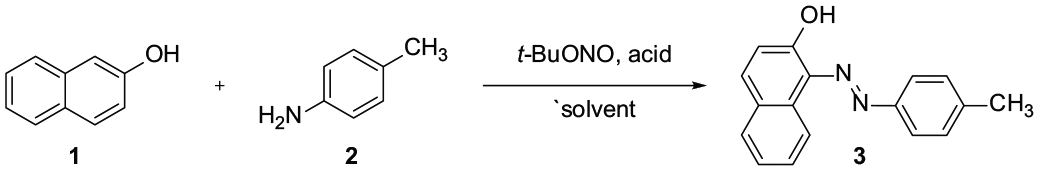
Abstract
A one-pot method for the synthesis of azo compounds by the reaction of β-naphthol with aryl amines using t-BuONO as the nitrosonium source in DCM at room temperature was developed. This method features mild reaction conditions, a simple experimental procedure, and is free of acidic or alkaline additives.

Introduction
Azo compounds, some of the most important synthetic highly colored dyes, are widely distributed.1–3 At present, about 3000 azo dyes are known to be in use in various fields around the world. 4 For instance, azo dyes are used in textile dyeing, histology dyeing, drug colorimetric analysis, cosmetics, food, digital printing, photographic colorants, and so on.5–12 However, azo compounds also have excellent optical and photoelectric properties13,14 and can be used to diagnose Alzheimer’s disease. 15 In addition, some azo compounds have been reported to possess antibacterial and antifungal activities.16,17 Due to their broad applications, many reports have focused on the preparation of synthetic azo compounds.18–20
The traditional approach for the preparation of azo compounds is usually divided into two steps: first, aromatic primary amines are diazotized to form diazonium salts using mineral acids, and then the so formed diazonium salts are coupled with active aromatic compounds under alkaline conditions. 21 This classical synthetic method is limited by the need for low reaction temperatures and, especially, the usage of acidic or alkaline reagents22,23 which violate the principles of green chemistry and cause permanent pollution to the environment. 24 Therefore, in recent years, many researchers have devoted themselves to exploring mild and green synthetic strategies, especially on developing novel catalysts for diazotization–diazo coupling. For example, a nanomagnetic-supported sulfonic acid was developed by Kolvari to catalyze diazotization–diazo coupling reactions in the absence of solvents. 25 Chermahini reported the synthesis of azo compounds using modified montmorillonite K-10. 26 According to Bagherzade’s research achievement, diethylamine functionalized polyethylene glycol could be used as a catalyst in the process of preparing azo dyes. 27 In addition, many investigations demonstrated that Fe(HSO4)3, 28 magnetic Fe3O4 nanoparticles, 29 granular PTFE (polytetrafluoroethylene), 30 and graphene quantum dots 4 were also effective catalysts for the synthesis of azo compounds. However, the above-mentioned methodologies still suffer from some disadvantages, such as tedious processes, high manufacturing costs, and the use of metals or mineral acids. In this study, we report a one-pot method for the synthesis of azo compounds at room temperature in the absence of any acidic or alkaline additive using tert-butyl nitrite as the nitrosonium source (Scheme 1).
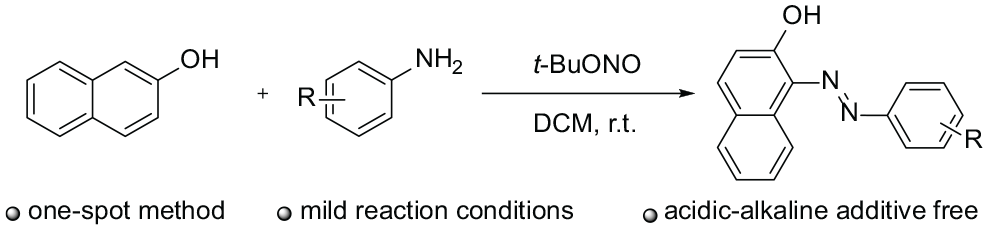
One-pot synthesis of azo compounds.
