Abstract
The acidic hydrolysis of astilbin to produce its aglycone, taxifolin, was investigated in this study. The effects of aq. HCl concentration and temperature on the reaction were studied, and the kinetic parameters were calculated. The results showed that with higher aq. HCl concentration and temperature, the hydrolysis of astilbin became faster. The activation energy of the hydrolysis reaction under 1 mol L−1 aq. HCl was calculated with a value of 148.6 kJ mol−1. The reaction was successfully applied to produce taxifolin from a sample of Rhizoma Smilacis Glabrae. A simple method for the purification of taxifolin from Rhizoma Smilacis Glabrae was developed with purity of 97.5%.

Taxifolin, also called dihydroquercetin, is a flavanonol commonly found in many plants, such as onions, 1 milk thistle, 2 and particularly, with high content, in the genus of Larix, for example, Dahurian Larch (Larix gmelinii Rupr). 3 Modern pharmacological studies have shown that taxifolin possesses many bioactivities and may have therapeutic promise for major disease states such as cancer, cardiovascular disease, and liver disease. 4 Theriault et al.5,6 found that taxifolin could significantly suppress the syntheses of cholesterol, triacylglycerol, and phospholipid in HepG2 cells by inhibition of the activity of HMG-CoA reductase, diacylglycerol acyltransferase, and triglyceride transfer protein. Sato et al. 7 found that taxifolin has the ability to prevent the aggregation and β-sheet formation of amyloid β-proteins, and may be a worthy candidate for Alzheimer disease therapeutics. More recently, the European Food Safety Authority confirmed the safety of Dahurian Larch extracts with taxifolin content > 90%. 8 The extract is intended to be added as an ingredient to many foods, for example, non-alcoholic beverages, yogurt, and chocolate confectionery. 8 Due to the safety and health promoting activities of taxifolin, it is the active component of many health products and drugs in America and Europe, such as silymarin (Legalon™), 4 Pycnogenol®, 4 and Ascovertin (a complex of taxifolin and vitamin C). 9
Dahurian Larch is usually the resource for taxifolin preparation at present. However, this tree grows in the most northerly area in the world with extremely stressful environmental conditions. 3 Thus, the tree is slow growing. Hence, to develop modern medicaments using taxifolin, a search for more plant sources and the development of simple and convenient methods for taxifolin purification is needed. Rhizoma Smilacis Glabrae (RSG) is a herbal material commonly used in China. Phytochemical analysis showed that astilbin is the dominant flavonoid in RSG with content ranging from 0.15% to 4.7%.10,11 Astilbin is the rhamnoside of taxifolin. Considering that the glucosidic bond could be hydrolyzed by acid treatment, RSG is a potential resource for taxifolin production. Acidic hydrolysis is a common chemical method in cleaving glucosidic bond, which has been extensively used to produce the aglycones of natural glycosides, such as saponin 12 and flavonoids.13,14 However, to the best of our knowledge, the specific acidic hydrolysis of astilbin to produce its aglycone has not been studied yet. Hence, in this study, the kinetics of acidic hydrolysis of astilbin was studied, and a method for the preparative purification of taxifolin from RSG was developed.
Results and discussion
Astilbin is the rhamnoside of taxifolin. Under acidic condition, it can be hydrolyzed to produce its aglycone, taxifolin (Scheme 1). Figure 1(a) shows the high-performance liquid chromatography (HPLC) chromatogram of astilbin after incubation with 1 mol L−1 aq. HCl at 80 °C for different times. As shown, after 10-min incubation, the peak area of astilbin decreased significantly, and a new peak, which is taxifolin, was found in the chromatogram. The retention time of astilbin was 15.4 min, and taxifolin was 0.6 min behind. After 45 min, the peak of astilbin disappeared, and only taxifolin was found in the chromatogram. Figure 1(b) shows the change of peak areas due to astilbin and taxifolin with time during the incubation. The peak area of astilbin quickly decreased with time, while that of taxifolin steady increased. After 20 min, only 12.5% of the initial astilbin was left. After 45 min, all astilbin had been hydrolyzed. The total peak area of astilbin and taxifolin slightly decreased with the time, which meant that a small part of these flavanonols had decomposed.
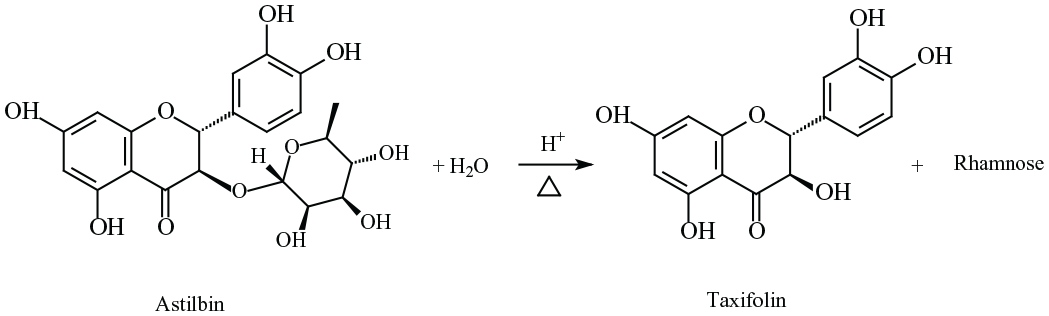
The acidic hydrolysis equation of astilbin.

