps, which included two Wittig reactions that were used to form the carbon skeleton with ethyl 2-oxoacetate and 2E,4E-hexadienal being reacted with the appropriate ylides. Impurities in the processes could easily be separated. Ethyl 6-hydroxy-2Z-hexenoate was converted to its E-isomer with catalysis by I2 and 2E,6Z,8E,10E-dodecatetraenoic acid was crystallized from a solution in 1% ethyl acetate in n-hexane.
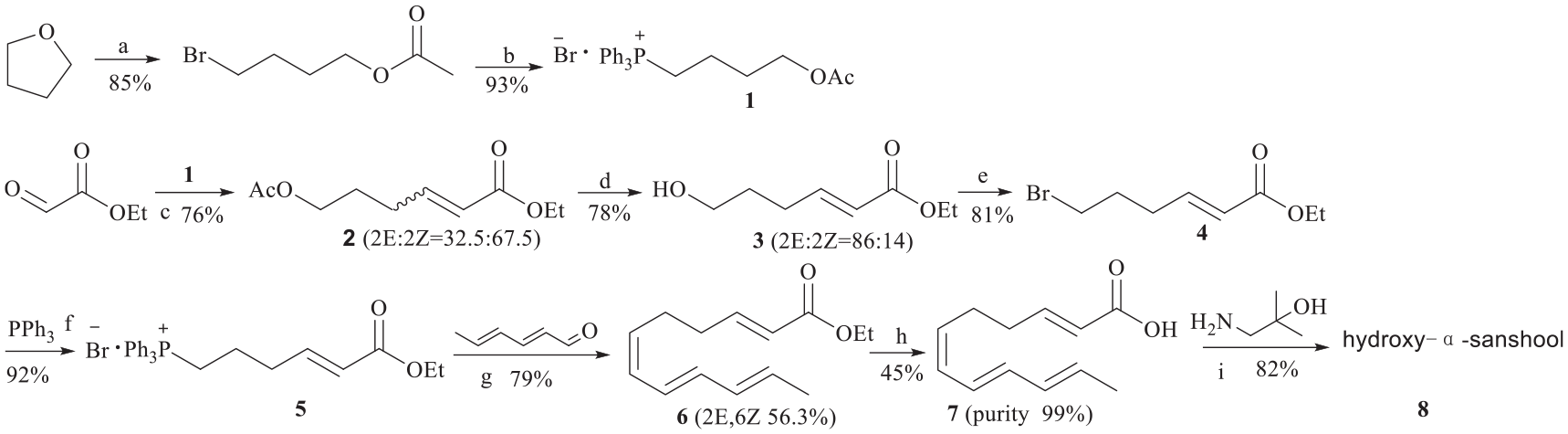
Introduction
The sanshools are representative unsaturated fatty acid amides found in Zanthoxylum (Zanthoxylum bungeanum Maxim.) and include α-sanshool, β-sanshool, γ-sanshool, δ-sanshool, and homologues containing one hydroxy group in the amino fragment (Figure 1).1–3 Hydroxy-α-sanshool is the main numb flavoring substance in Zanthoxylum, and its content directly determines the degree of numbness of Zanthoxylum, so establishing the quality of the Zanthoxylum.4,5

The sanshools.
Hydroxy-α-sanshool has been found to have several bioactivities. It acts as an agonist of transient receptor potential vanilloid type-1 (TRPV-1) and transient receptor potential ankyrin-1 (TRPA-1) 6 and is a selective blocker of some two-pore domain potassium channels (KCNK): TASK-1 (KCNK3), TASK-3 (KCNK9), and TRESK (KCNK18).7,8 The process of separating hydroxy-α-sanshool from Zanthoxylum is tedious and gives low yields. 9
Igarashi et al. 10 synthesized hydroxy-α-sanshool using a Suzuki–Miyaura coupling (SMC) (see Scheme 1). This SMC-based route involved the synthesis of complex building blocks and involved non-readily available starting materials. Sonnet et al. 11 synthesized α-sanshool using ethyl 4,6,8-decatrienoate as an intermediate although the process needs harsh conditions and involved the reduction of an acid to an aldehyde. Wu et al. 12 synthesized hydroxy-α-sanshool using 4-bromobutyraldehyde as a key intermediate. However, the starting material 4-bromo-1-butanol is unstable during its preparation, storage, and oxidation; therefore, 4-bromobutyraldehyde is difficult to prepare on a large scale.

