Abstract
Fifteen novel dithiocarbamate-derived naphthalimides as a type of potential anticancer and antimicrobial agents were synthesized and characterized by spectral and analytical techniques. The structures of

Introduction
The design and synthesis of highly efficient antitumor agents had attracted a lot of attention over the years. The development of the naphthalimide class of compounds as anticancer agents started in 1973. Two leading members of this family, amonafide and mitonafide, have been studied in phase I and phase II clinical trials. Unfortunately, the use and development of this kind of drug have been limited due to their unexpected toxicity. 1
In the past few decades, optimization of anticancer drugs has been an ongoing process. One promising strategy is the combination of an active moiety with known anticancer groups aimed at the development of potent cytotoxic substances. Recently, the Liu group reported the biological activities of dithiocarbamate (DTC) derivatives achieved by the introduction of the 1,2,3-trizaole ring leading to several compounds that showed an excellent, broad spectrum of anticancer activities and good anti-LSD1 activities.2–5 The Qian group investigated the anticancer activity of triazole-naphthalimides by introducing a 1,2,3-triazole ring to the 3 or 4 position of the naphthalimide and improved the anticancer cytotoxicity of the compounds over Amonafide.6,7 Our group recently reported tethering the biologically active group DTC to ruthenium(II) and dppfM(II) (M = Pt and Pd) units.8,9 These novel organometallic complexes-containing DTC exhibited good anticancer activity.
Inspired by the biological importance of naphthalimides as anticancer agents, it was necessary for us to attempt alternative structural modification of naphthalimides to find favorable or different biological activities. In this paper, the biologically active groups 1,2,3-triazole and DTC are tethered to a naphthalimide unit. The in vitro anticancer and antimicrobial activities of several novel 1,2,3-triazole-DTC-naphthalimides are described.
Results and discussion
Chemistry
The synthesis of the 1,2,3-triazole-dithiocarbamate-naphthalimides is shown in Scheme 1. Compound

Synthesis of 1,2,3-triazole-dithiocarbamate-naphthalimides. Reagents and conditions: (i) corresponding amine, CS2, anhydrous K3PO4, dimethylformamide (DMF), rt; (ii) NaN3, DMF, 60 °C; (iii) sodium ascorbate, CuSO4·5H2O, THF/H2O (1:1, V/V), rt.
X-ray diffraction studies
The single-crystal X-ray structures of