Results and discussion
Initially, we carried out the reaction using β-naphthol (
Optimization of the reaction conditions. a

Reaction conditions:
Isolated yield.
Without t-BuONO.
With the optimized conditions in hand, the scope of amines as the substrates was examined (Scheme 2). Initially, various substituted amines were reacted with β-naphthol in the presence of t-BuONO in DCM at room temperature. Delightfully, both electron-donating and electron-withdrawing functional groups such as alkyl (

The scope of amines as substrates. Reagents and conditions:
Conclusion
In summary, we have developed a one-pot approach for the synthesis of azo compounds by the reaction of naphthol with aromatic amines at room temperature. This protocol was characterized by easily accessible raw materials, mild reaction conditions, and a simple operational procedure. Most importantly, the azo compounds could be synthesized in the absence of any acidic or alkaline additive, which is in line with the theme of green chemistry. We expect this method to find considerable potential for application in industry.
Experimental procedure
Commercially available chemicals were obtained from commercial suppliers and used without further purification unless otherwise stated. Proton NMR (1H) and carbon (13C) NMR spectra were recorded on an Ascend TM 500 MHz NMR spectrometer. The following abbreviations are used for the multiplicities: s: singlet, d: doublet, t: triplet, q: quartet, m: multiplet, and br s: broad singlet for proton spectra. Coupling constants (J) are reported in Hertz (Hz). Melting points (uncorrected) were determined on an automatic melting point apparatus (ZRD-1) from Tianjin optical instrument factory. Analytical thin-layer chromatography was performed on Polygram SIL G/UV254 plates. Visualization was accomplished with shortwave UV light, or by KMnO4 staining followed by heating. Flash column chromatography was performed using silica gel (200300 mesh) from Qingdao Haiyang Chemical Co., Ltd with solvents distilled prior to use.
General procedure for the synthesis of azo compounds
To a solution of β-naphthol (1.0 mmol, 1.0 equiv.) in DCM (5.0 mL) was added the aryl amine (1.5 mmol, 1.5 equiv.) and t-BuONO (1.5 mmol, 1.5 equiv.). The reaction mixture was stirred in air at room temperature for 24 h and then concentrated in vacuum. The crude mixture was purified by column chromatography on silica gel with a mixture of petroleum ether and ethyl acetate as eluent to afford the target compounds.
1-(phenyldiazenyl)naphthalen-2-ol (3a ):
26
Red powder; yield 87%; m.p. 130–132 °C (lit. 26 128–130 °C); 1H NMR (500 MHz, Chloroform-d): δ 8.56 (d, J = 8.2 Hz, 1H), 7.76-7.70 (m, 3H), 7.60 (d, J = 7.8 Hz, 1H), 7.55 (t, J = 7.7 Hz, 1H), 7.48 (t, J = 7.9 Hz, 2H), 7.39 (t, J = 7.5 Hz, 1H), 7.30 (t, J = 7.4 Hz, 1H), 6.87 (d, J = 9.4 Hz, 1H).
1-(p-tolyldiazenyl)naphthalen-2-ol (3b ):
29
Red powder; yield 88%; m.p. 132–133 °C (lit. 29 130–132 °C); 1H NMR (500 MHz, Chloroform-d): δ 8.62 (d, J = 8.3 Hz, 1H), 7.73 (d, J = 9.3 Hz, 1H), 7.68 (d, J = 8.3 Hz, 2H), 7.63 (d, J = 7.9 Hz, 1H), 7.56 (t, J = 7.6 Hz, 1H), 7.40 (t, J = 7.5 Hz, 1H), 7.29 (d, J = 8.1 Hz, 2H), 6.93 (d, J = 9.3 Hz, 1H), 2.42 (s, 3H); 13C NMR (126 MHz, Chloroform-d): δ 168.6, 143.7, 139.0, 138.5, 133.7, 130.3, 129.9, 128.7, 128.6, 128.2, 125.5, 124.1, 121.8, 119.3, 21.4.
