Abstract
Ni(NH3)4(AFT)2 [NiC6H16N18O2, AFT = 4-amino-3-(5-tetrazolate)furazan] is synthesized and characterized by elemental analysis and Fourier-transform infrared spectroscopy for the first time. X-ray diffraction measurements are used to determine the crystal structure of compound
Keywords

Introduction
The preparation and design of high-density and low-sensitivity energetic materials, including pyrotechnics, propellants and explosives, are the focus of intense research interest in the chemical and materials industries. Energetic materials have found significant use in military and civilian applications during the past century.1–10 Although the early energetic materials demonstrate considerable performance, they often generate exhaust gases that are harmful to the environment. Further research is directed toward finding a balance between excellent performance and the environmental friendliness of energetic materials. 11
Efficient methods to promote the performance of energetic materials involve the utilization of nitrogen-rich heterocycles such as furazan, triazole, pyrazole, imidazole, tetrazole and their derivatives, which satisfy the pursuit of high-density, high-oxygen content and pollution-free generated gases.12–16 They all possess high positive heats of formation due to the presence of a large number of energetic bonds such as N–N, N=N, and N–C.17,18 What is more, nitrogen-rich heterocycles such as furazan rings can ameliorate the performance of energetic materials, especially in terms of oxygen balance.19–22 Simple nitrogen-rich heterocycles materials are not a current focus in energetic material research, due to the requirements of higher energy and safer properties. Scientists have studied the combination of two or more nitrogen-rich heterocycles that can sharply promote energetic properties.23–27 The combination of tetrazole and furazan rings can eliminate the acidic proton and optimize the detonation properties.28,29 A furazan derivative, 4-amino-3-(5-tetrazolate)furazan (HAFT), first synthesized by Shreeve and colleagues, 30 is made up of amino, tetrazole, and furazan rings. This compound can be used to construct new multi-dimensional (one-, two-, three-dimensional (1D, 2D, 3D)) energetic metal-organic frameworks (MOFs) with several metal ions. Hence, a series of complexes of HAFT have been reported such as [Cd(NH2NH2)(AFT)2·0.7H2O]n, [Cd(NH2NHCOCONHNH2)1.5(AFT)2·5H2O]n, 31 [Cd(H2O)2(AFT)2], 32 [Cu(C3H6N2H4)2(AFT)2], [Cu(NH3)4(AFT)2] 33 and [Zn(NH3)4(AFT)2], 34 which meet the needs of insensitive impact and thermal stability.
Herein, we have synthesized the new energetic complex Ni(NH3)4(AFT)2. This complex will allow us to study the coordination chemistry of the metal ion. In addition, we have studied its crystal structure, thermal decomposition mechanism, and sensitivity properties.
Results and discussion
Structure description
Single-crystal X-ray diffraction (XRD) indicated that compound

Molecular structure of complex
Selected bond lengths (Å) and bond angles (°) for compound

Since there are many N and O atoms present in this compound, a significant number of hydrogen bonds are also present. As can be seen in Table 2, three intermolecular hydrogen bonds exist in the Ni complex. There is no intermolecular hydrogen bond between two Ni(NH3)4(AFT)2 molecules, which are connected by van der Waals forces. As shown in Figure 2, the hydrogen bonds N(8)–H(8C)···N(5), N(7)–H(7A)···O(1), N(8)–H(8A)···N(1) and N(7)–H(7A)···N(4) and the molecule of Ni(NH3)4(AFT)2 form a two-dimensional structure. As illustrated in Figure 3, compound
Hydrogen bond lengths (Å) and bond angles (°) for compound

Symmetry codes: #1 1 – x, −0.5 + y, 0.5 – z; #2 1 – x, 0.5 + y, −0.5 + z; #3 1.5 – x, −y, −0.5 + z.

