Abstract
3,7-Bis[4-(diphenylamino)phenyl]-5H-dibenzo[a,d][7]annulen-5-one, containing triphenylamine- and dibenzosuberenone-based donor-acceptor-donor (D-A-D) conjugated moieties, is synthesized in high yield by Suzuki coupling of 5H-dibenzo[a,d][7]annulen-5-one with [4-(diphenylamino)phenyl]boronic acid. The most important part of the present study is the creation of a dye-sensitized solar cell structure using the synthesized organic molecule for the first time and the power conversion efficiency of this structure. Three solar cell devices are prepared and are named as first device, second device, and third device. The power conversion efficiency (%) value for first device, second device, and third device is calculated as 2.54, 2.38, and 2.54, respectively. Moreover, the chemical structure of the synthesized compound is determined by nuclear magnetic resonance, infrared, ultraviolet visible, fluorescence spectroscopy, and high-resolution mass spectrometry.

Introduction
The tricyclic structure of 5-dibenzosuberenone is that of an aromatic compound that contains a tropone skeleton and is formed by the fusion of two benzene rings to this skeleton. This core skeleton is found in the structure of drugs marketed as antidepressants. This class of molecule has attracted attention from both synthetic and biological communities due to their interesting chemical structures and potential biological activities.1–6 5-Dibenzosuberenone is also of considerable interest because of its physical and chemical properties in physical, theoretical, and instrumental organic chemistry.7,8 Although dibenzosuberenone has many dibenzo derivatives, the 2,3;4,5-dibenzotropone (5H-dibenzo[a,d][7]annulen-5-one) form has been utilized most in organic synthesis due to its symmetrical structure. This core skeleton is widely used, especially in materials chemistry, in photovoltaic applications, as a light-emitting diode, as an organic light-emitting diodes (OLED) in organic electronic applications, and in field-effect transistors,9–11 as well as in new bipolar molecular materials with high optoelectronic performances.12,13 In addition, the core skeleton has found various applications in polymer synthesis14–18 and in metal–ligand complexes and in the formation of coordination cages. 19 However, triphenylamine (TPA) derivatives are known for their strong electron-donor and blue light-emitting properties. TPA cores have shown exceptional properties, such as good solubility in organic solvents, hole-conducting properties, high electron mobility, two-photon absorption, and excellent optoelectronic properties.12,20,21
