Abstract
A new complex, [Co(DFB)2(3-Pyr)2(H2O)2] (where DFB = 3,5-difluorobenzoate, 3-Pyr = 3-pyridinol), is synthesized and characterized using different techniques (elemental analysis, Fourier transform infrared spectroscopy, and single-crystal X-ray diffraction). Looking at the crystal structure of the complexes, the cobalt atom is coordinated by two nitrogen atoms from two 3-Pyr ligands, two carboxylate oxygen atoms from two DFB anions, and two oxygen atoms from two water molecules. The complex has distorted octahedral geometry around the cobalt atom center complex and crystallizes in the P21/n space group (monoclinic system). Geometry optimization, frequency analysis, and energy quantum chemical calculations on the complex are performed by Density Functional Theory [B3LYP/6-31G (d,p) basis set] to predict the molecular properties. The novel complex is tested against the metabolic isoenzymes human carbonic anhydrases I and II. The novel complex shows Ki values of 317.26 ± 23.25 µM against hCA I and 255.41 ± 48.05 µM against hCA II; the IC50 values for these isoenzymes are 274.37 and 204.33 µM.
Keywords

Introduction
Coordination compounds are used in many fields. Different metal–ligand designs play an important role in the development of crystal engineering1,2 and provide materials that can be used in many areas by formation of targeted complexes. Although all metals are toxic at high concentrations, it is known that many metal ions play very important roles in biological systems.3,4 Also, cobalt binds to vitamin B12 and plays an important role in the biochemical systems.5,6 In addition, the antioxidant and antibacterial properties of cobalt nanoparticles and cobalt complexes are known.7–12 Studies on the roles of metals and metal complexes in biological systems have gained significant importance in recent years. In the literature, there are many studies on the antioxidant, 13 antibacterial,14,15 anticancer, 16 antitumor, 13 and enzyme inhibitory 17 properties of transition-metal complexes of carboxylic acids.
Human carbonic anhydrases (hCAs) are common in organisms and vary in their tissue distribution and intracellular localization. Sixteen α-isozymes (hCA I-XV) have been isolated and identified so far, but only 12 are catalytically active. hCAs are involved in pH and CO2 homeostasis, as well as in regulating many critical physiological processes such as gluconeogenesis, lipogenesis, or electrolyte secretion in various tissues and organs. 18 The isoenzymes hCA I and II are the most widely expressed isoforms and are identified in the anterior chamber of the eye, while hCA IV is responsible for the production of bicarbonate, the main component of aqueous humor in this organ. 18 hCA I and II isoenzymes inhibition has pharmacological applications in various fields: anticancer agents (coumarins, sulfonamides, dithiocarbamates, etc.) and various classes of pharmacological agents such as antiglaucoma, antiepileptic, diuretic, and antiobesity. Carbonic anhydrase inhibitors (CAIs) are mainly used for the treatment of glaucoma or other ocular conditions where lowering of the intraocular blood pressure has been deemed beneficial. Acetazolamide is also used for the treatment and prevention of acute mountain sickness (also known as altitude sickness) and in some types of epilepsy.19–22
