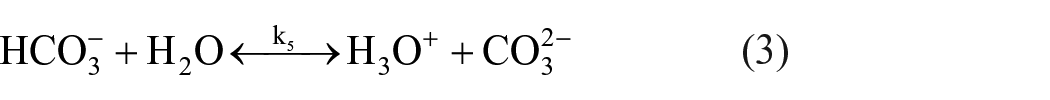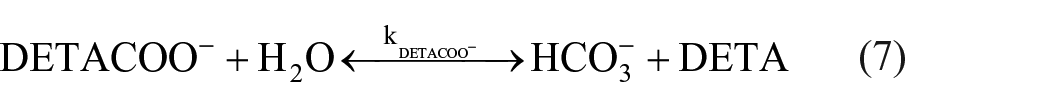Abstract
The absorption and desorption data of CO2 in aqueous solutions with a mass fraction of 10% and 20% of diethylenetriamine are measured at 313.15, 343.15, 373.15, and 393.15 K. The electrolyte non-random two-liquid theory is developed using Aspen V9.0 to correlate and predict the vapor–liquid equilibrium of CO2 in aqueous diethylenetriamine solutions. The model predicted the heat capacity and saturated vapor pressure data of diethylenetriamine, the mixed heat of a diethylenetriamine–H2O binary system, and the vapor–liquid equilibrium data of a diethylenetriamine–H2O–CO2 ternary system. The physical parameters and the interaction parameters of the model system are calculated. The model predicted CO2 solubility showing a 10% average absolute deviation from experimental data. The calculated values of the model are basically consistent with the experimental values.

