Abstract
Catalysts based on inexpensive and abundant transition metals are necessary for effective environmental remediation. In this study, a series of CuMn-CeO2 catalysts is synthesized by a simple coprecipitation method. The properties of the as-prepared catalysts are characterized by X-ray diffraction, Brunauer–Emmett–Teller analysis, scanning electron microscopy–energy-dispersive spectroscopy, transmission electron microscopy, and X-ray photoelectron spectroscopy. The introduction of dopants results in rougher surfaces with greater surface areas and pore volumes as well as more surface defects. Furthermore, the dopant-added catalysts exhibit higher activities for benzene oxidation, as the temperature required for 90% benzene removal with Cu4Mn1-CeO2 (656 K) was 231 K lower than that required with CeO2. The superior performance of the dopant-added CeO2 catalyst is attributed to the generation of defect-rich surfaces due to the isomorphous substitution of Cu and Mn in the crystal lattice of CeO2.
Introduction
Benzene is a harmful pollutant produced in many industrial activities such as in coating, petrochemical, and chemical processes. 1 Because its inhalation can induce leukemia and other cancers, benzene is classified as one of the most hazardous volatile organic compounds (VOCs). 2 However, the negative effects of benzene can be mitigated by effective control. Catalytic combustion is a reasonable strategy for the environmental remediation of benzene. In particular, catalytic processes can substantially decrease the combustion temperature, thus decreasing the cost of benzene removal. Although noble metals (e.g. Pt, Pd, and Rh) have been used as active materials, due to their high costs, transition metals are being actively studied as replacements.3–5
Cerium dioxide (CeO2) is widely used as a catalytic material for environmental remediation processes such as hydrocarbon combustion and carbon monoxide oxidation.6,7 The usefulness of this material is attributed to both the low cost of Ce and the high oxygen storage capacity of CeO2, which results from the low redox potential between Ce3+ and Ce4+.8,9 The catalytic performance of CeO2 can be improved by doping with various active metals. Lattice substitution with metal dopants increases the catalytic performance via the generation of defect structures that are favorable for catalytic oxidation. 9 Enhanced catalytic activities have been reported for CeO2-based catalysts substituted with transition metals (e.g. Fe, Cr, Ni, Co, Cu, Mn, and V). 10
In particular, Cu–Mn composite oxide catalysts have been synthesized for use in CO and VOC oxidation processes.11–13 According to a previous study, the efficient redox reaction can occur between Cu and Mn (Cu2+ + Mn3+
The goal of this work was to prepare Cu- and Mn-substituted CeO2 catalysts to investigate the influence of the dopant metals on the catalyst properties. In particular, the correlation between the characteristics of dopant-added CeO2 and the catalytic activity for benzene oxidation was examined.
Results and discussion
Characterization of the catalysts
Three different CeO2-based samples and a pristine CeO2 sample for comparison were synthesized to examine the effect of Cu and Mn addition on the catalyst characteristics and catalytic performance. The elemental compositions of the as-prepared catalysts were analyzed by energy-dispersive spectroscopy (EDS) mapping. As listed in Table 1, the Cu and Mn contents of the catalysts are 3.5–4.6 wt% and 1.1–1.8 wt%, respectively. Notably, the Cu and Mn fractions are similar irrespective of the nominal composition, which suggests that there is a limitation on the incorporation of metals into the CeO2 crystal lattice.
Elemental compositions of the CeO2-based catalysts, as determined by EDS mapping.

EDS: energy-dispersive spectroscopy; NA: not applicable.
The crystal structures of the catalysts were identified based on the X-ray diffraction (XRD) patterns shown in Figure 1. In the pristine CeO2 sample, all the observed peaks can be indexed to the different crystal faces of CeO2 (JCPDS: 81-0792). Although the peak intensities are substantially decreased, the three dopant-added samples also exhibit the same diffraction patterns. This observation indicates that Cu or Mn compounds were not formed despite the addition of Cu and Mn precursors during the catalyst synthesis. Thus, it is considered that Cu and Mn are incorporated into the cubic lattice of CeO2 via isomorphous substitution of Ce4+ ions. The successful incorporation of dopant ions into CeO2 has been observed with other metals such as Zr, Gd, Bi, Nd, Sm, and Ag.8,16 As shown in Table 1, the amounts of Mn are relatively small compared with those of Cu, which is presumably related to the difference in the ionic radii of Cu2+ (0.8 Å) and Mn4+ (0.58 Å). In other words, as the ionic radius of Cu2+ is close in size to that of Ce4+ (0.87 Å), Cu2+ ions can be easily substituted for Ce4+ ions in the crystal lattice of CeO2.