In this study, a new complex of Co(II) 3,5-difluorobenzoate (DFB) with 3-pyridinol, [Co(C7H3F2O2)2(C5H5NO)2(H2O)2], was synthesized and its structure was characterized by elemental analysis, Fourier-transform infrared (FT-IR) spectroscopy, and single-crystal X-ray diffraction methods. In addition, the antibacterial properties of the complex were investigated by the agar-well diffusion method against Pseudomonas aeruginosa, Klebsiella pneumoniae, Escherichia coli, and Staphylococcus aureus. Geometry optimization and frequency analysis of the difluorobenzoate complex was performed using the DFT/B3LYP/6-31G(d,p) method. The TD-DFT/B3LYP/6-31G(d,p) method was used to obtain the molecular orbital energies of the complex.
Results and discussions
X-ray structural determination
The experimental details are given in Table 1. The asymmetric unit of the centrosymmetric title compound contains one half of the complex molecule (Figure 1). The metal atom is located on a center of symmetry (Figure 2). The molecule contains two DFB and two 3-Pyr ligands and two coordinated water molecules, all ligands being monodentate. The four O atoms (O1, O4, and the symmetry-related atoms, O1′, O4′) in the equatorial plane around the Co atom form a slightly distorted square-planar arrangement, while the slightly distorted octahedral coordination is completed by the two N atoms (N1, N1′) of the 3-Pyr ligands in the axial positions (Figure 2). The near equality of the C1–O1 [1.250 (4) Å] and C1–O2 [1.256 (4) Å] bonds in the carboxylate group indicates a delocalized bonding arrangement, rather than localized single and double bonds, and may be compared with the corresponding distances: 1.256 (6) and 1.245 (6) Å in [Mn(diethylnicotinamide)2(4-chlorobenzoato)2(H2O)2], (I), 23 1.265 (6) and 1.275 (6) Å in [Mn(dimethylamino)2(H2O)4].2(H2O) (II), 24 1.260 (4) and 1.252 (4) Å in [Zn(diethylnicotinamide)2(4-fluorobenzoato)2(H2O)2] (III), 25 1.259 (9) and 1.273 (9) Å in Cu2(diethylnicotinamide)2(benzoato)4 (IV), 26 1.279 (4) and 1.246 (4) Å in [Zn2(diethylnicotinamide)2(4-hydroxybenzoato]·2H2O (V), 27 1.251 (6) and 1.254 (7) Å in [Co(diethylnicotinamide)2(2-hydroxybenzoato)2(H2O)2] (VI), 28 and 1.278 (3) and 1.246 (3) Å in [Cu(diethylnicotinamide)2(4-nitrobenzoato)2(H2O)2] (VII). 29 The average Co–O bond length (Table 2) is 2.099 (2) Å, and the Co atom is displaced out of the least-squares plane of the carboxylate group (O1/C1/O2) by −0.2616(1) Å. Atoms F1 and C1 are 0.039 (6) and −0.017 (3) Å, while atoms Co1 and O3 are −0.0891 (1) and −0.003(3) Å away from the adjacent A and B rings, respectively. Hence, they are nearly co-planar with the corresponding rings. The dihedral angle between the planar carboxylate group (O1/O2/C1) and the adjacent benzene ring A (C2–C7) is 15.9 (2)°, while that between benzene ring A and pyridine ring B (N1/C8–C12) is A/B = 74.4 (1)°. In the crystal structure, the O–H···O hydrogen bonds (Table 3) link the molecules into a network (Figure 3), and they are further linked by the C–H···O and C–H···F hydrogen bonds (Table 3) into a three-dimensional structure in which they may be effective in the stabilization of the structure. The π–π contacts between the benzene rings, Cg1–Cg1i [symmetry codes: (i) 2 − x, 1 − y, and 1 − z, where Cg1 is the centroid of the ring A (C2–C7)] may further stabilize the structure, with a centroid–centroid distance of 3.804 (2) Å.
Experimental details.