Solar cells, including dye-sensitized solar cells (DSSCs), bulk heterojunction donor-acceptor blends, quantum dot solar cells, organic–inorganic hybrid perovskite solar cells, and tandem solar cells have received significant interest in the market for low-cost power and mobile energy generation.22–24 DSSCs, as a new form of green energy device, show important characteristics such as ease of manufacture, reasonably low manufacturing costs, low contamination, and good molecular design versatility. 25 Photosensitizers play a critical role among all the components of DSSCs, being responsible for light harvesting and then passing electrons to a broad band gap of a semiconducting oxide. Therefore, optoelectronic activities of sensitizing dyes are essential for the photovoltaic efficiencies of a DSSC. Hence, molecular engineering of sensitizing dyes is one of the most productive ways of advancing DSSC performance. Owing to their simple growth and consistent efficiency, one of the most popular design approaches of metal-free organic dyes includes the use of D-π-A structures.26,27
Classic metal-free sensitizers are centered on the D-π-A method to accomplish productive separation and transfer of charges. Molecular engineering of donor groups has advanced from the use of a basic moiety of TPA to a moiety based on starburst phenylene, to regulate electronic grades or molecular configuration, and to provide efficient movement of charge. 28 A π-tunneling device is a modern design function, affecting the highest energy occupied molecular orbital (HOMO)/lowest energy unoccupied molecular orbital (LUMO) energy levels or otherwise spectroscopic characteristics. 29
Herein, we report on the synthesis of 3,7-bis[4-(diphenylamino)phenyl]-5H-dibenzo[a,d][7]annulen-5-one (
Results and discussion
Synthesis
Dibromodibenzosuberenone (
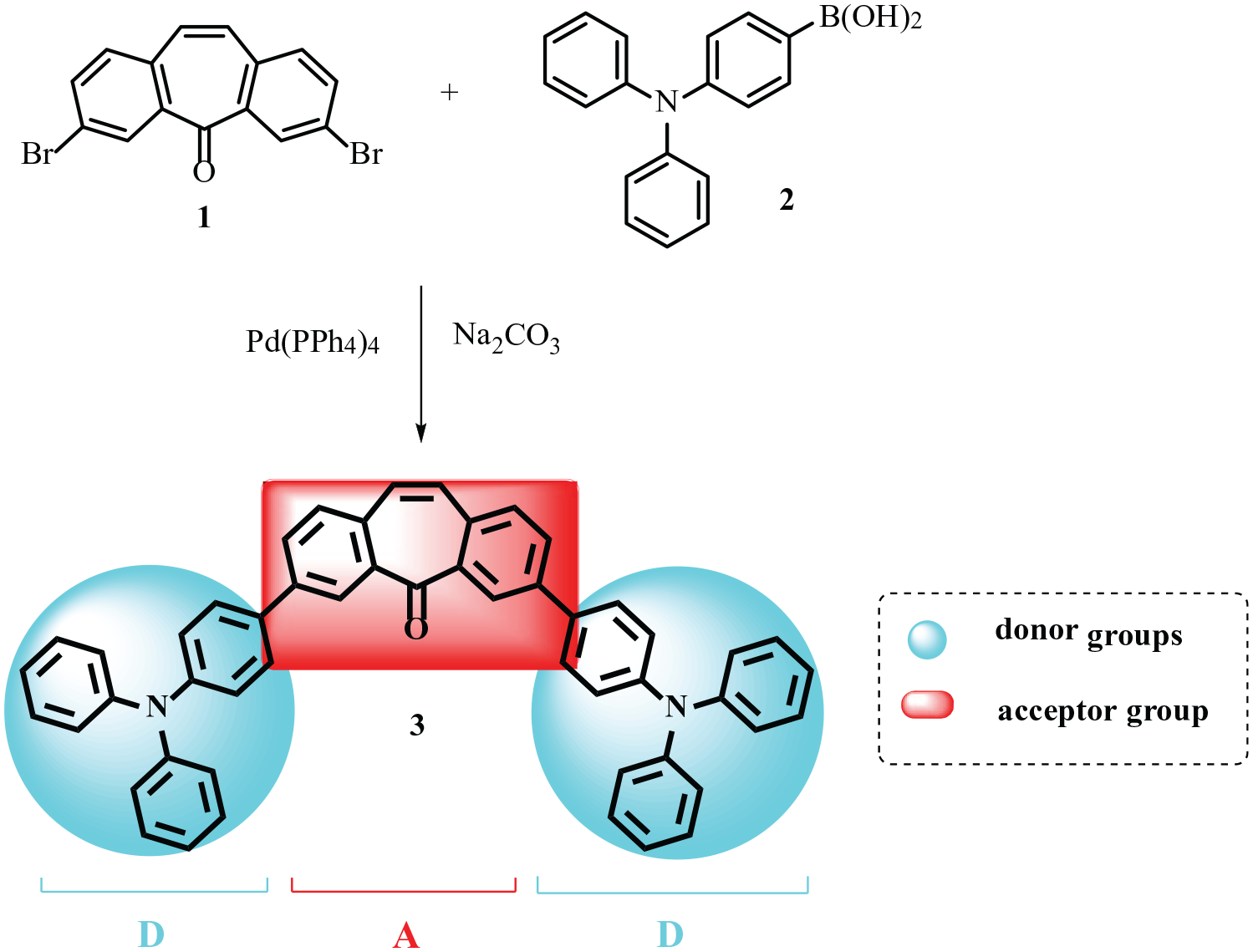
The synthesis of compound
NMR analysis
The 1H NMR spectrum of the compound

Structure of compound
Fourier transform infrared and HRMS analysis
The IR spectrum of
Photophysical properties
The result of the absorbance measurement on compound

Plot of the wavelength versus absorbance intensity for compound
As it is non-destructive and non-intrusive, photoluminescence (PL) is an essential technique for researching electronic equipment. In addition, the optical and electronic characteristics of a compound are closely related: a quantum system displays quantized energy states that are defined by the discrete wavelength of emission. The result of the PL measurement for compound

