Abstract
Porous carbon and La2O3/porous carbon materials are synthesized for the study of CO2 adsorption and separation by the volumetric method. The synthesized adsorbents are characterized by X-ray diffraction, N2 adsorption–desorption isotherms, Raman spectra and scanning electron microscopy with energy-dispersive X-ray analysis. Characterization results confirm the existence of porosity in the synthesized carbon materials and uniform distribution of lanthanum(III) oxide on porous carbon. The CO2 adsorption capacity for porous carbon and La2O3/porous carbon is 21 and 33 cm3 g−1, respectively, at 298 K and 1 bar. High adsorption of CO2 is obtained for La2O3/porous carbon because of the electrostatic interaction between La2O3 and CO2. Moreover, the N2 adsorption capacity is 2.8 cm3 g−1 for porous carbon and 2.2 cm3 g−1 for La2O3/porous carbon at 298 K and 1 bar. The change in N2 adsorption is due to the decrease in surface area. For La2O3/porous carbon, the selectivity of CO2/N2 is 33.5 and the heat of CO2 adsorption is 36.5 kJ mol−1 at low adsorption of CO2. It also shows constant CO2 adsorption capacity in each adsorption cycle.

Introduction
Global warming is a worldwide environmental issue which is contributed to carbon dioxide. It is liberated by the combustion of fossil fuels coinciding with the high growth of the automobile and petrochemical industries. 1 Day by day, the concentration of CO2 is increasing and has reached 400 ppm, which is more than pre-industrial levels. Hence, greater effort has to be made to control the CO2 concentration levels in the atmosphere. Carbon capture and separation (CCS) techniques on solid materials is an important method to lower CO2 concentration levels, as is the liquid amine solution absorption technique. In power plants, liquid amine solutions are mostly used to capture CO2. However, high energy is required for regeneration and occurs corrosion of the pipeline system. To overcome these drawbacks, solid materials are used for CCS. So far, materials such as activated carbon, 2 zeolites 3 and mesoporous silica 4 have been used for CO2 capture and separation.
Activated carbon is a cost-effective adsorbent and has a high surface area. However, the production of activated carbon on a large scale is hindered because of less renewable sources. Porous carbon synthesis from renewable sources is an important aspect. Renewable sources such as sawdust, 5 rice husk, 6 waste tea, 7 biodiesel industry solid residues, 8 cotton stalks, 9 olive and peach stones,10,11 palm oil ash 12 and coffee grounds 13 have been used to synthesize porous carbon materials. Pongamia pinnata fruit seeds are used for the production of biodiesel. During the production of biodiesel, a large quantity of pongamia pinnata fruit hulls is disposed of without commercial use. From these, porous carbon can also be synthesized by pyrolysis.
The CO2 adsorption of porous carbon can be enhanced by the incorporation of basic metal oxide and a heteroatom, which generates basic sites on the porous carbon. The Nitrogen-doped porous carbon-derived from Hazelnut-shell has shown CO2 adsorption capacity of 97 cm3 g−1 at 298 K and 1 bar.
14
Similarly,
Results and discussion
Structural characterization
Figure 1(a) shows the X-ray diffraction (XRD) of porous carbon and lanthanum oxide incorporated porous carbon. Two broad diffraction peaks were observed at 2θ = 24° and 43.5° for the (002) and (100) planes, respectively. These were the main characteristic peaks of porous carbon. 22 Moreover, lanthanum oxide–incorporated porous carbon showed diffraction peaks similar to porous carbon and no peaks related to La2O3 appeared. This indicated that La2O3 was highly dispersed on porous carbon and/or not detectable by XRD analysis. Chanapattharapol et al. 23 have also reported similar results with iron oxide doped MCM-41 for CO2 adsorption. The Raman spectra of porous carbon and La2O3/porous carbon are shown in Figure 1(b). Porous carbon shows Raman bands at 1334 and 1582 cm−1, which are related to the D-band and the G-band, respectively. 24 The D-band corresponds to disordered carbon, whereas the G-band corresponds to graphitic carbon. The ratio of the intensity of D and G-bands (ID/IG) was 0.87 for porous carbon and 0.95 for La2O3/porous carbon. The high ID/IG ratio of La2O3/porous carbon indicates a decrease in the graphitic nature of porous carbon by the incorporation of lanthanum oxide. 25