Molecular structure of

Molecular structure of

Molecular structure of
Biological evaluation
The in vitro antiproliferative activities of the synthetic compounds
Antiproliferative activity of the 1,8-naphthalimide-dithiocarbamate derivatives.
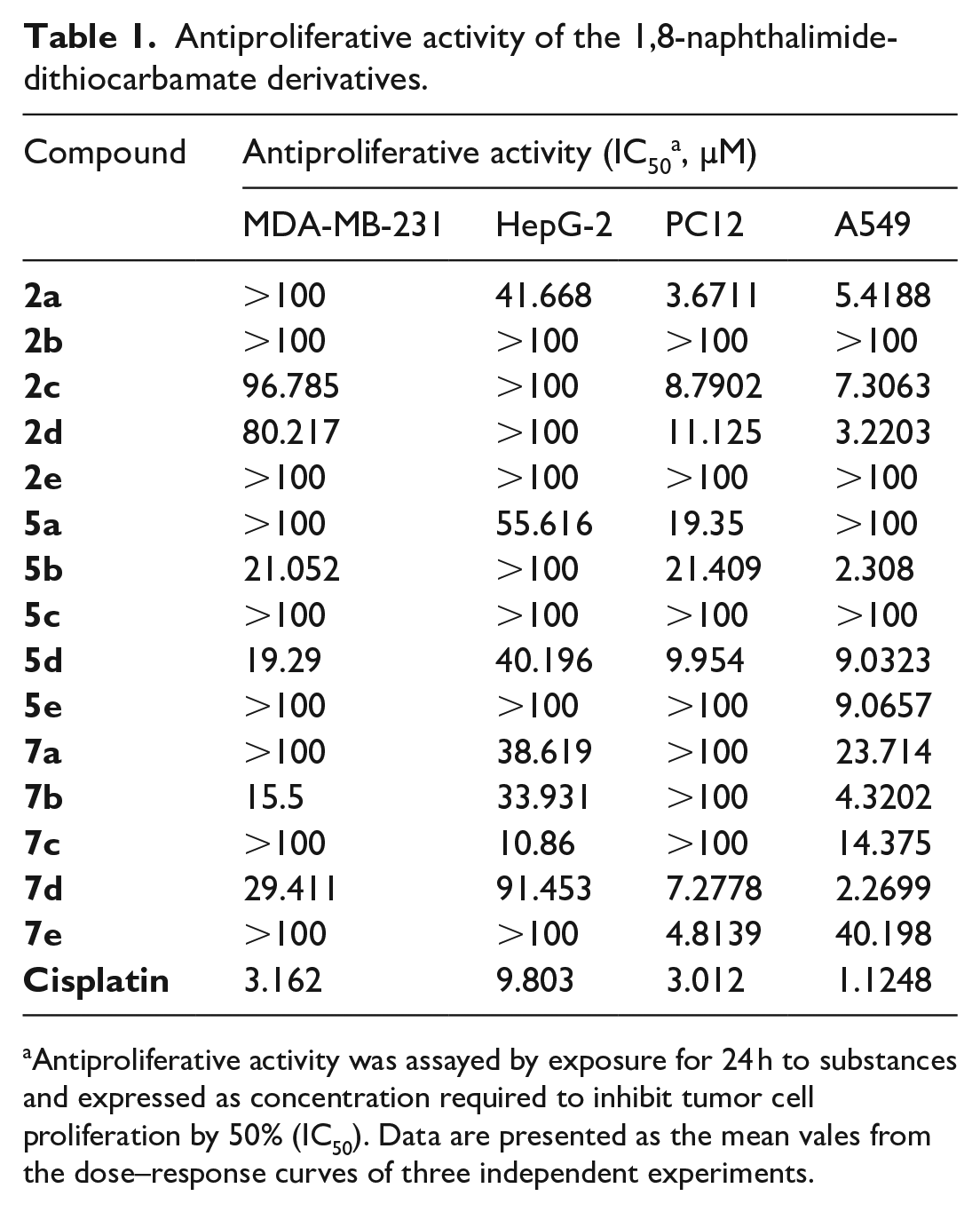
Antiproliferative activity was assayed by exposure for 24 h to substances and expressed as concentration required to inhibit tumor cell proliferation by 50% (IC50). Data are presented as the mean vales from the dose–response curves of three independent experiments.
As shown in Table 1, the DTCs
Recently, the Zhou group found that triazole-naphthalimides displayed good antimicrobial activities,12,13 and so, the antimicrobial activities of the 1,2,3-triazole-DTC-naphthalimides were also investigated. All the target compounds were evaluated for their antimicrobial activity in vitro against three bacteria (Escherichia coli, Bacillus subtilis, and Staphylococcus aureus) and one fungus (Candida albicans) (Table 2).
In vitro antibacterial and antifungal activities as MIC a (µM) of 1,8-naphthalimide-dithiocarbamate derivatives.