Crystalline structures of the as-synthesized CeO2-based samples measured by XRD.
The N2 adsorption–desorption isotherms and pore diameter distributions of the CeO2-based samples are presented in Figure 2. All the samples exhibit type IV isotherms, as defined by IUPAC (Figure 2(a)). 17 Moreover, the observation of a hysteresis loop at relative pressures of 0.6–1.0 P/P0 indicates that each catalyst has a mesoporous structure (Figure 2(b)). From these isotherms, the specific surface area, mean pore diameter, and pore volume were calculated using the Brunauer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) methods.18,19 As listed in Table 2, the surface area and pore volume are significantly increased when Cu2+ and Mn4+ ions are incorporated into CeO2. This change is attributed to the development of more defects with the addition of Cu and Mn. 16 The formation of a defect structure means that the amount of oxygen vacancies on the catalyst surface increases. The introduction of dopant metals into the CeO2 lattice leads to a charge imbalance due to the lower oxidation numbers of the dopants. 20 The excessive negative charge can be compensated by desorption of negatively charged lattice oxygen, resulting in the formation of oxygen vacancies.

(a) N2 adsorption–desorption isotherms and (b) pore size distributions of the as-prepared CeO2-based catalysts.
Surface area, pore characteristics, and Ce3+/Ce4+ and Oads/Olatt ratios of the CeO2-based catalysts.

The chemical environments of Ce and O on the catalyst surface were investigated using X-ray photoelectron spectroscopy (XPS, Figure 3). Deconvolution of the Ce 3d XPS spectra gave eight peaks that correspond to either Ce3+ (901.7 and 882.9 eV) or Ce4+ (915.9, 906.7, 899.6, 897.1, 887.8, and 881.2 eV). 20 The O 1s XPS spectra were deconvoluted into three peaks at 534.2, 530.6, and 528.2 eV that are attributed to chemisorbed water (Owater), adsorbed oxygen (Oads), and lattice oxygen (Olatt), respectively. 9 It has been reported that oxidation reactions are affected by the concentrations of surface defects such as Ce3+ and oxygen vacancies. 21 Therefore, the Ce3+/Ce4+ and Oads/Olatt ratios were calculated, as summarized in Table 2. Among the samples, pristine CeO2 has the lowest Ce3+/Ce4+ ratio (0.086), which suggests that more defect structures are developed with the addition of dopants.

(a) Ce 3d and (b) O 1s XPS spectra of the CeO2-based samples.
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) analyses were employed to examine the morphological characteristics of the catalysts. The SEM micrographs show that all the catalysts have rough surfaces that are formed by the aggregation of primary particles (Figure 4). In the pristine CeO2 sample, most of the elementary particles are round, whereas irregular particle shapes are observed in the Cu- and Mn-doped catalysts. The TEM micrographs clearly show round particles in the CeO2 sample and distorted particles in the Cu1Mn4-CeO2 sample (Figure 5), with most of the particles having sizes of 40–70 nm. Furthermore, the surface roughness becomes relatively high in the dopant-added catalysts. This phenomenon reflects the formation of a structure containing more defects, as discussed earlier. The electron microscopy results indicate that the particles in each catalyst sample are homogeneous without phase separation, which is in agreement with the XRD results (Figure 1).

Surface morphologies of the CeO2-based samples obtained by SEM.