1-({4-[tert-butyl]phenyl}diazenyl)naphthalen-2-ol (3c ):
Red solid; yield 92%; m.p. 87–89 °C (lit. 31 86–88 °C); 1H NMR (500 MHz, Chloroform-d): δ 8.60 (d, J = 8.3 Hz, 1H), 7.73-7.70 (m, 3H), 7.62 (d, J = 7.8 Hz, 1H), 7.56 (t, J = 7.7 Hz, 1H), 7.51 (d, J = 8.7 Hz, 2H), 7.39 (t, J = 6.9 Hz, 1H), 6.92 (d, J = 9.4 Hz, 1H), 1.37 (s, 9H); 13C NMR (126 MHz, Chloroform-d): δ 169.4, 151.6, 143.3, 139.2, 133.7, 130.0,128.8, 128.7, 128.2, 126.7, 125.5, 124.4, 121.8, 119.0, 34.9, 31.5.
1-({4-fluorophenyl}diazenyl)naphthalen-2-ol (3d ):
Red floccule; yield 90%; m.p. 144–146 °C (lit. 28 143–145 °C); 1H NMR (500 MHz, Chloroform-d): δ 15.80 (s, 1H), 8.57 (d, J = 8.2 Hz, 1H), 7.77-7.70 (m, 3H), 7.62 (d, J = 7.9 Hz, 1H), 7.54 (t, J = 7.7 Hz, 1H), 7.38 (t, J = 6.9 Hz, 1H), 7.16 (t, J = 8.6 Hz, 2H), 6.93 (d, J = 9.3 Hz, 1H); 13C NMR (126 MHz, Chloroform-d): δ 166.5, 162.5 (d, J = 249.3 Hz), 143.0 (d, J = 2.9 Hz), 138.9, 133.5, 129.9, 128.7, 128.6, 128.3, 125.5, 123.4, 121.7, 121.3 (d, J = 8.5 Hz), 116.6 (d, J = 23.2 Hz).
1-({4-chlorophenyl}diazenyl)naphthalen-2-ol (3e ):
26
Red floccule; yield 64%; m.p. 153–155 °C (lit. 26 158–160 °C); 1H NMR (500 MHz, Chloroform-d): δ 16.03 (s, 1H), 8.53 (d, J = 8.2 Hz, 1H), 7.72 (d, J = 9.4 Hz, 1H), 7.67 (d, J = 8.9 Hz, 2H), 7.60 (d, J = 7.8 Hz, 1H), 7.55 (t, J = 7.7 Hz, 1H), 7.45-7.38 (m, 3H), 6.87 (d, J = 9.4 Hz, 1H).
1-([4-bromophenyl]diazenyl)naphthalen-2-ol (3f ):
29
Red powder; yield 74%; m.p. 168–170 °C (lit. 32 169–170 °C); 1H NMR (500 MHz, Chloroform-d): δ 16.06 (s, 1H), 8.53 (d, J = 8.2 Hz, 1H), 7.73 (d, J = 9.4 Hz, 1H), 7.62-7.54 (m, 6H), 7.41 (t, J = 7.0 Hz, 1H), 6.86 (d, J = 9.4 Hz, 1H).
1-({4-iodophenyl}diazenyl)naphthalen-2-ol (3g ):
33
Dark red powder; yield 69%; m.p. 164–166 °C; 1H NMR (500 MHz, Chloroform-d): δ 16.05 (s, 1H), 8.46 (d, J = 8.1 Hz, 1H), 7.74 (d, J = 8.7 Hz, 2H), 7.68 (d, J = 9.4 Hz, 1H), 7.56 (d, J = 7.8 Hz, 1H), 7.54-7.50 (m, 1H), 7.41 (d, J = 8.7 Hz, 2H), 7.38 (t, J = 7.5 Hz, 1H), 6.81 (d, J = 9.4 Hz, 1H); 13C NMR (126 MHz, Chloroform-d): δ 172.6, 144.5, 140.7, 138.7, 133.5, 130.4, 129.1, 128.8, 128.3, 126.1, 124.9, 121.9, 120.2, 91.8.
