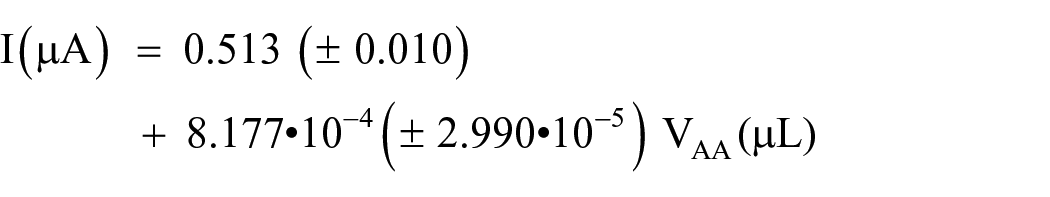Abstract
Chili peppers are rich in ascorbic acid and capsaicin. In this article is proposed an easy, fast, sensitive, and inexpensive method for determining the pungency and content of ascorbic acid in chili pepper extracts. The voltammetric (cyclic and differential pulse) behavior of capsaicin on a glassy carbon electrode has been evaluated at different pH values. A calibration curve has been obtained for the peak current, IP, of capsaicin as a function of the capsaicin concentration, C, in differential pulse voltammetry in phosphate buffer solution at pH 7.0: IP(µA) = 0.0147 (±5.346·10−3) + 0.0507 (±8.984·10−4) C(µM), with limit of detection, LOD = 0.198µM, limit of quantification, LOQ = 0.660µM, and dynamic linear range from 0.660 to 20.9 µM. A variant of the standard addition method has been used for simultaneous determination of the pungency and ascorbic acid content of extracts of Capsicum annuum cubana red. In this case, the calibration for ascorbic acid was I(µA) = 0.467 (±0.012) + 2.039·10−3 (±4.601·10−5) VAA(L)), with LOD = 17.56 µL, LOQ = 58.55 µL, and dynamic linear range from 58.6 to 500 µL, being VAA the volume of 10-mM ascorbic acid added to 50 mL of solution. The ascorbic acid content was compared to that of a sweet pepper. The method is cheap, simple, and fast (30 min vs c.a. 2 h compared to the spectrophotometric method), its sensitivity being comparable to other more expensive and/or more laborious methods.
Introduction
Capsaicin (8-methyl-N-vanillyl-6-nonenamide) is a phenolic compound responsible for the characteristic taste and pungency of chili peppers, representing up to 1% of their weights, being the most consumed condiment by humans, together with salt. In addition, capsaicin can also be used to relieve pain 1 and to relieve muscle aches, as well as some types of neuropathic pain with fewer side effects than other painkillers. 2 Moreover, it is the active substance in aerosol or pepper spray, which is used to disperse street riots, such as a non-lethal chemical weapon or a self-defense weapon, as it irritates the eyes causing tears, pain, and even temporary blindness. 3
Capsaicin has a nonpolar phenolic structure and thus is poorly solubilized in water. The mixed properties of ethanol allow its use as a solvent for this chemical. Therefore, its extraction in food depends on whether alcoholic beverages (such as wine, rum, brandy, and so on) are used in their preparation, which influences the spicy taste.
Capsaicin-containing spices also have beneficial effects by increasing the body temperature, which accelerates the heart and metabolism, this causing an increase in blood flow, a reduction in blood pressure, and a vasodilator effect that can dissolve blood clots. 4 If used sparingly, they are beneficial as they provide antimicrobial, analgesic, fungicide, bactericide, and anticancer properties. 5
The level of pungency of the different spices was first evaluated with the Scoville organoleptic test, which expresses the level of pungency on a scale called Scoville heat unit (SHU). 6 This approximation is subject to a great error (c.a. 50% for the same extract) due to the different perception of the operators. Since the pungency of pure capsaicin is 1.6 million SHU per gram, this parameter is now determined by measuring capsaicin plus the dihydrocapsaicin content in foods, in grams, and multiplying by the previous value.