Primary particle sizes of Cu1Mn4-CeO2 and CeO2 as revealed by TEM micrographs.
Catalytic performance
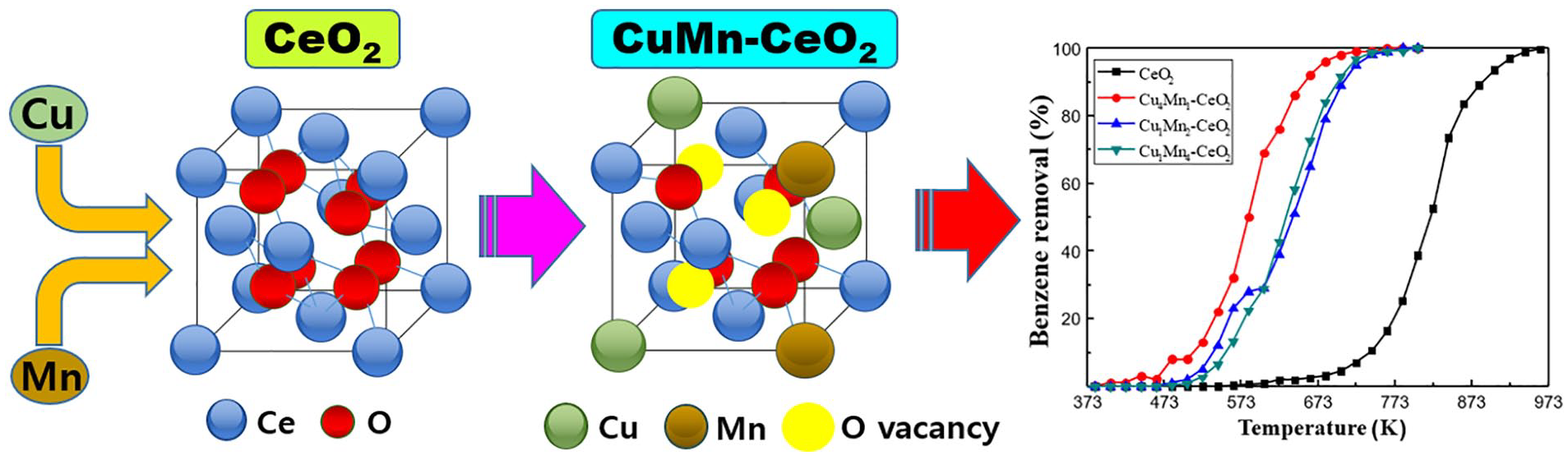
Figure 6 exhibits the catalytic activities of the as-prepared catalysts with respect to temperature. The catalytic reaction became activated at 400–480 K for the dopant-added samples, whereas activation occurred at 560 K with CeO2. Similarly, benzene is completely removed at approximately 740–780 K and 970 K for the dopant-added and pristine CeO2 catalysts, respectively. It should be mentioned that no secondary organic compounds were detected by gas chromatography–flame ionization detector (GC-FID) in the outlet gas, which implies that benzene-oxidized products were either carbon dioxide or monoxide. Table 3 shows the oxidation temperatures for removal of various fractions of benzene using each catalyst. The results indicate that Cu4Mn1-CeO2 has the highest catalytic performance among the four samples because it shows the lowest temperatures for removal of 10%, 50%, 90%, and 100% of benzene. The catalytic activity of Cu1Mn2-CeO2 is generally similar to that of Cu1Mn4-CeO2, with the former being slightly higher below 600 K, but slightly lower above 600 K. Overall, the conversion temperatures are substantially lower for the dopant-added samples than for CeO2. For example, Cu4Mn1-CeO2 can convert 90% of benzene at 656 K, whereas CeO2 requires a temperature of 887 K for 90% benzene removal.

Catalytic activities of the as-prepared CeO2-based catalysts for benzene oxidation.
Conversion temperatures for benzene oxidation with the CeO2-based catalysts.

