Abstract
The first concise synthesis of natural flavonoid glycosides, oroxins C (
The first concise synthesis of natural flavonoid glycosides, oroxins C (

Introduction
Flavonoid glycosides, glycosylated secondary metabolites widely distributed in the plant kingdom (such as vegetables, fruits, and medicinal plants),1–3 having tremendous structural diversity, exhibit a multitude of biological and physiological activities, such as antimicrobial, 4 antitumor, 5 anti-inflammatory, 6 antidiabetic, 7 and hepatoprotectant activities. 8 Compared with flavonoids, flavonoid glycosides possess similar stability, bioactivity, and better solubility and are often more efficacious than their aglycones in pharmaceutical studies. 9 Although flavonoid glycosides are widely distributed and have been demonstrated to possess important biological activities, access to this class of natural products by separation and purification, especially in desirable amounts, presents a formidable task. This results in a great obstacle for further pharmacological studies of homogeneous flavonoid glycosides. Chemical synthesis could provide a feasible way to solve the problem, and many groups have made great efforts to provide sufficient amounts of flavonoid glycosides through chemical synthesis.10–12
Recently, Wu et al.
13
reported two novel flavonoid glycosides, oroxin C (baicalein 7-O-β-
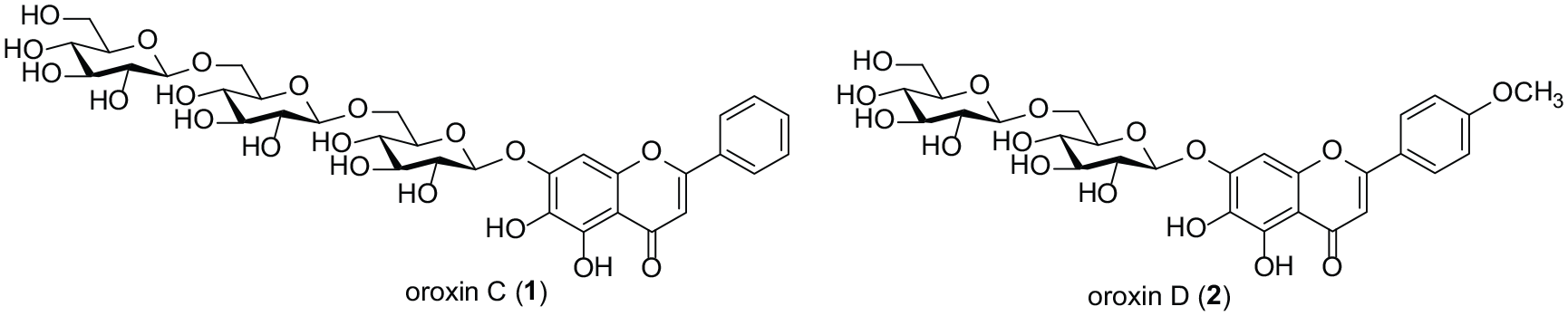
Chemical structure of flavonoid glycosides, oroxins C (
Results and discussion
In terms of their chemical structures, the glycoside residues of the target compounds

Retrosynthesis of target flavonoid glycosides, oroxins C (
As shown in Scheme 2, disaccharide bromide

Reagents and conditions: (a) NH2NH2·HOAc, DMF, 81% for
The partially protected baicalein and scutellarein acceptors