(a) XRD patterns, and (b) Raman spectra of porous carbon and La2O3/porous carbon.
The porosity of the synthesized adsorbents was determined by measuring the N2 adsorption–desorption isotherms at 77 K. Figure 2 shows the N2 isotherms of porous carbon and La2O3/porous carbon, and the textural properties are presented in Table 1. Pristine porous carbon showed high adsorption of N2 below the relative pressure P/P0 = 0.1 and a hysteresis loop between the relative pressure of 0.4 and 1.0. As per the classification of porous materials by IUPAC, the isotherm of the porous carbon material is similar to type I and type IV isotherms. 26 This indicated that the synthesized porous carbon has micropores and mesopores. The specific surface area, pore volume and pore size of porous carbon were 826 m2 g−1, 0.89 cm3 g−1 and 4.3 nm, respectively. Similarly, La2O3/porous carbon showed an isotherm curve similar to that of pristine porous carbon. However, the amount of N2 adsorption was less, which indicated that the pores of pristine porous carbon were occupied with lanthanum oxide. Hence, a change in the textural properties was observed. The specific surface area, pore volume and pore size of La2O3/porous carbon were 715 m2 g−1, 0.76 cm3 g−1 and 4.2 nm, respectively. Lou et al. 27 have also observed a change in textural properties by the occupation of ruthenium nanoparticles within the pores of porous carbon material.

(a) N2 adsorption–desorption isotherms (closed symbol: adsorption, open symbol: desorption) and (b) pore size distribution of porous carbon and La2O3/porous carbon.
Textural properties and metal composition of the synthesized samples.

BET surface area.
Total pore volume at P/P0 = 0.99.
Micropore volume obtained from the t-plot.
Mesopore volume obtained by subtracting Vmicro from Vtotal.
Average pore size by BET.
From EDX, NF: not found.
Morphological images with chemical composition for porous carbon and La2O3/porous carbon are shown in Figure 3. Disordered carbon particles were observed for porous carbon (Figure 3(a)). In La2O3/porous carbon, the loaded La2O3 covers the surface of the disordered porous carbon (Figure 3(b)). From energy-dispersive X-ray (EDX) results, the amount of lanthanum was 2.97 wt%.

SEM with EDX images of (a) porous carbon and (b) La2O3/porous carbon.
Study of CO2 and N2 adsorption
The adsorption of CO2 and N2 has been studied by the volumetric method using porous carbon and La2O3/porous carbon. The CO2 and N2 adsorption isotherms are shown in Figure 4. CO2 adsorption increased on increasing the pressure, but no equilibrium was attained for both adsorbents. The amount of CO2 adsorption was 21 cm3 g−1 for porous carbon and 33 cm3 g−1 for La2O3/porous carbon at 298 K and 1 bar. The high adsorption of CO2 with La2O3/porous carbon was due to the electrostatic interaction between lanthanum oxide and CO2. Along with the CO2 adsorption study, N2 adsorption was also studied under similar conditions to those used for the CO2 adsorption study. The amount of N2 adsorption was 2.8 cm3 g−1 for porous carbon and 2.2 cm3 g−1 for La2O3/porous carbon at 298 K and 1 bar. The decrease in N2 adsorption was due to a change in the surface area.
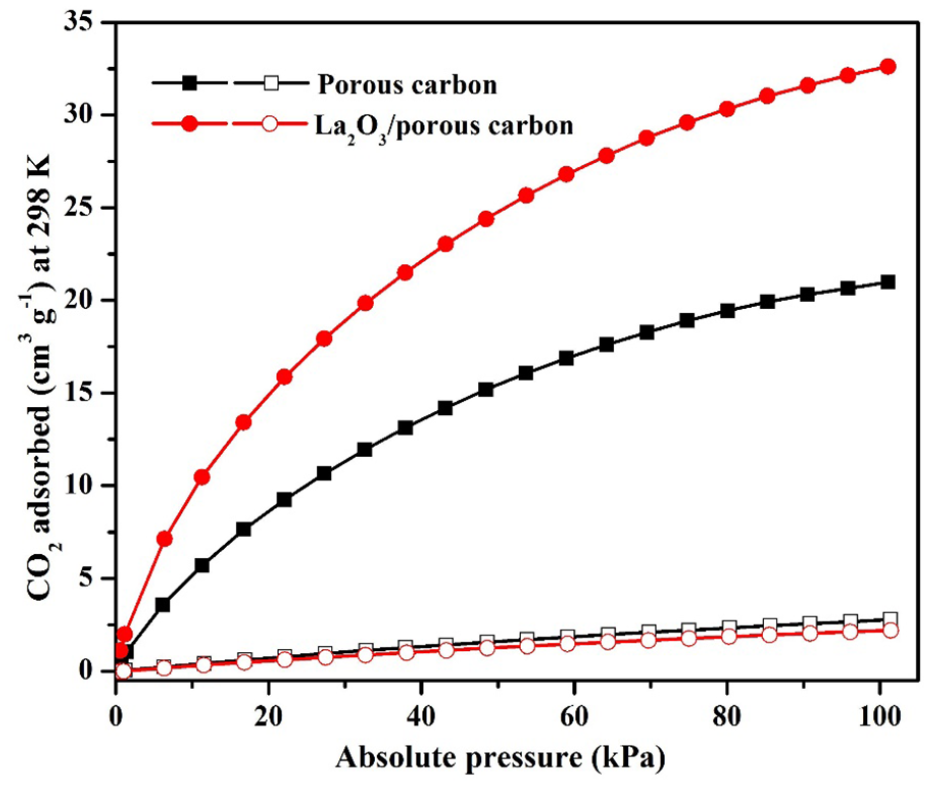
CO2 and N2 adsorption of porous carbon and La2O3/porous carbon (closed symbol: CO2 adsorption, open symbol: N2 adsorption).
In flue gas, carbon dioxide is a major component. So, it is essential to study CO2/N2 selectivity. The initial slope method was used to calculate CO2/N2 selectivity. 28 Figure 5 shows the CO2/N2 selectivity with porous carbon and La2O3/porous carbon. For porous carbon, the CO2/N2 selectivity was 14.5, whereas for La2O3/porous carbon, the selectivity was 33.5. High selectivity was obtained for La2O3/porous carbon because of the high adsorption of CO2 and the selectivity value was higher than those of some reported adsorbents such as HKUST-1 and Mg-MOF-74.29,30