C. albicans: Candida albicans; E. coli: Escherichia coli; B. subtilis: Bacillus subtilis; S. aureus: Staphylococcus aureus.
Minimum inhibitory concentration against C. albicans, B. subtilis, S. aureus, and Staphylococcus aureus.
As shown in Table 2, most of compounds
Conclusion
In summary, a series of novel naphthalimide derivatives with DTC or 1,2,3-triazole-DTC moieties were designed and synthesized, and their antitumor and antibacterial activities were evaluated in vitro. It was found that the introduction of a triazole ring helps to improve the activities of the DTC-naphthalimides and that some naphthalimides with substituents at the 4-position have the higher activities. This work demonstrates that incorporating the biological active naphthalimide and DTC units, or a 1,2,3-triazole-DTC moiety, might result in novel classes of lead compounds with potential antitumor and antimicrobial activities.
Experimental section
General procedures
All manipulations were carried out at room temperature under a nitrogen atmosphere using standard Schlenk techniques, unless otherwise stated. All reagents were purchased from commercial sources and were used without further purification. 1H and 13C NMR spectra were obtained using a Bruker 500 MHz magnetic resonance spectrometer. 1H and 13C NMR chemical shifts are relative to tetramethylsilane (TMS). Mass spectrometry was recorded with a Bruker amaZon SL spectrometer. The starting materials 2-(3-bromopropyl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (
General procedure for synthesis of DTC-naphthalimides 2a–e
To a solution of amine (1.1 mmol) in dimethylformamide (DMF) (5 mL) was added dropwise carbon disulfide (0.17 g, 2.2 mmol) and anhydrous potassium phosphate (0.23 g, 1.1 mmol). The resulting mixture was stirred at room temperature for 30 min. Then, the naphthalimide compound
Synthesis of the azide naphthalimide compound 3
The naphthalimide
General procedure for synthesis of 1,2,3-triazole-DTC naphthalimides 5a–e and 7a–e
The azide naphthalimide derivatives
Crystal structures determination for 2b , 5a , and 7b
Single crystals of compounds
Cytotoxicity assay in vitro
Cytotoxic activities were evaluated by the MTT [3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide] method in the MDA-MB-231, HepG-2, PC12, and A549 cell lines. Briefly, the cell suspensions (198 μL) were plated in 96-well microtiter plates at a density of 5 × 104 cells mL−1 and incubated for 12 h at 37 °C in a humidified incubator with 5% CO2. The test compounds with different concentrations were added to each well and further incubated for 24 h under the same conditions. Then, 20 μL of the MTT solution was added to each well and incubated for 4 h. The old medium (200 μL) containing MTT was then gently replaced by dimethyl sulfoxide (DMSO) and pipetted to dissolve any formazan crystals formed. Absorbance was then determined on a Spectra Max Plus plate reader at 570 nm. Dose–response curves were generated, and the IC50 values were determined. CDDP, a commonly approved agent for the treatment of many tumors, was used as the positive control.
Antibacterial assays
A total of four microbial strains were used in the study. Staphylococcus aureus (ATCC 27217), Escherichia coli (ATCC 15597), Bacillus subtilis (ATCC 63501), and Monilia albican (ATCC 10231), which were taken from the microbiology laboratory of Yunnan Minzu University, were used in the study. For experiments, bacteria were inoculated onto Colombia agar (Merck, Germany) plates supplemented with hemine (5 µg mL−1), menadione (1 µg mL−1), and 5% horse blood. Incubation was performed at 37 °C under anaerobic conditions for 5–7 d in an automated anaerobic chamber (Electrotek, UK) with 5% CO2. After harvesting the bacteria, bacterial suspensions prepared in the sterilized test tubes containing Colombia broth supplemented with hemine (5 µg mL−1), menadione (1 µg mL−1), and the inoculum were adjusted according to the turbidity of the 0.5 McFarland standard. For the determination of the MIC, 100 µL of chemicals was added into the first wells of the U-bottom-polistiren microplates and serially diluted in the range of 0.125–256 µg mL−1. Then, 100 µL of bacterial suspensions was added onto them, except the negative control well. Polistiren microplates were incubated for 48 h in an anaerobic cabin, and after the incubation time, wells which contained minimum concentration of the extracts where no visible growth can be seen were expressed as MIC, the minimum concentration which completely inhibited bacterial growth. All experiments were performed in duplicates.
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820966971 – Supplemental material for 1,2,3-triazole-dithiocarbamate-naphthalimides: Synthesis, characterization, and biological evaluation
Supplemental material, sj-pdf-1-chl-10.1177_1747519820966971 for 1,2,3-triazole-dithiocarbamate-naphthalimides: Synthesis, characterization, and biological evaluation by Qiu Mei Chen, Zhen Li, Guang Xun Tian, Yi Chen and Xiang Hua Wu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support from National Natural Science Foundation of China (No. 21002086) and the Natural Science Foundation of Yunnan Province (No. 2010ZC071).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
