Abstract
In recent years, several deuterated drugs have entered clinical trials and have been approved for use. Deuterated methylamine and dimethylamine as important intermediates play significant roles in the preparation of deuterated drugs. In this study, we have developed a new method to prepare deuterated methylamine and dimethylamine. This method employs Boc-benzylamine as the starting material and TsOCD3 as the deuterated methylation reagent. Our method gives relatively high yields and involves simple purifications, which provide a favourable supplement for the development and synthesis of deuterated drugs in the future.
Introduction
Deuterated drugs are small molecule drugs in which the hydrogen atoms are replaced by the heavier stable isotope deuterium. In modern clinical medicine, deuterated drugs play an important role in the field of drug discovery and disease treatment. Compared with traditional drugs, deuterated drugs have better metabolic stability and improved efficacy. 1 Moreover, deuteration can slow down the system clearance rate and prolong the half-life of the drug in the body.2,3 Therefore, deuterated drugs can reduce the toxicity and side effects by reducing the dose of a single administration while not affecting the pharmacological activity of the drug.4,5 The first deuterated drug Austedo (deutetrabenazine), (Figure 1) was approved by the US Food and Drug Administration (FDA) in 2017 for the treatment of involuntary movements or chorea in patients with Huntington’s disease. 6 Many pharmaceutical companies and research institutions have committed to the development of deuterium-containing drugs. Several deuterium-containing drugs have entered clinical trials, such as BMS-986165, VX-984, and CTP-354.7–9 Recently, the clinical study of Donafinil showed significant overall survival in the treatment of advanced hepatocellular carcinoma, and this new drug application was formally accepted by the State Food and Drug Adminis-tration of China in May 2020.

The chemical structures of deuterium-containing drugs.
Among these deuterium drugs, the deuteration of methyl groups is a common method in the design of deuterated drugs. 10 Taking the JAK inhibitor BMS-986165 as an example, it exhibits a unique ability to selectively bind to the pseudokinase (JH2) domain of tyrosine kinase 2 (TYK2) and inhibits the TYK2 function through an allosteric mechanism. 11 The deuteration of the N-methyl group was shown to slow down the generation of a less selective primary amide metabolite in vivo by suppressing an N-demethylation metabolic pathway via a deuterium kinetic isotope effect (DKIE). 11 Similarly, Donafinil was designed from Sorafenib with a deuteriated methyl group. From the structures of BMS-986165 and Donafinil, we find that deuterated methylamine and dimethylamine, as the important intermediates, play significant roles in the preparation of deuterated drugs.
Generally, methylation of amino compounds can be achieved by nucleophilic substitution reactions with methyl iodide 12 or by reductive amination with aldehydes.13,14 Therefore, the deuterated methylation of amino compounds can be achieved with deuterated methylating agents such as deuteroiodomethane and TsOCD3. 15 However, the purity of the nucleophilic substitution reaction between the amine and deuteroiodomethane is relatively low due to the high activity of deuteroiodomethane. The amino compounds can go on to react with the deuterated methylation reagent to form a mixture of secondary, tertiary, and quaternary ammonium salts. Furthermore, the reductive amination of formaldehyde is another efficient and feasible method; however, this method did not lead to deuterated methylamine and dimethylamine because deuterated formaldehyde is difficult to obtain and very expensive. 16 Hence, there is a requirement to develop a new approach to the synthesis of deuterated methylamine and dimethylamine.