Analytical separation, quantitation, and identification of capsaicin and dihydrocapsaicin in foods have been made by gas chromatography, 7 spectroscopy,8,9 capillary electrophoresis, 10 optical biosensors, 11 and, mainly, high-performance liquid chromatography (HPLC).12–14 The most accurate methods need sample manipulation and pretreatment, being long lasting and expensive, and requiring sophisticated instrumentation. But fast, simple, and inexpensive methods of food analysis are demanded. To meet these requirements, electrochemical techniques such as electrochemical sensors based on carbon nanotubes,15–18 graphene,19,20 functionalized graphite, 21 nanoparticles, 22 or ionic liquids 23 have been proposed. The use of a bare glassy carbon electrode without extra materials is definitely more advantageous than those requiring extra materials, as long as the sensitivity is in the needed range. Glassy carbon electrodes are cheap and easily available. In addition, peppers in general, and chili peppers in particular, are plants rich in ascorbic acid. 9
The objective of this article is to propose an easy, fast, sensitive, and inexpensive method for determining the pungency and ascorbic acid content in chili pepper extracts (Figure 1).

Structures of the compounds studied.
Results and discussion
The voltammetric characteristics of capsaicin and dihydrocapsaicin (oxidation potentials and peak currents) are virtually the same, 24 which also occurs also with the spectroscopic responses. 9 This is because the difference between both molecules, a double bond, is far from the electroactive group and the diffusion coefficients are virtually the same. This makes it possible to determine the pungency, mainly due to the presence of these two capsaicinoids, by determining both compounds from electrochemical (or spectroscopic) measurements.
The cyclic voltammetric behavior of capsaicin on carbon electrodes has been previously reported in the literature. 24 To select the measuring pH, cyclic voltammograms were recorded at different pH values, and the direct scans of the cyclic voltammograms at some pH values are shown in Figure 2. These results agree with those previously published. A main oxidation peak is observed at potentials low enough to avoid the usual interferences appearing in natural samples, with the exception of ascorbic acid, as will be discussed below.
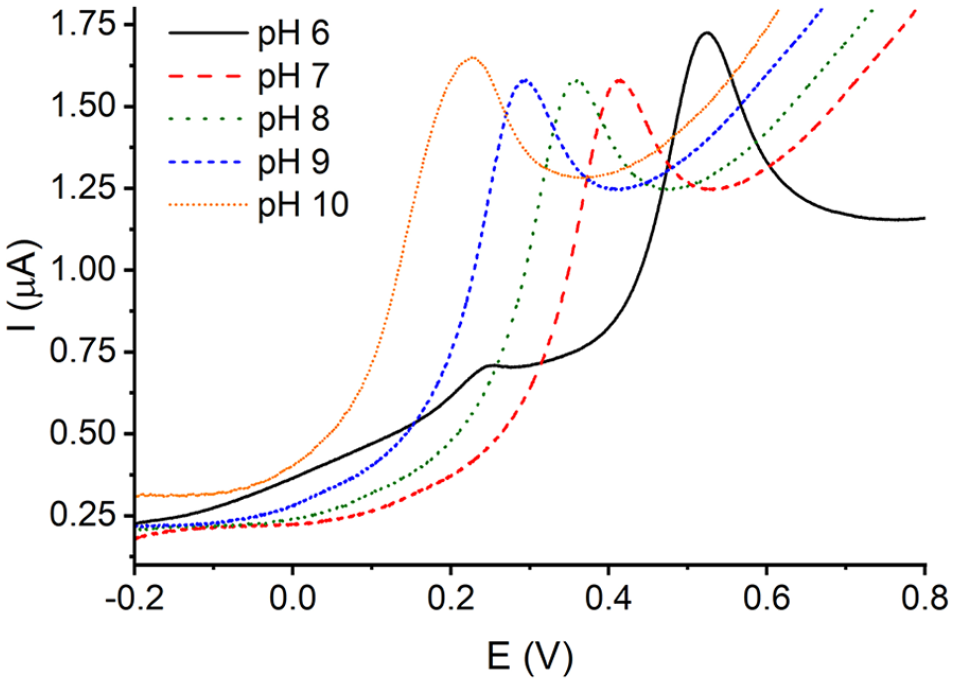
Direct scans on the cyclic voltammograms of 2·10−5 M capsaicin in PBS solutions of different pH at 0.1 V s−1. Scan from negative to positive potentials.