Reported processes in the literatures
The synthesis of Wu et al. was the easiest to carry out although the preparation of ethyl 6-bromo-2E-hexenoate is not straightforward. Although 4-bromobutyraldehyde can be prepared by reducing ethyl 4-bromobutyrate, the reaction must be performed in the absence of water and at a low temperature.13–15 An alternative method is the bromination of ethyl 6-hydroxy-2E-hexenoate.16–19 6-Hydroxy-2E-hexenoate can be prepared from tetrahydrofuran-2-ol which is obtained by reduction of γ-butyrolactone using diisobutylaluminium hydride (DIBALH) albeit at an extremely low temperature or from 4-hydroxy-1-butyraldehyde. This can be obtained by oxidation of 1,4-butanediol with pyridinium chlorochromate (PCC) or MnO2,20–22 but the yield is low and the purification difficult.
Considering the difficult preparation of 4-bromobutyraldehyde or 4-hydoxybutyraldehyde, a new route starting from glyoxylic acid was designed (Scheme 2). Ethyl (E)-6-bromohex-2-enoate

Designed synthetic route in our work
Results and discussion
Chemistry
According to the literature,
23
the impurities in 4-bromobutyl acetate are 1,4-dibromobutane and 1,4-diacetoxybutane. 1,4-Diacetoxybutane can be easily removed following the preparation of the quaternary phosphonium salt
The phosphonium salt
We tried to record the nuclear magnetic resonance (NMR) spectrum of our synthetic hydroxy-α-sanshool
Conclusion
In summary, hydroxy-α-sanshool was synthesized in eight steps using two Wittig reactions as the key assembly steps. The cis-isomer of ethyl 6-hydroxyhex-2-enoate can be isomerized to the desired trans-isomer using I2 as the catalyst. 2E,6Z,8E,10E-Dodecatetraenoic acid was crystallized from a solution in 1% ethyl acetate in n-hexane. The work-ups were simple and the overall yield of the product was high. All the intermediate and target compounds were characterized by 1H NMR, 13C NMR, and MS spectra.
Experiment
All the reagents were purchased from commercial suppliers without further purification unless otherwise specified. 1H NMR and 13C NMR spectra were recorded on a Bruker Avance 400 MHz spectrometer. NMR spectra were recorded in DMSO-d6 solutions at room temperature (20 °C ± 2 °C). 1H and 13C chemical shifts are quoted in parts per million downfield from TMS. Supplemental MS spectra were recorded on a Bruker Esquire 3000 instrument. High-resolution mass spectra (HRMS) were obtained on a MicrOTOF-Q II mass spectrometer with a supplemental source (Waters, Manchester). As for known compounds, only 1H NMR and 13C NMR spectra were confirmed with previously reported literature, and the main intermediates were characterized by 1H NMR, 13C NMR spectra, and mass spectra.
4-Bromobutyl acetate
It was prepared according to the reported procedure. 23 light yellow liquid 27.6 g, yield 85%. 1H NMR (400 MHz, DMSO-d6, ppm) δ 4.03 (t, J = 6.8 Hz, 2H), 3.42 (t, J = 6.4 Hz, 2H), 2.01 (s, 3H), 1.81–1.88 (m, 2H), 1.50–1.57 (m, 2H). 13C NMR (101 MHz, DMSO-d6, ppm) δ 171.2, 67.7, 36.0, 31.6, 29.9, 25.8.
[4-(Acetyloxy)butyl]triphenylphosphonium bromide (1)
4-Bromobutyl acetate (23.5 g, 0.12 mol) and PPh3 (30 g, 0.12 mol) were added to 250 mL acetonitrile and refluxed overnight. Acetonitrile was distilled off under reduced pressure to obtain a pale yellow viscous oil. After 150 mL ether was added, the mixture was frozen for 1 h, to give a white solid, the solid was then filtered and washed with ethyl acetate and ether, respectively, to afford
(Z/E)Ethyl 6-acetoxy-2-hexenoate (2 )
To a solution of
Ethyl 6-hydroxy-2E-hex-2-enoate (3)
The strong acidic styrene type cation exchange resin 732 (15 g) was added into a solution of compound
Ethyl 6-bromo-2E-hexenoate (4)
CBr4 (4.4 g, 12.64 mmol) was added to a solution of PPh3 (13.2 g, 50.56 mmol) in 50 mL CH2Cl2 cooled in ice bath, and the reaction was stirred for 1 h. To the reaction was added a solution of compound
(6-Ethoxyl-6-oxo-4E-hexenyl)triphenylphosphonium bromide (5)
Compound
Ethyl 2E,6Z,8E,10E-dodecatetraenoate (6)
To a solution of
2E,6Z,8E,10E-dodecatetraenoic acid (7)
Compound
Hydroxy-α-sanshool (8)
To a mixture of
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820974323 – Supplemental material for Synthesis of hydroxy-α-sanshool
Supplemental material, sj-pdf-1-chl-10.1177_1747519820974323 for Synthesis of hydroxy-α-sanshool by Jianjun Zhou, Yan Xiao, Taiping Chen, Jiyu Gao, Wencai Huang and Zicheng Li in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank State Key Laboratory of Biotherapy, West China Hospital, Sichuan University, for 1H NMR, 13C NMR, and high-resolution mass spectra (HRMS) determination.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Sichuan Provincial Administration of Traditional Chinese Medicine 2018HJZX04.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