1-({4-methoxyphenyl}diazenyl)naphthalen-2-ol (3h ):
29
Red powder; yield 80%; m.p. 136–138 °C (lit. 29 137–138 °C); 1H NMR (500 MHz, Chloroform-d): δ 15.72 (s, 1H), 8.70 (d, J = 8.3 Hz, 1H), 7.82 (d, J = 9.0 Hz, 2H), 7.76 (d, J = 9.2 Hz, 1H), 7.69 (d, J = 7.9 Hz, 1H), 7.57 (t, J = 7.7 Hz, 1H), 7.40 (t, J = 7.5 Hz, 1H), 7.03 (dd, J = 10.4, 9.1 Hz, 3H), 3.88 (s, 3H).
Ethyl-4-({2-hydroxynaphthalen-1-yl}diazenyl)benzoate (3i ):
Red powder; yield 66%; m.p. 139–141 °C (lit. 34 149 °C); 1H NMR (500 MHz, Chloroform-d): δ 8.43 (d, J = 8.1 Hz, 1H), 8.12 (d, J = 8.6 Hz, 2H), 7.67-7.64 (m, 3H), 7.58-7.51 (m, 2H), 7.40 (t, J = 7.4 Hz, 1H), 6.73 (d, J = 9.6 Hz, 1H), 4.41 (q, J = 7.2 Hz, 2H), 1.44 (t, J = 7.1 Hz, 3H); 13C NMR (126 MHz, Chloroform-d): δ 178.0, 166.1, 146.8, 142.3, 133.5, 131.4, 131.1, 129.5, 129.0, 128.4, 127.8, 126.8, 126.2, 122.2, 116.9, 61.2, 14.5.
1-({4-nitrophenyl}diazenyl)naphthalen-2-ol (3j ):
4
Red powder; yield 86%; m.p. 250–252 °C (lit. 4 249–250 °C); 1H NMR (500 MHz, Chloroform-d): δ 16.13 (s, 1H), 8.42 (d, J = 8.0 Hz, 1H), 8.32 (d, J = 9.1 Hz, 2H), 7.71-7.68 (m, 3H), 7.60-7.51 (m, 2H), 7.44 (t, J = 7.4 Hz, 1H), 6.70 (d, J = 9.6 Hz, 1H).
1-({3-(trifluoromethyl)phenyl}diazenyl)naphthalen-2-ol (3k ):
Bright red powder; yield 88%; m.p. 159–160 °C (lit. 35 193–195 °C); 1H NMR (500 MHz, Chloroform-d): δ 16.06 (s, 1H), 8.47 (d, J = 8.5 Hz, 1H), 7.93 (s, 1H), 7.79 (d, J = 8.0 Hz, 1H), 7.69 (d, J = 9.5 Hz, 1H), 7.61-7.49 (m, 4H), 7.39 (t, J = 7.4 Hz, 1H), 6.80 (d, J = 9.5 Hz, 1H); 13C NMR (126 MHz, Chloroform-d): δ 173.9, 145.1, 141.4, 133.4, 132.3 (q, J = 32.9 Hz), 130.7, 130.3, 129.4, 128.9, 128.4, 126.5, 125.1, 123.9 (q, J = 272.6 Hz), 123.3 (q, J = 3.6 Hz), 122.1, 121.4, 114.9 (q, J = 4.0 Hz).