Introduction
With the development of economies and society, the demand for fossil fuels is increasing rapidly, which makes the emission of CO2 and global warming increasingly serious. A recent study by Rosenfeld 1 shows that global warming problem is more serious than previously estimated. As a greenhouse gas, carbon dioxide is a major factor contributing to the growth of global warming.2,3 China’s coal-based energy structure demands that any potential approach toward large-scale and effective CO2 emission reduction is CO2 capture and storage. 4 As for CO2 capture, the solvent absorption method is mainly adopted in industry to realize large-scale CO2 treatment, which has advantages of fast absorption, a simple process, and large processing capacity, but also has disadvantages of high consumption and significant corrosion of equipment. 5 In order to develop CO2 capture and storage technology with low energy consumption and low cost, it is necessary to model, expand simulations, and utilize thermodynamic properties such as phase equilibrium and chemical equilibrium, and optimize CO2 capture processes. There are three kinds of commonly used thermodynamic models for evaluating the thermodynamic properties of acidic gas absorption processes: the first is the KE model with simple and non-strict form. 6 This model has poor universality, but the fitting effect is a good in the specific load range. It cannot be extended to the absorption process of a mixed gas, and it cannot be used to predict the reaction heat of each component. The second category is the more rigorous activity coefficient model and equation of state. For the activity coefficient model, more variables are required and the model is more complex. As the most widely used model in this class, Deshmukh and Mather 7 model has been successfully used to calculate the phase equilibrium of the monoethanolamine–H2O–CO2 (MEA–H2O–CO2) system. The third category encompasses more complex and more rigorous models, including non-random dual fluid electrolyte (e-NRTL) and extended UNIQUAC models, which have been widely used in aqueous amine systems, 8 while extended UNIQUAC models are rarely used. In the CO2 capture based on amines, the reaction between CO2 and the aqueous solution of amine will form numerous ions. Due to interactions between ions, the non-ideal nature of the electrolyte system is relatively strong. How to accurately describe the non-ideal nature of the solution is the key to simulate the electrolyte solution system. The e-NRTL model proposed by Mock et al. 9 can correct the non-ideal nature of the multi-component electrolyte system and can be used to simulate the capture process of CO2. Therefore, this paper selects the e-NRTL model for the relevant thermodynamic simulation.
Diethylenetriamine (DETA) is a polyamine containing three amine functionalities (two primary and one secondary amine), which is expected to have high CO2 loading capacity.10,11 Compared to typical alkanolamines, DETA has a high capacity for binding CO2, a high absorption rate and a high boiling point.12,13 Therefore, aqueous solutions of DETA can be potential solvents for CO2 capture. Due to the advantages mentioned above, a series of studies have been carried out on DETA recently, including mixing of DETA with PZ, 14 impregnating of plaza-activated carbon, alumina15,16 and polymethyl methacrylate polymeric materials, 17 anhydrous solvent ionic liquid based on DETA.18,19
In this work, new experimental CO2 solubility data in aqueous DETA solutions (mass fraction of 10% and 20% of DETA) are generated over a wide pressure range (up to 480 kPa) and temperature (313.15–393.15 K) to extend the limited experimental data found in the literature. The detailed process is described in the literature.20,21
Results and discussion
Chemical equilibrium
The chemical reaction of DETA is complex because DETA is a tertiary amine consisting of primary and secondary amino groups in its structure. So a wide range of chemical reactions is possible with a large number of species formed. The following possible parallel reversible chemical reactions in liquid phase for the (DETA–H2O–CO2) system are presented below. The constant-volume method, combined with gas chromatography analysis was used to measure the equilibrium solubility at different concentrations. The schematic diagram of the CO2 absorption measurement device is presented in Figure 1.
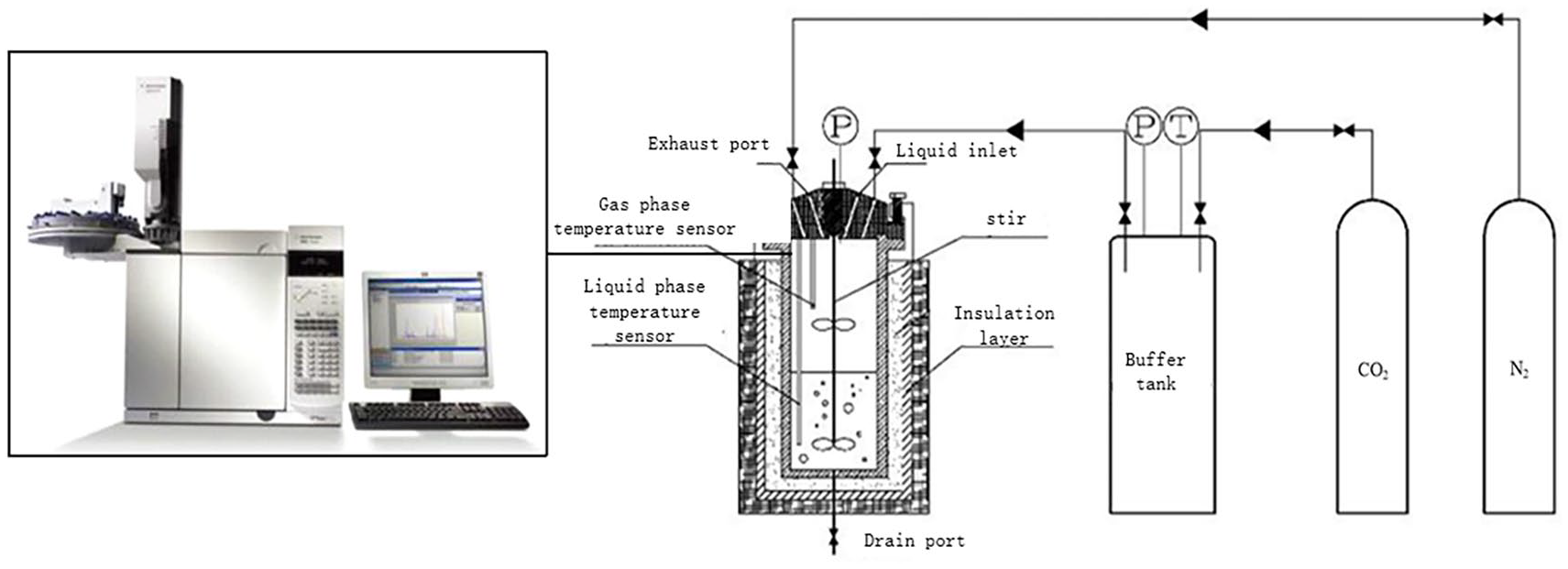
Schematic diagram of the constant temperature CO2 absorption solubility measurement device.
Dissociation of water
Formation of bicarbonate
Formation of carbonate
Amine deprotonation
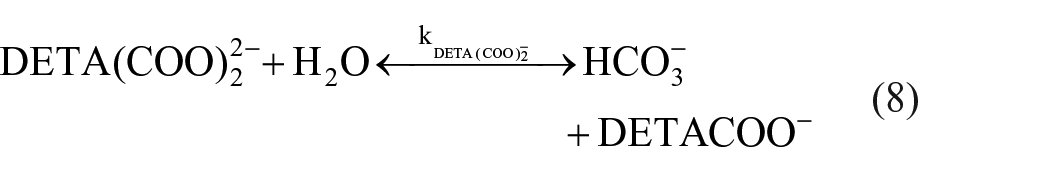
Dissociation of carbamate
Due to the complex species of the system, there are many isomers. The main reaction equations are outlined. The chemical equilibrium constants of equations (1)–(8) are a function of temperature, which can be expressed by the following formula
where K is the reaction equilibrium constant; T is the reaction temperature; A, B, C, and D are the parameters of each reaction equilibrium constant, the values of which are taken from the literature22,23 and are listed in Table 1.
The parameters of the equilibrium constants of the reactions in the DETA–H2O–CO2 system.