The differences in catalytic performance are related to the surface area, Ce3+ concentration, and amount of surface-adsorbed oxygen (Oads). As shown in Table 2, among the samples, CeO2 has the lowest surface area and Ce3+ concentration, which is presumably the main reason for the poor performance of this catalyst. It is believed that the higher fraction of Oads/Olatt in CeO2 than in Cu1Mn2-CeO2 and Cu1Mn4-CeO2 was insufficient to achieve high performance due to the very low surface area of CeO2. In contrast, the surface area, Ce3+/Ce4+ fraction, and Oads concentration are greatly increased in the case of Cu4Mn1-CeO2, resulting in significantly improved catalytic activity.
According to Mars and Krevelen,
15
the catalytic oxidation of VOCs is activated by the adsorption of gaseous oxygen molecules (O2g). Efficient adsorption requires the generation of sufficient defect sites on the catalyst surface. Subsequently, the adsorbed oxygen molecules are activated to form “reactive” oxygen species (i.e.
Conclusion
Cu- and Mn-incorporated CeO2 catalysts were prepared to control benzene emissions. Catalyst characterization confirmed that the dopant metals were incorporated into CeO2 to form solid solutions. Elemental analysis revealed that the concentration of Cu was relatively higher than that of Mn, irrespective of the initial Cu/Mn ratio; this was ascribed to the facile substitution of Cu2+ ions for Ce4+ ions in the crystal lattice of CeO2 due to their similar ionic radii. The dopant-added catalysts exhibited higher catalytic activities than those of CeO2 over the entire temperature range. This performance enhancement can be explained by the augmentations in the surface area, Ce3+ concentration, and oxygen vacancies caused by the addition of dopants. The major findings of this study suggest that CeO2 isomorphously substituted with transition metals is promising for catalytic VOC oxidation due to its defect-rich structure.
Experimental
Synthesis of the catalysts
Cu- and Mn-substituted CeO2 materials were synthesized using Cu(NO3)2·3H2O (99%, Daejung Chemicals, South Korea), Mn(NO3)2·6H2O (97%, Junsei Chemical Co., Ltd, Japan), and Ce(NO3)3·6H2O (98%, Samchun Chemicals, South Korea) as Cu, Mn, and Ce precursors, respectively. Different samples were synthesized by varying the Cu/Mn ratio between 0.25 and 4. The total amount of Cu and Mn was fixed to 10 mol% irrespective of the Cu/Mn ratio. As a representative example, the sample with Cu/Mn ≒ 0.5 was synthesized as follows. First, 9.97 g of Ce(NO3)3·6H2O, 0.20 g of Cu(NO3)2·3H2O, and 0.49 g of Mn(NO3)2·6H2O were dissolved in 200, 19, and 20 mL of deionized water, respectively. The three solutions were then mixed together and 598 mL of a 40% NaOH solution was added slowly as a precipitating agent. After stirring for 1 h, the slurry was transferred to a polytetrafluoroethylene (PTFE) bottle and hydrothermally reacted at 373 K for 24 h. Next, the solid was collected by centrifugation at 5000 rpm for 3 min and washed with water and ethanol. The solid was dried at 343 K for at least 4 h. Finally, the solid was heat-treated at 673 K (heating rate: 1 K min−1) for 3 h in static air to obtain the final catalyst (Cu1Mn2-CeO2). The same procedure was used to synthesize the other catalyst samples, except that the initial reactant amounts were varied according to the desired Cu/Mn ratio.
Characterization
The crystal structures of the catalysts were analyzed by powder XRD (D/MAX-2200 Ultima/PC; Rigaku, Japan) using a Cu Kα radiation source over the diffraction angle (2θ) range of 5°–90°. The nitrogen adsorption characteristics at 77 K (BELSORP-max; BEL Japan Inc.) were used to determine the specific surface area, pore size, and total pore volume. These values were calculated based on the BET and BJH methods. SEM-EDS (Quattro S, FEI, Thermo Fisher Scientific, USA) was used to examine the morphology and the elemental dispersion of the catalysts. TEM (300 kV, Tecnai G2F30, FEI, Thermo Fisher Scientific, USA) was used to analyze the size of the catalyst particles. XPS (MultiLab 2000; Thermo Scientific) was used to identify the surface element species. A GC system (Master GC; DANI Instruments, Italy) equipped with an FID was used to analyze the benzene concentration in the outlet gas during catalytic tests.
Catalytic tests
Catalytic activity tests were conducted using a fixed-bed reactor with a quartz tube (I.D.: 10 mm). Catalyst powders with particle sizes of 90–180 μm were collected by sieving with 80–170 mesh sieves. A quartz wool plug was placed in the center of the quartz tube, and then the tube was filled with the catalyst powder to a height of 7.6 mm and another quartz wool plug was placed above the catalyst. First, a pretreatment test was carried out under flowing 1000 ppm-benzene/air at a rate of 100 mL min−1 at 373 K for 1 h to remove moisture and contaminants in the catalyst. Next, the temperature was increased from 373 to 973 K at a rate of 2 K min−1. During the catalytic test, 1000 ppm-benzene/air was introduced at a flow rate of 100 mL min−1, which corresponds to a gas hourly space velocity (GHSV) of 10,000 h−1. A portion of the outlet gas was injected into the GC system and analyzed at the intervals of 20 K. The rate of benzene removal (Rb,t) was determined using the following equation
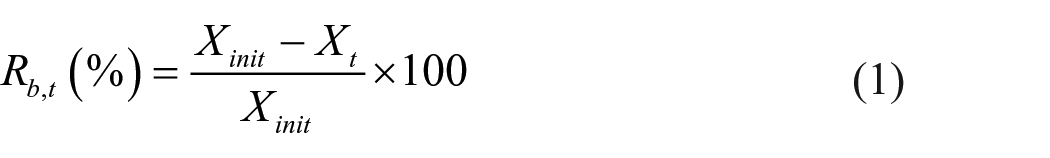
where Xinit and Xt are the initial benzene concentration and the benzene concentration at a specific temperature, respectively.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research fund of Chungnam National University.