The 2D chain of complex

Packing structure of complex
Thermal decomposition behavior
The thermal behavior of compound
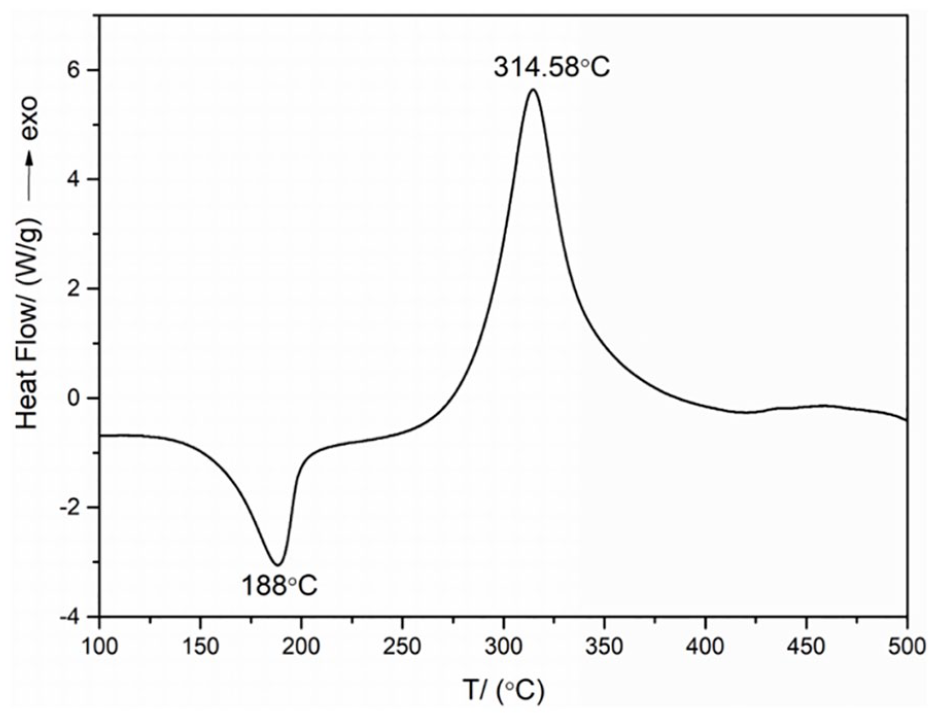
DSC curve of compound

TG-DTG curves of compound

TG-DSC curves of compound
Non-isothermal kinetic analysis
The apparent activation energy (Ek/Eo), pre-exponential factor (Ak), and linear correlation coefficients (rk/ro) have been calculated by Kissinger’s 36 and Ozawa-Doyle’s 37 methods. These data are given in Table 3. The equations for Kissinger’s and Ozawa-Doyle’s methods are as follows


in which the linear heating rate is β, the peak temperature (K) is Tp, A is the pre-exponential constant (s−1), the gas constant (8.314 J K−1 mol−1) is represented by R, and the apparent activation energy (kJ mol−1) is referred to as E.
Parameters determined from the DSC curves at different heating rates (β) for compound

Subscript k, data obtained by the Kissinger method; subscript o, data obtained by the Ozawa method.
Based on the above data, the kinetic parameters, including the extrapolated onset temperature (Te) and peak temperature (Tp), have been calculated. The values of Te0 and Tp0 (extrapolated temperature and peak temperature when the heating rates approach to 0) can be obtained through equation (3)

in which the coefficients are a, b and c.
The acquired Ea (the average of Ek and Eo) and Ak values can be used for expressing the Arrhenius equation of compound
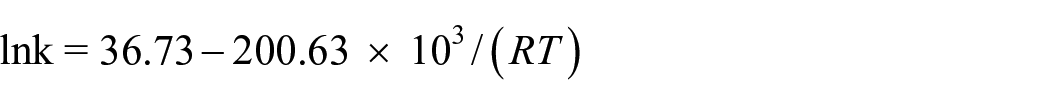
Thermal stability, safety parameters, and thermodynamic functions
As is already known, the self-accelerating decomposition temperature (TSADT or Te0), thermal ignition temperature (TTIT or Tbe0) and critical temperature of thermal explosion (Tb or Tbp0) are important parameters, which can ensure safe storage and process operations for evaluating the thermal stability of energetic materials. The TSADT, TTIT and Tb values of compound


Values of TSADT, Tbe0, Tbp0, ΔS≠, ΔH≠, and ΔG≠ for compound

The Ozawa method can be employed for obtaining the apparent activation energy (E0). In line with A and Ea, the thermodynamic parameters of the decomposition process of compound



where kB and h represent the Boltzmann constant (1.381 × 10–23 J K−1) and the Planck constant (6.626 × 10–34 J s), respectively.
Compared with Cd(pztza)2 and Cd(pzpha)(CH3OH)2,
38
the critical temperatures of which are 275.85 °C and 264.75 °C, compound
Energy of combustion and enthalpy of formation
The energy of combustion (ΔcHθ), the enthalpy of formation (ΔfHθ), and the constant-volume energy of combustion (ΔcU) of complex
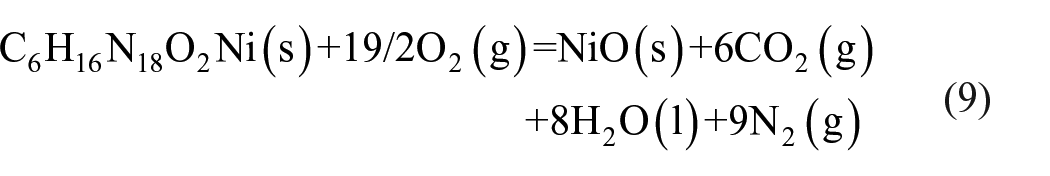