Phase equilibrium
The vapor–liquid equilibrium is defined by the equality of the fugacity at a given temperature and pressure. In the process of thermodynamic calculations, different reference states correspond to different phase equilibrium relations, so the appropriate reference state must be selected for our case study. In the model, CO2 was set as the Henry component, so the reference states of the ions and CO2 in the solution were in a state of infinite dilution, and the reference states of water and DETA were pure liquid. Therefore, DETA and H2O have the following phase equilibrium relationship

The phase equilibrium relationship between CO2 and ions is as follows

where


The model results
DETA systems
By fitting the data of the liquid heat capacity 26 and saturated vapor pressure for DETA, 27 the coefficients of the Antoine equation and specific heat capacity equations (14) and (15) were obtained. The results are shown in Tables 2 and 3. The experimental values and the calculated values from the model for the specific heat capacity are shown in Figure 2. The average relative deviation of DETA is 0.41%. Figure 3 compares the experimental values of the saturated vapor pressure with the model fitting values, and the average relative deviation of saturated vapor pressure is 1.3%. It can be seen from the figures that the experimental values are in good agreement with the model fitting values, and the parameters obtained can accurately describe the properties of the pure absorbent
Values of the specific heat capacity equation parameter (kJ/kmol/K).

DETA: diethylenetriamine.
Values of the Antoine equation parameter (atm).

DETA: diethylenetriamine.

Comparison of the e-NRTL model predictions with experimental data for the specific heat capacity of DETA from 300 to 400 K.

Comparison of the e-NRTL model predictions with experimental data for the saturated vapor pressure of DETA from 350 to 550 K.
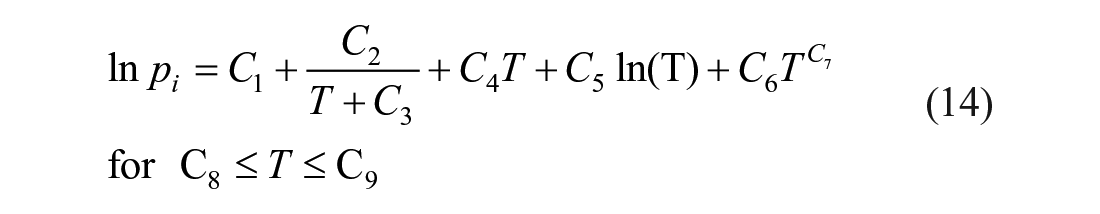