CO2/N2 selectivity on porous carbon and La2O3/porous carbon.
The adsorption behaviour of an adsorbent can be calculated by fitting of the experimental CO2 adsorption with the Freundlich and Langmuir-Freundlich isotherm models. 31 The isotherm models can be written as follows

where Q is the adsorption capacity at equilibrium (cm3 g−1); Qmax is the maximum adsorption capacity (cm3 g−1); P is the pressure (kPa); kF and K are the Freundlich and Langmuir–Freundlich constants, respectively; and n is the heterogeneity factor. The experimental CO2 adsorption of porous carbon and La2O3/porous carbon are fitted with the Freundlich and Langmuir–Freundlich isotherm models, as shown in Figure 6. The fitting parameters are presented in Supplemental Table S1. The Langmuir–Freundlich isotherm model was well-fitted with the experimental CO2 adsorption, regression co-efficient R2 > 0.999. The Qmax was higher for La2O3/porous carbon because of the strong interaction of CO2 with lanthanum oxide.

Fitting of isotherm models for CO2 adsorption on porous carbon and La2O3/porous carbon.
In gas adsorption studies, the heat of adsorption (Qst) is an important parameter. It describes the interaction between the adsorbate and adsorbent. The Qst can be calculated using the virial equation 32
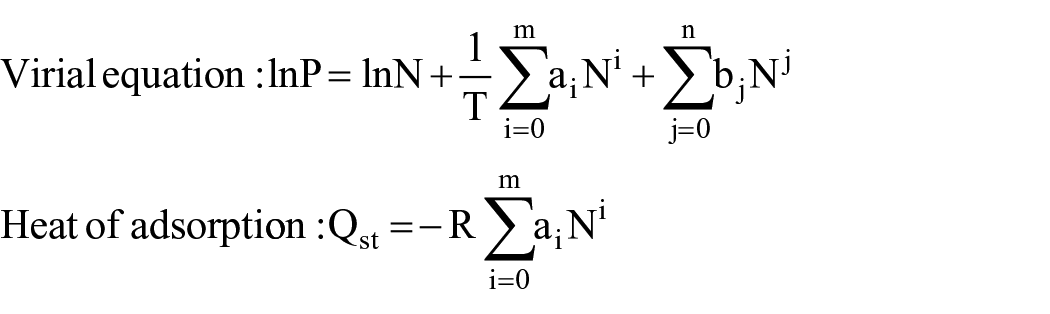
where P is the pressure in Torr, N is the gas uptake in cm3 g−1, T is the temperature in K, R is the universal gas constant (8.314 JK−1 mol−1), Qst is the heat of adsorption in kJ mol−1, ai and bj are the virial coefficients, and m and n are the number of coefficients. To calculate the heat of CO2 adsorption for porous carbon and La2O3/porous carbon, CO2 adsorption at 303 K was measured for both samples (see Supplemental Figure S1). The amount of CO2 adsorption was less at 303 K compared with CO2 adsorption at 298 K because of the increase in the kinetic energy of CO2. The measured CO2 adsorption isotherms were fitted with the virial equation (Supplemental Figure S2). From the virial fitting parameters, the heat of CO2 was calculated. The heat of CO2 adsorption of porous carbon and La2O3/porous carbon is shown in Figure 7. For porous carbon, Qst was 18.9–14.5 kJ mol−1, whereas for La2O3/porous carbon was 36.5–33.4 kJ mol−1. A high Qst was obtained for La2O3/porous carbon because of the strong interaction between CO2 and lanthanum oxide. At low adsorption of CO2, a high Qst was obtained. With an increase in CO2 adsorption, the Qst decreased due to a decrease in the number of active adsorption sites.