The standard molar enthalpy of combustion (ΔcHθ) can be verified by using equation (10). The parameter Δn is the change of the total molar amount of gases in the reaction process, and R and T are 8.314 J mol−1 K−1 and 298.15 K, respectively. The enthalpy of combustion of compound
Hess’ law and the combustion reactions were employed for obtaining the standard enthalpy of formation (ΔfHθ) of compound
Detonation property
The detonation parameters, including the detonation velocity (D), the detonation pressure (P), and the heat of detonation (Q), are significant for energetic performance evaluation in energetic materials. Currently, the largest exothermic principle proposed by Kamlet and Jacobs
39
is a valuable reference for research. Pang and colleagues
40
adopted a new empirical strategy to calculate these parameters that can be used as a basis to make predictions about metal-based energetic materials. It is easy to estimate the detonation reaction by using this strategy, which includes the arbitrary theory of Kamlet and Jacobs (K-J) and the hypothesis of the Becker–Kistiakowsky–Wilson (BKW) equation. The detonation reaction of compound
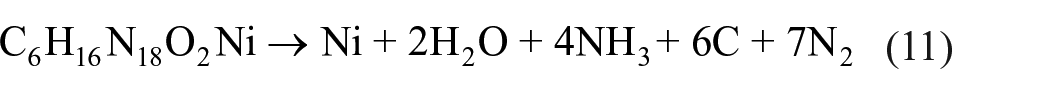




where D and P represent the detonation velocity (km s−1) and the detonation pressure (GPa), respectively; the density of explosive (g cm−3) is ρ; N can express the moles of detonation gases per gram of explosive; M is the average molecular weight of these gases; and the heat of detonation (kcal g−1) is referred to as Q.
We can easily obtain many details from the above equations. The detonation velocity (D), the detonation pressure (P), and the heat of detonation (Q) were 5.54 km s−1, 13.15 GPa, and 0.452 kcal g−1 (1.894 kJ g−1) for compound
Detonation properties of compound

From Table 5, it can be seen that. Zn(NH3)3(AFT)2 34 has the lowest detonation velocity (D). The highest detonation pressure (P) of 18.36 GPa is exhibited by Pb(HCONHNH2)2(AFT)2. As for the heat of detonation (Q), Cu(pn)2(AFT)2 has the highest value of the compounds presented in Table 5.
Specific heat capacity
The continuous specific heat capacity of compound
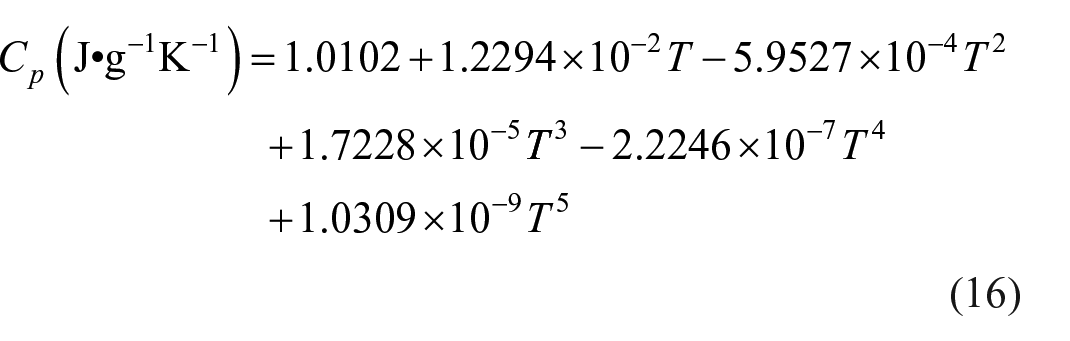
Impact sensitivity
The impact sensitivity (IS) is considered to be a significant property of energetic materials from the view point of personal safety. Energetic materials are required to be insensitive, which can reduce the risk of unnecessary accidents. The data on the IS were obtained using a drop hammer IS device with a 2.5 kg hammer weight at a height of 1.2 m. The experiment revealed that compound
Conclusion
In conclusion, Ni(NH3)4(AFT)2 has been synthesized for the first time and structurally characterized. Ni(NH3)4(AFT)2 crystallized in the orthorhombic space group Cmce. Moreover, the DSC and TG-DTG curves show that Ni(NH3)4(AFT)2 possesses one endothermic decomposition peak and two exothermic decomposition peaks, the former is sharp and the latter is smooth, and the thermal decomposition processes can be divided into three stages. The endothermic process is in the range of 130–510 °C with a peak temperature of 188 °C. The exothermic process occurs from 230 to 400 °C, with a peak temperature of 314.58 °C. The self-accelerating decomposition temperature (TSADT), thermal ignition temperature (TTIT), and critical temperature of thermal explosion (Tb) for Ni(NH3)4(AFT)2 are 265.69, 278.31, and 308.94 °C, respectively, which indicate good thermal safety. The non-isothermal kinetic analysis data indicate the Arrhenius equation can be expressed as lnk = 36.73 – 200.63 × 103/(RT). At the same time, complex
Experimental
Most energetic materials are flammable and explosive substances. In some experiments, many harmful substances are produced, such as strong acids and bases. Therefore, extreme care should be taken when conducting these experiments. Appropriate protective equipment must be used.
Materials and methods
All chemicals were commercially available reagents of analytical grade and used directly. A VarioEL III elemental analyzer (Elementar Co., Germany) was used to record the elemental analysis. An EQUINX55 infrared spectrometer was used to acquire the Fourier-transform infrared spectroscopy (FTIR) spectrum as a KBr pellet in the range of 4000–500 cm−1. DSC was performed using a DSC-Q2000 apparatus (TA, USA) under a nitrogen atmosphere at a flow rate of 50 mL min−1, with heating rates (β) of 5, 10, 15, and 20 °C min−1 from 70 to 470 °C. The TG-DTG experiment was implemented with an SDT-Q600 apparatus (TA, USA) under the conditions of a flow of nitrogen gas with a flow rate of 100 mL min−1 and a heating rate of 10.0 °C min−1. A ZBL-B IS instrument (Nachen Co., China) was used for determining the IS.
Preparation of HAFT
A three-step literature procedure was used to synthesized HAFT in Scheme 1. 43