(a) The chromatograms of astilbin before and after hydrolysis; (b) peak area change of astilbin and taxifolin versus time during hydrolysis. Hydrolysis was conducted with temperature of 80 °C and aq. HCl concentration of 1 mol L−1. Peaks: 1—astilbin; 2—taxifolin.
As shown in Scheme 1, the hydrolysis of astilbin is a second-order reaction. However, because the concentration of water remains almost constant in the solution, the reaction could be considered as pseudo-first-order. Thus, a first-order reaction model was applied for kinetic analysis of astilbin hydrolysis under various conditions. The model is expressed as
where C0 and Ct are the concentrations of astilbin at 0 and t min under the given hydrolysis conditions (in this study, the concentrations were represented by the peak areas), respectively. Parameter k is the hydrolysis rate constant (s−1). The time needed for 50% astilbin hydrolysis is called the half-life time (t1/2), and is calculated by the following equation
The major factors influencing the acidic hydrolysis of flavonoid glycosides are acidity (reflected by aq. HCl concentration) and temperature.13,14 Figure 2 plotted the calculated k and t1/2 versus HCl concentration. All linear fitting coefficients for the k calculation were bigger than 0.99, which implied the accuracy of pseudo-first-order estimates. As shown, the values of k rapidly increased, while t1/2 quickly decreased, with the rise of aq. HCl concentration. With 0.25 mol L−1 of aq. HCl, the values of k and t1/2 were 1.4 × 10−4 s−1 and 4968 s, respectively. The k value increased to 2.1 × 10−3 s−1, while t1/2 decreased to 324 s with 2 mol L−1 of aq. HCl. These results showed that the hydrolysis of the glucosidic bond in astilbin increased with increasing acidity. It was also found that with 0.25 mol L−1 of HCl, the total area of astilbin and taxifolin was equal to the initial area of astilbin, and was almost unchanged with incubation time (data not shown). The results implied that the molar absorption coefficient of taxifolin was equal to that of astilbin, and they were relatively stable under such acidic conditions. However, their total area decreased more quickly with 2 mol L−1 of aq. HCl than that with 1 mol L−1 of aq. HCl, which meant the decomposition of astilbin and taxifolin was faster. The decomposition of flavonoids under strong acidic condition was found in many other studies.13,14

The calculated k and t1/2 versus aq. HCl concentration.
The hydrolysis rate constants of astilbin with 1 mol L−1 of HCl at different temperatures are listed in Table 1. As shown, the k values quickly increased with the rise of temperature (T). According to the Arrhenius equation, the activation energy (Ea) of the reaction can be calculated from the intercept by plotting lnk versus 1/T to give a straight line. In the present, the linear coefficient (R2) of lnk versus 1/T was 0.954. The Ea of the hydrolysis of astilbin using 1 mol L−1 of aq. HCl was 148.6 kJ mol−1. The relatively high activation energy implied that the hydrolysis could hardly occur at room temperature. Even at 60 °C, the k value was as small as 0.89 × 10−4 s−1 and t1/2 was as long as 7800 s. However, acidic hydrolysis at high temperatures (e.g. >90 °C) may also cause the degradation of flavonoids. 14 Hence, based on the optimization study, hydrolysis conditions with temperature of 80 °C, aq. HCl concentration of 1 mol L−1, and duration of 1 h were chosen for RSG extract hydrolysis in this study.
The calculated k, t1/2, sand Ea of astilbin hydrolysis with 1 mol L−1 of aq. HCl at different temperatures.

In Figure 3, line a is the chromatogram of RSG extract before hydrolysis. The biggest peak in the chromatogram is astilbin, which is the dominant flavanonol in RSG. 15 After hydrolysis, the peak of astilbin disappeared from the chromatogram (line b). However, the peak of taxifolin was found (peak 2 in chromatogram b). The retention time difference between astilbin and taxifolin was 0.6 min, which was in accordance with Figure 1(a). Besides RSG, astilbin was also found in many other Smilacaceae genus plants, for example, Smilax corbularia 16 and Smilax china. 17 Sakurai et al. identified two new taxifolin-O-glucosides in Taxillus kaempferi (DC.) Danser. 18 As revealed in this study, the glucosidic bond of taxifolin glucosides could be successfully hydrolyzed by acid treatment. Hence, these plants may also be the potential resource for taxifolin production.