The heat of CO2 adsorption of porous carbon and La2O3/porous carbon.
Multiple CO2 adsorption cycles have been studied to determine the adsorption stability of La2O3/porous carbon at 298 K (Figure 8). Before studying the CO2 adsorption cycle, the adsorbent was degasified at 473 K for 2 h under vacuum to remove the adsorbed CO2. The amount of CO2 adsorption was constant in each adsorption cycle. The CO2 adsorption of La2O3/porous carbon was compared with the CO2 adsorption capacity of previously reported adsorbents (Table 2). The synthesized La2O3/porous carbon showed 33 cm3 g−1 of CO2 adsorption at 298 K and 1 bar, which was in-between the CO2 adsorption capacity of mesoporous N-doped CeO2 and S-doped microporous carbon.34,36
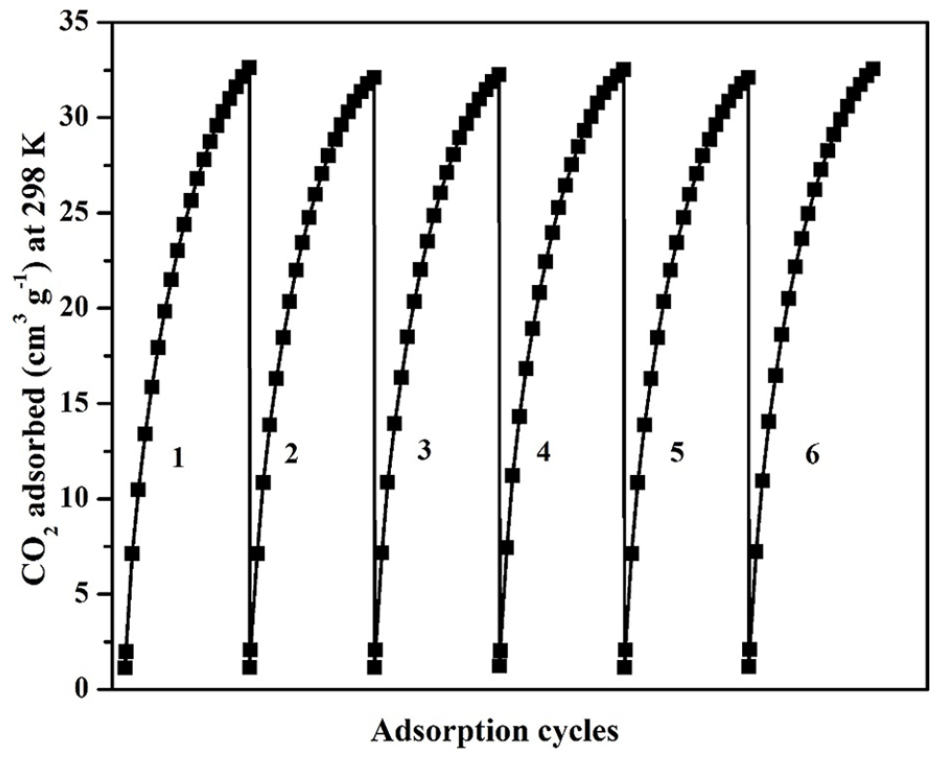
Multiple CO2 adsorption cycles of La2O3/porous carbon.
Comparison of the CO2 adsorption capacity of La2O3/porous carbon with reported adsorbents.