Plot of the wavelength versus fluorescence for the compound
There are significant benefits of organic molecules that are used in solar cell applications.
30
Organic molecules, in fact, involve more adsorption layers than metal materials and absorb a wider range.
31
The scope of organic materials is limitless and they can be quickly refreshed.
32
Furthermore, curiosity in pure organic molecules is rising as computer simulations to model such compounds are simpler than using actual metal compounds. In addition, compound
Photovoltaic properties
In the light of the information given above, current density (J)–voltage (V) measurements were performed to examine the photovoltaic properties of three compound

A plot of current density versus voltage for the device base on compound
The obtained short-circuit current density (JSC), the open-circuit voltage (VOC) current corresponding to the maximum power point (JMP), the voltage corresponding to the maximum power point (VMP), the fill factor (FF), and the power conversion efficiency (%) values for each device are given in Table 1.
JSC, VOC, JMP, VMP, FF, and calculated power conversion efficiency (%) values for each device.

The power conversion efficiency (%) values for the three devices were calculated as 2.54, 2.38, and 2.54 under illumination with a AM1.5, 100 mW/cm2 lamp using the relation given in our previous study.33,34 It is well-recognized that the rate of electron transport in a solar cell configuration will impact the power conversion efficiency (%) value of an organic molecule sensitized solar cell device. The achieved high short-circuit current density value for the first device and the third device indicates that effectual injection of electrons occurs into the conduction band (CB) of a counter electrode (TiO2) conduction band from the excited state of the organic molecule. Compared to the first and third solar cell devices, the second device has a slightly lower JSC value. One possible reason for the low JSC value is the inadequate amount of compound
Conclusion
We have successfully synthesized and characterized the triphenylamine-dibenzosuberenone derived D-A-D-based conjugated organic molecule
Experimental
General remarks
The one- and two-dimensional 1H and 13C NMR spectra were recorded on a Varian-400 or a Bruker-400 spectrometer in CDCl3 using tetramethylsilane as the internal reference. All spectra were recorded at 25 °C and coupling constants (J values) are given in Hertz. Chemical shifts are given in parts per million (ppm). Abbreviations used to define the multiplicities are as follows: d = doublet; dd = doublet of doublets; m = multiplet. Mass spectra were recorded on an Agilent Technologies 6530 Accurate-Mass Q-TOF-LC/MS. IR spectra were recorded on Perkin Elmer Fourier transform infrared (FT-IR) spectrometer. Absorption spectrometry was performed using a Perkin Elmer Lambda 35 spectrophotometer. Steady-state fluorescence measurements were conducted using a Shimadzu RF-5301PC spectrofluorometer. Absorbance and emission spectroscopy measurements were performed on 5 µm samples in CH2Cl2. The fluorescence quantum yield of compound
Synthesis of 3,7-bis[4-(diphenylamino)phenyl]-5H-dibenzo[a,d][7]annulen-5-one (3)
To a 50-mL, two-necked, round-bottomed flask was added dibromodibenzosuberenone (
Preparation of the DSSC devices
Zn2SnO4 nanowires were synthesized on gold-coated (3 nm) Si substrates following a procedure published elsewhere.
37
This process is able to reliably reproduce nanowire films roughly 20 μm thick. The nanowires are then transferred into fluorine-doped tin oxide (FTO) substrates following a printing procedure
37
and annealed at 500 °C for 4 h to transform the precursor on FTO into a Zn2SnO4 film. The annealed FTO substrates with the nanowires are placed in a suspension of compound
Supplemental Material
sup_mat – Supplemental material for Synthesis and characterization of a triphenylamine-dibenzosuberenone-based conjugated organic material and an investigation of its photovoltaic properties
Supplemental material, sup_mat for Synthesis and characterization of a triphenylamine-dibenzosuberenone-based conjugated organic material and an investigation of its photovoltaic properties by Musa Erdoğan and Sabit Horoz in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Research Foundation of Kafkas University for financial support under project 2019-FM-66.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