Binary systems: DETA–H2O
The binary interaction coefficients of the e-NRTL model were obtained by fitting the mixed heat data of the DETA–H2O system. 20 Figure 4 compares the experimental values and the model calculation values of the mixed heat data of the DETA–H2O system. The average relative deviation of the mixed heat data of {DETA–H2O} is about 5%. It can be seen from the figure that the thermodynamic model can accurately predict the vapor–liquid equilibrium and mixed heat data of the system.

Excess molar enthalpy for the binary mixtures {DETA (2) + H2O (1)}.
Ternary systems: DETA–H2O–CO2
As the system of polyamines is relatively complex compared with that of monoamines, the thermodynamic reactions between substances in the system should be clarified before setting up the thermodynamic model. Hartono
28
determined the species distribution of the DETA–H2O–CO2 system by nuclear magnetic resonance (NMR) spectrometry, and found that there were 24 species in the system. The main species are DETACOO−,
For the amine + H2O + CO2 system, there is not only the interaction between the amine and the CO2 and H2O molecules, but also the interaction between ion pairs and molecules. The values of the action parameters can be obtained by fitting the solubility data of CO2 in amine aqueous solutions of different concentrations at different temperatures by applying the short-range term of e-NRTL. DETA has two symmetric primary amines (p) and a tertiary amine (s), so the species in aqueous solution is complex and there are many isomers. For the DETA + H2O + CO2 system, regression parameters are required as follows: a binary interaction parameter of H2O–DETA, an interaction parameter between the molecule {H2O, DETA, CO2} and the ion pair {DETAH+(s), DETAH+(p),
Figures 5 and 6 describe the CO2 solubility in DETA solutions with mass fractions of 10% and 20%, respectively, and compare the calculated results with the experimental values. The measured temperatures of solubility were 313.15, 343.15, 373.15, and 393.15 K, respectively, and the CO2 loading ranged from 0.0 to 1.9. The deviation of experimental data and fitting data is shown in the Table 4. As DETA is a ternamine, the species of the system is relatively complex. In the model calculation, some isomers were approximated, and the calculation error at low temperature was slightly larger. Under the conditions of low pressure, the partial pressure of CO2 in the gas phase is relatively low, the amount of CO2 in the vapor phase is relatively small, and the average relative deviation will be large. At low temperature, the error in the fitting results is relatively large. The reason may be that at low temperature, the solution has a strong CO2 absorption capacity and a low gas pressure, resulting in a large measurement error, which will affect the final simulation results.

Solubility of CO2 in a DETA solution with a mass fraction of 10%.
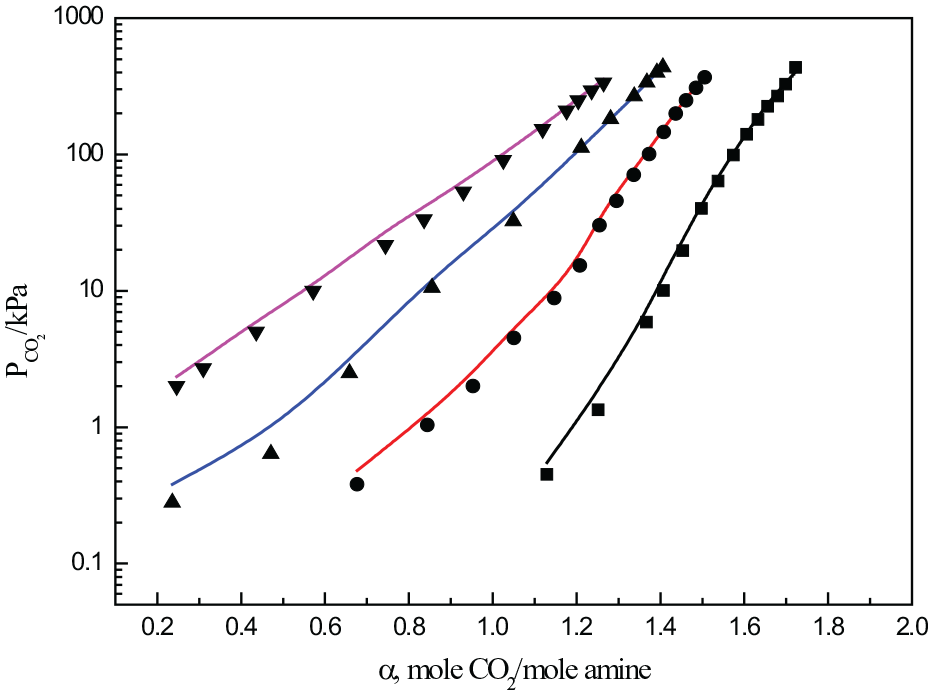
Solubility of CO2 in a DETA solution with a mass fraction of 20%.
The average relative deviations.