The shape of the curve obtained at pH = 10 is distorted, probably due to the occurrence of at least two processes appearing at very close potentials. A similar behavior can be observed in the curve for pH = 6, which shows some signals at potentials lower than the main peak. In these cases, the signals should not be used for the quantification of capsaicin if this is feasible. The rest of signals are suitable for this purpose and pH 7.0 was selected, first, because the PBS buffer has its maximum buffering capacity near this pH value and, second, because PBS at pH = 7 is a reaction medium widely used in food chemistry. The determinations were made by using differential pulse voltammetry (DPV) because the high sensitivity of the technique. Figure 3 shows some results at different capsaicin concentrations and the plot of the net peak current versus concentration.

Top: Differential pulse voltammograms in PBS solutions of pH = 7.0 of capsaicin at different concentrations given in the graph in mol L–1. Bottom: Calibration made at 0.325 V.
From the plot shown in this figure, the following linear relation was obtained
From the analysis, of the data, it follows that LOD = 0.198 µM, LOQ = 0.660 µM, and dynamic linear range (DLR) was from 0.660 to 20.9 µM.
The next step was to evaluate the pungency of the aqueous and ethanolic extracts of Capsicum annuum cubana red prepared as described in the experimental section, recording the differential pulse voltammograms, and using the calibration shown in Figure 3 to obtain the capsaicin + dihydrocapsaicin concentration.
In Figure 4 are shown the voltammograms corresponding to the extracts of cubana red, together that corresponding to ascorbic acid and capsaicin.

Differential pulse voltammograms of 2-mL cubana red (CR) and sweet pepper (SP) extracts (in water, w, and in ethanol, e) in 48 mL of PBS solution, 4·10−5 M ascorbic acid (AA), and 2.24·10−5 M capsaicin (CP). pH = 7.0.
The voltammograms of the extracts showed a peak corresponding to ascorbic acid at potentials close to 0.1 V and (in the case of the ethanolic extract of cubana red) the peak corresponding to capsaicin, at potentials close to 0.3 V. Therefore, to obtain the pungency, it is necessary to separate the two contributions. This could be accomplished by deconvoluting the signals, but this would imply a decrease in the simplicity of the method. An alternative was chosen to obtain the ascorbic acid content and pungency, using a variant of the standard addition method. Due to the difficulty of preparing capsaicin solutions (and taking into account the high cost of this chemical), different volumes of a solution of 0.01 M ascorbic acid were successively added to the extract and the corresponding differential pulse voltammograms were recorded. For clarity, Figure 5 shows a set of voltammograms selected from the 10 experiments made, included the one corresponding to the sample without addition of ascorbic acid. This figure clearly shows that the addition of ascorbic acid increases the intensity of the voltammograms.

Top: Differential pulse voltammograms of 2-mL cubana red ethanolic extract in 48 mL of PBS solution at pH = 7.0 and different volumes of 0.01 M ascorbic acid. Bottom: Plot of the net currents at the potentials given in the graph versus ascorbic acid volume added.
The intensities measured at the potentials corresponding to the peaks of ascorbic acid and capsaicin, after subtracting the blank intensity at the same potentials, are also given in Figure 5. The points corresponding to ascorbic acid are linearly distributed and the extrapolation to zero intensity gives the concentration (through the extrapolated volume) of this compound, as expected for a standard addition method. In this case, the ascorbic acid concentration is 907 ppm.
From the plot shown in this figure, the following linear relation was obtained for ascorbic acid
From the analysis, of the data, it follows that LOD = 17.56 µL, LOQ = 58.55 µL, and DLR was from 58.6 to 500 µL.
The same method was used for the water extract, obtaining 1430 ppm (no signal of capsaicin was observed). In the case of sweet pepper, the ascorbic acid content obtained by the standard addition method was 1180 and 960 ppm for the extracts in water and in ethanol, respectively.
Once the intensity of the ascorbic acid signal has been extrapolated, the intersection of the line corresponding to the capsaicin with the abscissa obtained from the extrapolation to zero of the ascorbic acid line gives the expected intensity of the capsaicin voltammogram in the absence of ascorbic acid.
For capsaicin, the regression equation for the data shown in Figure 5 is
In this case, the independent variable is the volume of ascorbic acid added, which is not related to the capsaicin concentration. The value related to this variable is the signal corresponding to the sample in the absence of ascorbic acid, at the value of ascorbic acid obtained at Y = 0, which will be called Xa. In this case, the correct statistical variable must be the standard deviation of the intercept of the plot of the Y values versus the X − Xa values. When this representation was made, the standard deviation for the intercept found was ±2.8%. This value is accurate enough since pungency measurements are generally affected by higher errors.