1-({3-nitrophenyl}diazenyl)naphthalen-2-ol (3l ):
32
Red powder; yield 90%; m.p. 194–196 °C (lit. 32 194–196 °C); 1H NMR (500 MHz, Chloroform-d): δ 16.03 (s, 1H), 8.54 (s, 1H), 8.50 (d, J = 8.2 Hz, 1H), 8.08 (d, J = 8.1 Hz, 1H), 7.91 (d, J = 8.0 Hz, 1H), 7.73 (d, J = 9.5 Hz, 1H), 7.65-7.53 (m, 3H), 7.43 (t, J = 7.4 Hz, 1H), 6.80 (d, J = 9.5 Hz, 1H).
1-({2,4-dimethylphenyl}diazenyl)naphthalen-2-ol (3m ):
Dark red solid; yield 93%; m.p. 149–150 °C (lit. 36 159–160 °C); 1H NMR (500 MHz, Chloroform-d): δ 8.59 (d, J = 8.2 Hz, 1H), 7.94 (d, J = 8.3 Hz, 1H), 7.70 (d, J = 9.4 Hz, 1H), 7.59 (d, J = 7.8 Hz, 1H), 7.54 (t, J = 8.2 Hz, 1H), 7.37 (t, J = 7.4 Hz, 1H), 7.14 (d, J = 8.3 Hz, 1H), 7.07 (s, 1H), 6.90 (d, J = 9.4 Hz, 1H), 2.50 (s, 3H), 2.36 (s, 3H).
1-({3-chloro-4-methylphenyl}diazenyl)naphthalen-2-ol (3n ):
37
Red powder; yield 78%; m.p. 133–135 °C; 1H NMR (500 MHz, Chloroform-d): δ 15.92 (s, 1H), 8.52 (d, J = 8.2 Hz, 1H), 7.75 (d, J = 2.0 Hz, 1H), 7.69 (d, J = 9.4 Hz, 1H), 7.58 (d, J = 7.8 Hz, 1H), 7.54 (t, J = 8.2 Hz, 1H), 7.45 (dd, J = 8.2, 2.0 Hz, 1H), 7.38 (t, J = 7.4 Hz, 1H), 7.28 (d, J = 8.2 Hz, 1H), 6.86 (d, J = 9.4 Hz, 1H), 2.39 (s, 3H); 13C NMR (126 MHz, Chloroform-d): δ 169.8, 144.6, 139.9, 135.7, 133.5, 131.7, 130.2, 129.0, 128.7, 128.2, 125.8, 124.2, 121.9, 118.9, 117.8, 20.0.
1-({3-bromo-4-methylphenyl}diazenyl)naphthalen-2-ol (3o ):
38
Dark red powder; yield 74%; m.p. 217–218 °C (lit. 38 217–218 °C); 1H NMR (500 MHz, Chloroform-d): δ 15.91 (s, 1H), 8.53 (d, J = 8.2 Hz, 1H), 7.94 (d, J = 2.1 Hz, 1H), 7.70 (d, J = 9.4 Hz, 1H), 7.59 (d, J = 7.8 Hz, 1H), 7.55 (t, J = 7.7 Hz, 1H), 7.51 (dd, J = 8.1, 1.9 Hz, 1H), 7.38 (t, J = 7.4 Hz, 1H), 7.29 (d, J = 8.2 Hz, 1H), 6.87 (d, J = 9.4 Hz, 1H), 2.42 (s, 3H); 13C NMR (126 MHz, Chloroform-d): δ 169.7, 144.7, 139.9, 137.5, 133.5, 131.5, 130.2, 129.0, 128.7, 128.2, 125.9, 125.9, 124.2, 122.2, 121.9, 118.4, 22.9.
Supplemental Material
Supplemental material for One-pot synthesis of azo compounds in the absence of acidic or alkaline additives
Supplemental material, Supplemental_Material for One-pot synthesis of azo compounds in the absence of acidic or alkaline additives by Ting-Ting Liu, Jiao-Zhao Yan, Xin-Wang Cheng, Pan Duan and Yao-Fu Zeng in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Foundation of Hu’nan Educational Committee (18C0476) and the Natural Science Foundation of Hunan Province (2020JJ5475).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.