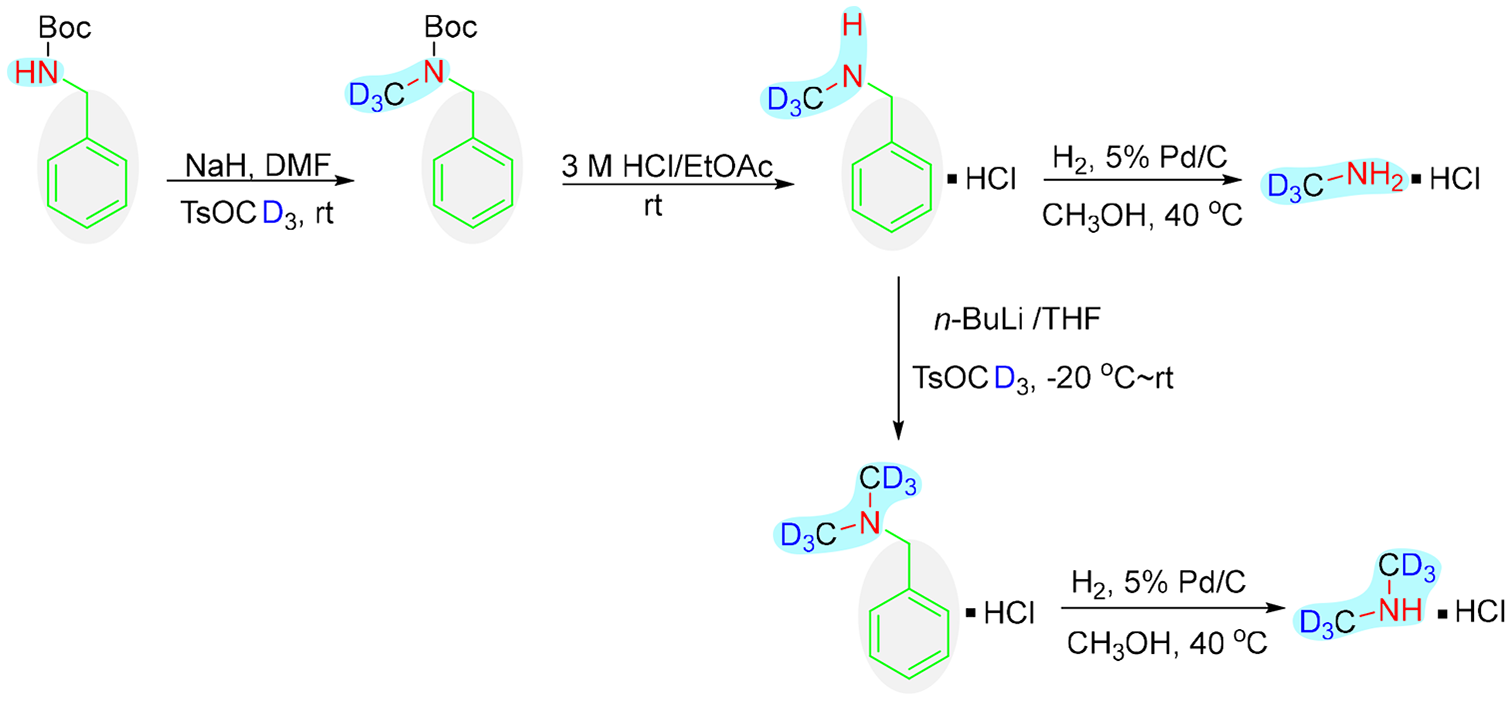
In this study, we report a novel synthetic method towards deuterated methylamine and dimethylamine. Boc-protected benzylamine as the starting material, reacted with the deuterated methylation reagent TsOCD3 at low temperature in the presence of NaH or n-butyllithium. Next, the Boc and benzyl protecting groups were removed to afford the targeted deuterated methylamine and dimethylamine. This novel method exhibits relatively high yields, a simple operation, and satisfactory purity.
Results and discussion
Initially, we attempted to synthesize deuterated methylamine by utilizing benzylamine as the starting material and n-butyllithium as a strong base. Although there were no tertiary amine or quaternary ammonium salt by-products, the yield of deuterated methylamine was relatively low. Next, we selected Boc-benzylamine as the starting material for the reaction. As shown in Scheme 1, Boc-benzylamine reacted with TsOCD3 to give intermediate

The synthetic routes to deuterated methylamine hydrochloride 4 and deuterated dimethylamine hydrochloride 6.
Similar to the deuterated methylamine hydrochloride
Conclusion
Deuterated methylamine and deuterated dimethylamine are important deuterated intermediates that are employed in the synthesis of various deuterated drug molecules. Convenient and controllable methods for synthesizing deuterated methylamine and dimethylamine are of great significance. In this study, an efficient and practical route conducive to the reduction of production costs of deuterium methylamine hydrochloride and deuterium dimethylamine hydrochloride is described. This novel method exhibits relatively high yields, a simple operation, and satisfactory purity.
Experimental
Materials and instruments
All chemicals and reagents used in this work were of analytical grade and obtained from commercial suppliers without further purification. 1H nuclear magnetic resonance (NMR) and 13C NMR spectra were recorded on a Bruker AM-400 NMR spectrometer in CDCl3 or dimethyl sulfoxide (DMSO)-d6. The chemical shifts are reported in δ (ppm) relative to tetramethylsilane (TMS) as the internal standard. Electrospray ionization mass spectrometry (ESI-MS) spectra were recorded on a Thermo MSQ PLUS mass spectrometer. High-resolution mass spectroscopy (HRMS) spectra were obtained on a Bruker Solarix 94 FT mass spectrometer.
tert-Butyl benzyl(methyl-d3)carbamate (2 )
At 0 ℃, compound
N-Benzylmethan-d3-amine hydrochloride (3 )
Compound
Methan-d3-amine hydrochloride (4 )
Compound
N-benzyl-N-(methyl-d3)methanamine-d3 hydrochloride (5 )
At -20℃, N-benzylmethan-d3-amine hydrochloride
Bis(methyl-d3)amine hydrochloride (6 )
Compound
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820969636 – Supplemental material for A practical synthesis of deuterated methylamine and dimethylamine
Supplemental material, sj-pdf-1-chl-10.1177_1747519820969636 for A practical synthesis of deuterated methylamine and dimethylamine by Zhaogang Liu, Xiangyu Ren and Peng Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
The Supporting Information is available free of charge at HRMS spectrum and 1H and 13C (PDF).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