Preparation of HAFT.
Synthesis of [Ni(NH3)4](AFT)2 (1 )
HAFT (0.3828 g, 2.5 mmol) was dissolved in deionized water (5 mL) and charged into a glass reactor with a water bath. The reaction solution was stirred with a mechanical agitator and heated at ambient temperature. Ni(NO3)2 (0.1914 g, 1.25 mmol) dissolved in 5 mL of deionized water was added to HAFT solution during 15 min. Then, aqueous ammonia solution (1 mL) was added to the above-mentioned reacting mixture under constant stirring. The solution was stirred for an additional 60 min. Finally, filter residue was filtered out. The mother liquor was sealed and left at room temperature (Scheme 2). Appropriate crystals for X-ray measurements were obtained after 2 months. m.p.: 155–155.8 °C. Anal. calcd. for NiC6H16N18O2: C, 16.72; H, 3.74; N, 58.50; found: C, 16.74; H, 3.72; N, 58.46. IR (KBr): 3491 (w, –NH2), 3365 (m, –NH), 1639 (m, –C=O), 1594 (m, N=N), 1200 (m, –C–N), 1171 (s, –C–N), 987 (m, –NH3), 865 (w, –NH2), 667 (s, –NH3).
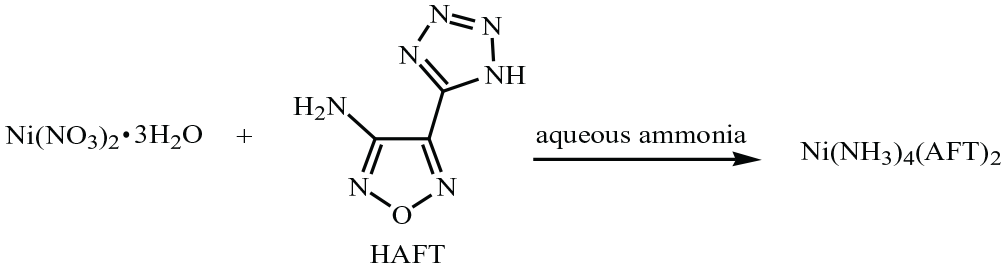
Synthesis of compound
Crystal structure determination and refinement
The single-crystal X-ray determination was carried out with a Bruker SMART APEX CCD X-ray diffractometer using graphite-monochromated Mo-Ka radiation (λ = 0.71073 nm) and an ϕ–ω scan mode at 296(2) K. The structures were resolved by direct methods (SHELXTL-97) and optimized by the full-matrix least-squares method on F 2 with anisotropic thermal parameters for all non-hydrogen atoms.44,45 The hydrogen atoms were determined with theoretical models. CheckCIF was used for checking the finalized Crystallographic Information File (CIF) file. The refinement parameters and crystallographic data for Ni(NH3)4(AFT)2 are collected in Table 6.
Crystal data and structure refinement details of compound

CCDC: Cambridge Crystallographic Data Centre.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are very grateful for the financial support from the National Natural Science Foundation of China (No. 21673168) and the Natural Science Basic Research Foundation of Shaanxi Province (No. 2018JM2061).