Reagents and conditions: (a) Ac2O, AcONa, 80 °C, 90% for
With an effective synthetic access to the bromide donors (
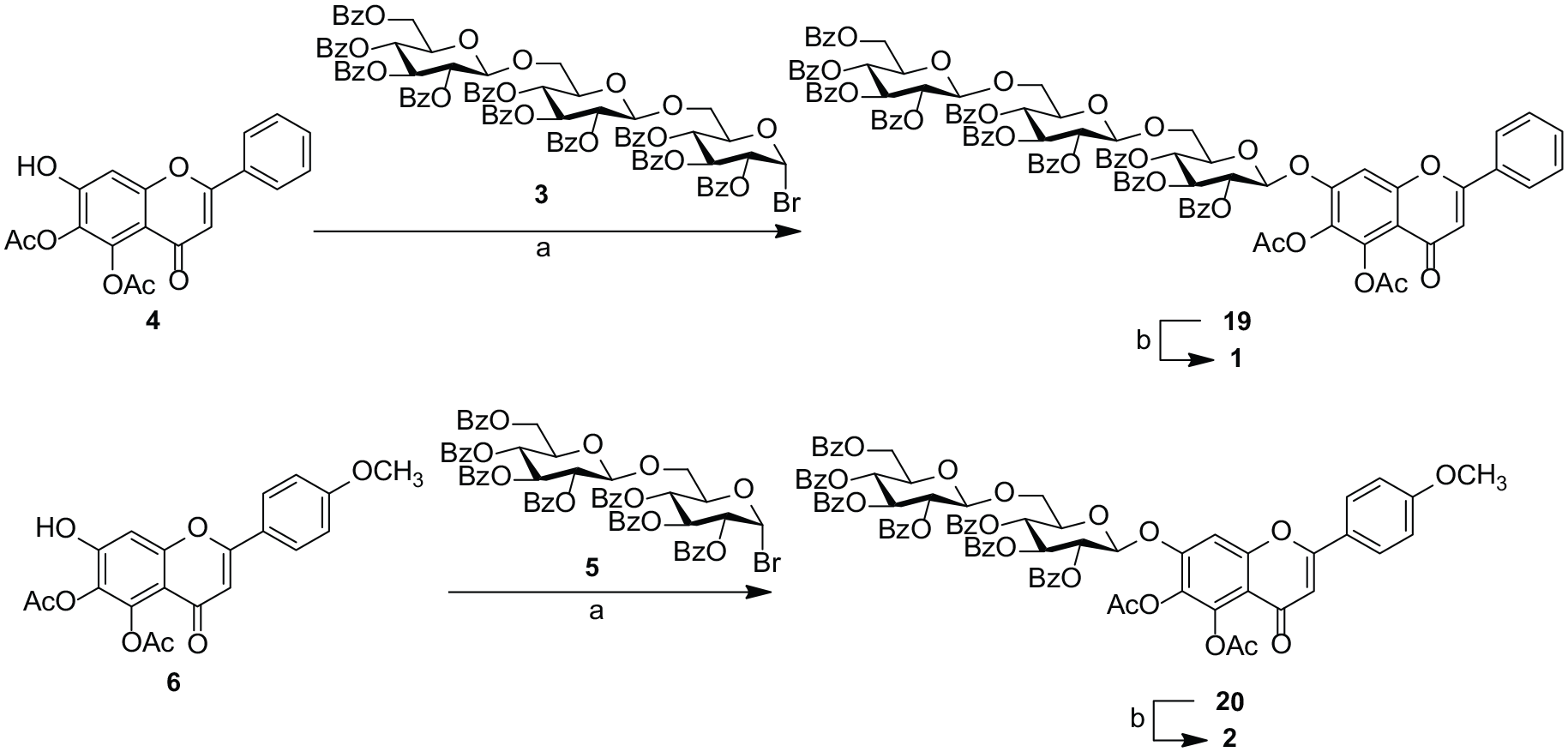
Reagents and conditions: (a) Ag2O, quinoline, 79% for
To examine the potential antidiabetic ability of the natural flavonoid glycosides, oroxins C (
Inhibitory activities of