Conclusion
In this work, new experimental CO2 solubility in aqueous DETA solutions (mass fraction of 10% and 20% of DETA) have been measured over a wide range of pressure (up to 480 kPa) and temperature (313.15–393.15 K) to extend the limited experimental database. A thermodynamic model based on e-NRTL was established and used to correlate VLE data to predict CO2 partial pressure for the DETA–CO2–H2O system. The model predicted results are in good agreement with experimental VLE data with an average relative deviation of 10%. The parameters of the thermodynamic model can be used to simulate and optimize CO2 absorption process.
Experimental section
Materials
The molecular formula, CAS number, purity, and source are reported in Table 5. The aqueous solutions are prepared using ultrapure water, which was prepared using a Center 120 FV-S ultrapure water generator. All reagents were used as supplied without any further purification.
Specifications and sources of the chemicals used in this work.

Used without any further purification.
Apparatus and procedure
The constant-volume method, combined with gas chromatography analysis was used to measure the solubility at different concentrations. The schematic diagram of the CO2 absorption measurement device is presented in Figure 1. A detailed description of the vapor–liquid equilibrium apparatus can be found in the literature.
29
It is comprised of two stainless steel tanks for the buffer and reaction. Both tanks are equipped with temperature probes and pressure transducers. The principle of this method is to absorb a known volume of gas known volume of polyamine solution. The amount of CO2 gas introduced into the reaction tank
At low partial pressure (<10 kPa), the pressure transducer error may have a greater impact on the results. Therefore, the CO2 partial pressure was obtained using gas chromatography (Agilent 7890). The apparatus was verified by determining the solution of CO2 in 30 mass% MEA solution at T = 313.15 and 393.15 K. The results are compared with reported data.25,30,31 Our results are in agreement with reported data, and details are given in our previous work. 32 The solubility of DETA aqueous solutions with mass fractions of 10% and 20% at 313.15, 343.15, 373.15, and 393.15 K are measured using the above-mentioned CO2 absorption device. The values are shown in Table 6.
CO2 solubility (

Standard uncertainties u(c) = ±0.01 mol/L, u(T) = ±0.1 K, ur (p) = ±0.5%, ur(
Supplemental Material
supporting_information – Supplemental material for Experimental and an electrolyte non-random two-liquid model to predict the vapor–liquid equilibrium of CO2 in aqueous solutions of diethylenetriamine
Supplemental material, supporting_information for Experimental and an electrolyte non-random two-liquid model to predict the vapor–liquid equilibrium of CO2 in aqueous solutions of diethylenetriamine by Ruilei Zhang, Yandong Tang, Weifeng Shan, Haijun Liu, Haijun Li and Jian Chen in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Langfang Science and Technology Project (Grant No. 2018013116), the Fundamental Research Funds for the Central Universities (Grant No. ZY20180241), and the Key Research and Development Program of Hebei Province (Grant No. 18273718).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.