The chromatograms of RSG extract before (a) and after hydrolysis (b), and the purified taxifolin product (c). Peaks: 1—astilbin; 2—taxifolin.
A simple method for the preparative separation of taxifolin from RSG was developed. The total flavonoids in RSG was first purified by a macroporous resin adsorption-desorption process. Then, the flavonoids were hydrolyzed with 1 mol L−1 aq. HCl at 80 °C for 1 h followed by ethyl acetate extraction and recrystallization from water to give taxifolin with purity of 97.5%. Using 1000 g of a sample of RSG, 3.96 ± 0.21 g of taxifolin was obtained. The taxifolin was further characterized by MS analysis (Figure 4). The molecular weight ([M–H]−, 303.0602 m/z) and fragmentation pattern (285.0463, 177.0246, 125.0285 m/z) were the same as those of taxifolin in the literature, 19 which confirmed its chemical identity.

The MS spectra of purified taxifolin product.
Conclusion
Astilbin is successfully converted into taxifolin through acidic hydrolysis. The reaction is acidity- and temperature-dependent. By the reaction, RSG is a potential resource for taxifolin production. A simple method for the purification of taxifolin from RSG through acidic hydrolysis of astilbin was developed with purity of 97.5%.
Experimental
Chemicals and materials
The sample of RSG was gift from Guandong Shixin Pharmaceutical Co., Ltd (Jieyang, China) and was authenticated by the HPLC fingerprint we developed previously. 15 The specimens are deposited in Jiangxi Key Laboratory of Natural Product and Functional Food. Astilbin (>98%) was purified from RSG in our laboratory and was identified by UV, IR, MS, and nuclear magnetic resonance (NMR). HPLC-grade acetonitrile was purchased from Anhui Tedia High Purity Solvents Co., Ltd (Anqing, China). All other reagents used were analytical grade.
HPLC analysis
A Symmetry C18 column (250 mm × 4.6 mm i.d., 5 μm; Waters, USA) and an Agilent 1260 HPLC system equipped with an autosampler and diode array detector (DAD) detector were used. The analysis conditions were in according to our previous work. 15 Acetonitrile (A) and 0.1% acetic acid aqueous solution (B) were used as the mobile phase with flow rate of 1 mL min−1 and linear gradient program of 0–15 min, 16%–20% A; 15–40 min, 20%–40% A. The injection volume was 10 μL, and the detected wavelength was 291 nm. The column temperature was set at 40°C.
Astilbin hydrolysis study
A 0.5 mL aliquot of astilbin (2 mg mL−1 in 50% methanol) was mixed with 9.5 mL of aq. HCl solution with different concentrations (0.25, 0.5, 1, and 2 mol L−1). The mixture was incubated in a water bath at different temperatures (60, 65, 70, 75, and 80 °C). The mixture was sampled for HPLC analysis at different time intervals.
Preparation of taxifolin from RSG through hydrolysis
RSG sample was homogenized and filtered through 40 mesh sieve. RSG sample (1000 g) was extracted twice by 10 L of 50% ethanol at room temperature with stirring for 30 min. After filtering, the extract was concentrated to about 10 L. Then, the extract was pumped through a H103 resin fixed bed. After adsorption, the resin was desorbed with 3 L of 80% ethanol. The eluent was concentrated to about 400 mL, and concentrated aq. HCl was added with final concentration of 1 mol L−1. The mixture was then incubated at 80 °C for 1 h for hydrolysis. After cooling to room temperature, the mixture was extracted three times using equal volumes of ethyl acetate. The ethyl acetate fraction was concentrated to dryness by vacuum rotary evaporation at 60 °C. Then, the residue was dissolved by about 200 mL of boiling water. The solution was stored under 4 °C for 48 h. Taxifolin crystals were dried at 60 °C after filtration. A schematic diagram of the preparation procedures was presented in Figure 5. The purity of taxifolin in the product was determined by HPLC with the calibration curve of Y = 28.94X, where Y was peak area and X was the concentration of taxifolin. MS analysis was performed on Q-TOF 5600-plus mass Spectrometer (AB Sciex Corporation, Foster City, USA). The mass spectrometer was operated in the negative ion mode. Ultrapure nitrogen was used as the ion source gas 1 (50 lb/in2), ion source gas 2 (50 lb/in2), and curtain gas (40 lb/in2). The Turbo Ion Spray voltage and temperature were set at −4500 V and 500 °C, respectively. Declustering potential, collision energy, and collision energy spread were set at 100, −40, and 10 V, respectively. Data acquisition was performed with Analyst 1.6 software (AB Sciex).

Schematic diagram for preparation of taxifolin from RSG through acidic hydrolysis.
Statistical analysis
Data were expressed as the mean ± standard deviation (SD) of triplicates. Origin 8.0 software (Origin Lab Co., Northampton, USA) was used for data analysis and plotting.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 31760461).