Conclusion
In this work, we have studied CO2 adsorption and separation by the volumetric method using porous carbon and La2O3/porous carbon. The high CO2 adsorption capacity was obtained on La2O3/porous carbon compared to bulk porous carbon at 298 K and 1 bar because of electrostatic interaction of La2O3 with CO2 and CO2/N2 selectivity was also higher on La2O3/porous carbon. The heat of CO2 adsorption was 36.5 kJ mol−1 at low coverage of CO2 for La2O3/porous carbon and CO2 adsorption capacity was constant in each adsorption cycle. Therefore, a basic metal oxide can be incorporated on porous carbon to increase the CO2 adsorption and separation.
Experimental
Lanthanum nitrate hexahydrate [La(NO3)3.6H2O, 99.9%] and orthophosphoric acid (H3PO4, 85%) were purchased from Sigma-Aldrich, India, and used without purification. Distilled water was used to synthesize the adsorbents. High purity gases (carbon dioxide, nitrogen and helium) were purchased from BOC, India.
Porous carbon was synthesized by using pongamia pinnata fruit hulls which were collected from the forest region of Telangana, India. First, the fruit hulls were washed, dried then made into a powder. The powder was activated using phosphoric acid at room temperature for 24 h followed by drying at 373 K for 12 h. The dried sample was calcined under nitrogen gas with a flow rate of 50 mL/min at 723 K for 4 h, with a heating rate of 5 K/min, and then cooled to room temperature. The sample was washed with distilled water until the pH reached 7, then it was dried at 373 K overnight to afford porous carbon. 39 Porous carbon supported lanthanum oxide was synthesized by the impregnation method. 40 About 0.1 g of lanthanum nitrate hexahydrate was dissolved in 10 mL of distilled water, stirred for 10 min then 1 g of porous carbon was added. The mixture was stirred at room temperature for 1 h and then dried at 373 K overnight. The dried compound was calcined under nitrogen gas at 873 K for 3 h.
XRD patterns were recorded on a Rigaku Ultima-IV X-ray diffractometer using Ni-filtered Cu-Kα radiation operated at a voltage of 40 kV and a current of 30 mA in the scan range of 2θ = 10°–80° with a step size of 0.02°/s. A Micrometric ASAP 2020 porosity analyzer was used to measure N2 adsorption–desorption isotherms at 77 K. Prior to the adsorption study, about 0.1 g of the sample was degassed at 473 K for 2 h under vacuum. The multipoint Brunauer–Emmett–Teller (BET) surface area was calculated in the relative pressure range of 0.05–0.3, the total pore volume at a relative pressure of 0.99, the micropore volume by the t-plot method and the mesopore volume was calculated by subtracting the micropore volume from the total pore volume. The pore size distribution was plotted using non-local density functional theory. A LabRAM HR800 spectrometer was used to record the Raman spectra. The morphology and elemental composition of each sample were determined from scanning electron microscopy with energy-dispersive X-ray (SEM with EDX) analysis using a ZEISS Sigma 300 scanning electron microscopy analyzer.
The adsorption of CO2 and N2 was carried out using a Micromeritics ASAP 2020 gas adsorption analyzer at low pressure and at 298 K. A thermostatic bath connected with water circulating jacket was used to control the sample temperature, and helium gas was used to determine the free space of the sample tube. Before the gas adsorption measurement, 0.1 g of the sample was activated at 473 K for 2 h under vacuum to remove moisture or adsorbed gases and then cooled to the gas adsorption temperature. Ultra-high pure gases were used to measure the adsorption isotherms. The initial slope method was used to calculate the selectivity of CO2/N2. The virial method was used to calculate the heat of CO2 adsorption using adsorption isotherms obtained at 298 and 303 K. The CO2 adsorption cycles were also studied at 298 K to calculate the adsorption stability of an adsorbent.
Supplemental Material
Supplementary_material_7 – Supplemental material for Study of CO2 adsorption and separation using modified porous carbon
Supplemental material, Supplementary_material_7 for Study of CO2 adsorption and separation using modified porous carbon by Madhavi Jonnalagadda, Rumana Anjum, Harshitha Burri and Suresh Mutyala in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors acknowledge the Science and Engineering Research Board, Department of Science and Technology, New Delhi, India, for financial support (grant no. EMEQ-283/2014).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