From this value, and using the calibration given in Figure 3, a capsaicin concentration of 5.95·10−6 M was obtained, which corresponds to 227 ppm or 3635 SHU in the Scoville scale.
To evaluate the method, the contents of capsaicin and ascorbic acid in the extracts were determined following the spectrophotometric method described in the literature. 9 For the ethanolic extract of cubana red, 903 ppm of ascorbic acid and 228.5 ppm of capsaicin (3658 SHU) were obtained, very close to the values measured with the electrochemical methodology. In the rest of the cases, the differences between the values obtained were ±0.7%. Although the spectrophotometric determination of ascorbic acid was relatively rapid, the analysis of capsaicin involved evaporation of extract to dryness in a hot water bath, dissolution of the residue in a basic solution of phosphomolybdic acid, shaking for 1 h, and a final step of filtration and centrifugation for 15 min. In contrast, simultaneous electrochemical determination of both compounds was performed in 30 min in total.
Finally, the lowest content in capsaicin that can be determined by this technique is c.a. 5 ppm (80 SHU), this being a value comparable to the spectrophotometric method 9 and other methods mentioned in “Introduction.”
Conclusion
The use of DPV with a glassy carbon electrode allows the simultaneous determination of the pungency and ascorbic acid content of extracts of C. annuum cubana red, and by extension of other chili peppers. The method is cheap, simple, and fast, its sensitivity being comparable to other more laborious methods.
Materials and methods
Materials
General
Capsaicin 97% was from Sigma. All other chemicals used, including ascorbic acid, were Merck analytical grade reagents. pH measurements were made with a Metrohm 780 pH meter, calibrated daily with the corresponding pH standards (Sigma-Aldrich).
Capsicum cubana red was obtained from seed cultivation during the spring–summer of 2019. The sweet red pepper was purchased from a local market (Figure 6).

Capsicum annuum cubana red.
Electrochemical measurements
A CHI650A electrochemical workstation from IJ Cambria was used. The reference electrode was a Metrohm 6.0733.100 Ag | AgCl |KCl (3 m), and the auxiliary electrode was a platinum rod.
A glassy carbon working electrode (IJ Cambria) with 7.5 mm2 area was used.
Methods
Extracts preparation
The seeds were manually separated from the pulp. The pulp was minced manually and then the samples were ground for 3 min in a mortar.
The extracts were produced by suspending 2 g of powdered pulp in 10 mL of ultrapure water preheated at 90 °C (or pure ethanol at 65 °C) and stirring with a vortex for 5 min at 300 r min−1 at room temperature. After centrifuging during 3 min at 3200 r min−1, the supernatant liquid was filtered through a 0.45-µm nylon syringe filter.
Electrochemical measurements
A 0.1 M phosphate buffer solution (PBS) at pH 7 was used as supporting electrolyte. The aqueous solutions were prepared using ultrapure water type I (resistivity 18.2 MΩ cm at 298 K) obtained from an ultrapure water Millipore system. The ionic strength was 0.3 M adjusted with solid NaCl. The pH adjustment was made with solid NaOH.
Solutions were purged with purified N2 for at least 10 min to avoid the presence of oxygen that could originate undesired redox reactions on the electrode.
The electrode was electrochemically pretreated with five voltammetric cycles made between +2 and −2 V in a saturated sodium chloride solution. Then, it was polished using first a silicon carbide paper, followed by diamond (0.25 µm) slurry and two alumina (0.3 and 0.05 µm) slurries. After each polishing, residual material was removed by sonication of the electrode in an ultrapure water bath for 30 min.
The parameters selected in the DPV were pulse amplitude 0.05 V, pulse width 0.05 s, and pulse period 0.2 s.
All potentials were measured in the Ag/AgCl(3M) scale.
Spectroscopic measurements
The contents of capsaicin and ascorbic acid in the extracts were determined by using a Perkin Elmer Lambda 750S and Hellma quartz cuvettes of 1-cm pathlength and following the methods described by Orobiyi et al. 9
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Operative Framework Funding FEDER-Andalucía 2014-2020 Program (UCO-1263679) and PAIDI group FQM 198 (Junta de Andalucía, Spain).