NA: not active.
The IC50 values in μM were calculated from the dose response curve of six concentrations of each test compound in triplicate.
“-”: not determined.
Conclusion
In conclusion, we have succeeded in the first concise synthesis of the natural flavonoid glycosides, oroxins C (
Experimental
Chemistry
Commercial reagents were used without further purification unless specialized. Solvents were dried and redistilled prior to use in the usual way. Thin-layer chromatography (TLC) was performed on precoated E. Merck Silica Gel 60 F254 plates. Flash column chromatography was performed on silica gel (200–300 mesh). Optical rotations were determined with a Perkin–Elmer Model 241 MC polarimeter. 1H NMR and 13C NMR spectra were taken on a JEOL JNM-ECP 600 spectrometer with tetramethylsilane as the internal standard, and chemical shifts are recorded in δ values. Mass spectra were recorded on a Q-TOF Global mass spectrometer.
2,3,4,6-Tetra-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-α-d -glucopyranosyl bromide (5 )
A solution of
2,3,4,6-Tetra-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-β-d -glucopyranosyl -(1→6)-2,3,4-tri-O-benzoyl-α-d -glucopyranoside (12 )
A solution of
2,3,4,6-Tetra-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-β-d -glucopyranosyl -(1→6)-2,3,4-tri-O-benzoyl-α-d -glucopyranosyl bromide (3 )
A solution of
5,6,7-Tri-O-acetyl-4’-O-methyl scutellarein (18 )
A solution of compound
5,6-Di-O-acetyl-4’-O-methyl scutellarein (6 )
A solution of compound
5,6-Di-O-acetyl-7-O-[2,3,4,6-tetra-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-α-d -glucopyranosyl] baicalein (19 )
To a solution of
5,6-Di-O-acetyl-7-O-[2,3,4,6-tetra-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-β-d -glucopyranosyl-(1→6)-2,3,4-tri-O-benzoyl-α-d -glucopyranosyl]-4’-O-methyl scutellarein (20 )
Similar procedure as that used for the synthesis of
Baicalein 7-O-β-d -glucopyranosyl-(1→6)-β-d -glucopyranosyl-(1→6)-β-d -glucopyranoside (1 )
To a solution of
Scutellarein 4’-methyl ether 7-O-β-d -glucopyranosyl-(1→6)-β-d -glucopyranoside (2 )
Similar procedure as that used for the synthesis of
Biological activities assay
Assay for α-glucosidase inhibitory activities
Inhibitory α-glucosidase activities were determined spectrophotometrically in a 96-well microtiter plate based on p-nitrophenyl-α-
Assay for α-amylase inhibitory activities
The α-amylase inhibitory activities were measured with the method reported by Xiao et al. 21 and Yoshikawa et al. 22 with slight modifications. Substrate was prepared by heating starch (250 mg) in 12 mL of 0.4 M NaOH solution for 5 min at 100 °C, and then cooled to 0 °C and adjusted to pH 7 with 2 M HCl. Sample solutions were prepared by dissolving each solution in acetate buffer (pH 6.5). The sample (20 μL) and the substrate (40 μL) were mixed in a microplate well. After preincubation at 37 °C for 15 min, 5 mg/mL α-amylase solution (20 mL) was added and the solution was incubated at 37 °C for 15 min. The reaction was stopped by adding 50 mL 1 M HCl, and then 50 mL iodine solution was added. The absorbances were measured at 650 nm by a microplate reader. Acarbose was used as positive control.
Assay for lipase inhibitory activities
Lipase inhibitory activities were measured according to the method of Han et al. 23 with slight modifications. Substrate was prepared by sonication of a mixture of glyceryl trioleate (80 mg), lecithin (10 mg), and sodium cholate (5 mg) suspended in 9 mL of 0.1 M TES buffer (pH 7.0). Samples were prepared by dissolving each sample in 0.1 M TES buffer. The sample (20 μL) and the substrate (20 μL) were mixed in microplate wells. After preincubated for 5 min, 10 μL of lipase solution (20 μg/mL) was added to each reaction mixture and incubated for 30 min at 37 °C. The amount of released fatty acid was measured at 405 nm. Inhibition of lipase activity was expressed as the percentage decrease in the absorbance when porcine pancreatic lipase was incubated with the test compounds. Orlistat was used as positive control.
Supplemental Material
Supporting_information – Supplemental material for Concise synthesis and antidiabetic activity of natural flavonoid glycosides, oroxins C and D, isolated from the seeds of Oroxylum indium
Supplemental material, Supporting_information for Concise synthesis and antidiabetic activity of natural flavonoid glycosides, oroxins C and D, isolated from the seeds of Oroxylum indium by Gang Li, Guanghui Wang, Yangliu Tong, Junheng Zhu, Tongtong Yun, Xiaoping Ye, Fahui Li, Shengli Yuan and Qingchao Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financial supported by the Foundation of Facility Horticulture Laboratory Project of Shandong Universities (2018YY011, 2018YY042).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