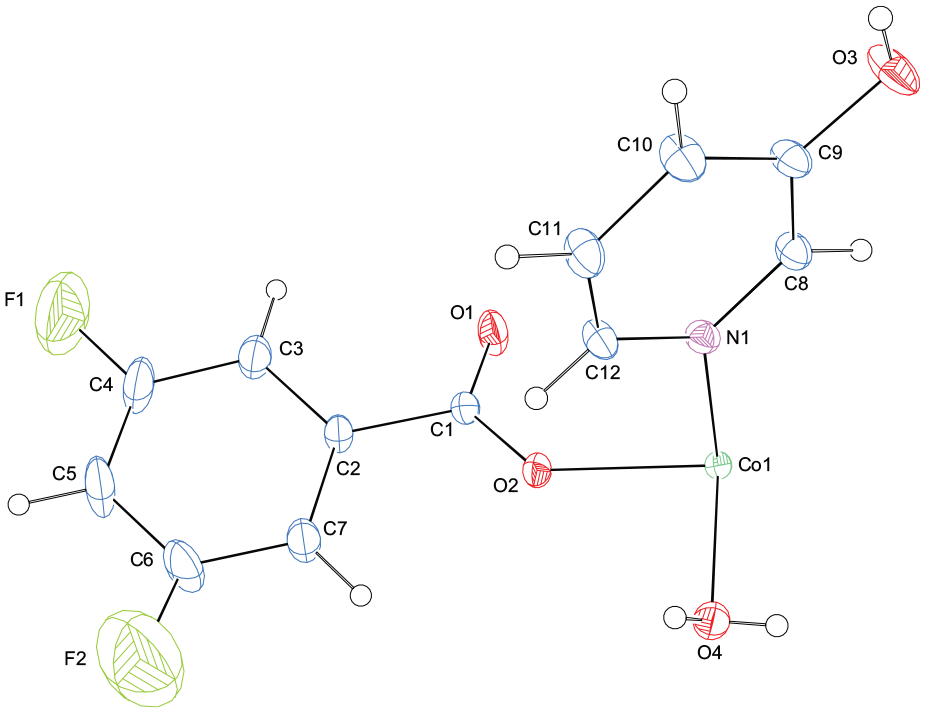
The asymmetric unit of the title compound with the atom numbering scheme. Thermal ellipsoids are drawn at the 50% probability level.

The molecular structure of the title compound with the atom numbering scheme. Thermal ellipsoids are drawn at the 50% probability level.
Selected geometric parameters (Å, °).

Symmetry code: (i) –x + 1, −y + 1, −z + 2.

A partial packing diagram of the complex. Only the O–H···O hydrogen bonds are shown as dashed lines. Remaining hydrogen atoms have been omitted for clarity.
Hydrogen-bond geometry (Å, °).

Symmetry codes: (i) –x + 1, −y + 1, −z + 2; (ii) –x + 3/2, y + 1/2, −z + 3/2; (iii) x−1, y, z; (iv) –x − 1, −y + 1, −z + 1.
Calculated thermochemical values of the complex with B3LYP/6-31G(d, p) level of theory in gas phase.

Etot is the total internal thermal energy, Ctot is the total constant volume heat capacity and Stot is the total entropy. Gcorr is the correction for the Gibbs free energy. Hcorr is the correction for the enthalpy.
Infrared spectra
In the IR spectra of the complex, the ν(O–H) vibration of the coordinated water molecules is seen at 3504 cm−1. When we examine the synthesized complex, the carbonyl group COO− asymmetric and symmetrical vibrations are observed at 1541–1390 cm−1, respectively. The vibration recorded at 643 cm−1 supports the presence of the Me–O bond.34,35 However, the absorption band C–N group of 3-pyridinol was observed at 1052 cm−1. 36 (See in the Supplemental material.)
Theoretical results
Geometry optimization, the molecular orbital diagram, and the electron density of the studied complex are given in Figure 4. Vibrational frequency analyses are given in Figure 5 for comparison.

Optimized geometry and the molecular orbital diagram of the complex.

3D and 2D total electron density surfaces of the complex.
The dipole moment (μ) of the complex is 0.0003 Debyes and the polarizability (α) is 327.056 a.u. The electronic energy (ε0), the thermal energy correction (ε0 + Etot), the zero-point energy (ε0 + εZPE), the thermal free-energy correction (ε0 + Gcorr), and the thermal enthalpy correction (ε0 + Hcorr) values are given in Table 4.
The calculated parameters of the complex using B3LYP/6-31G(d,p) basis set at DFT/TD-DFT.

LUMO: lowest unoccupied molecular orbital; HOMO: highest occupied molecular orbital.
Complex interactions, bond lengths, and bond angles were compared with the experimental data. The calculated bond lengths and angles are in agreement with the experimental data. The experimental value for the Co1–O2 distance is 2.049 Å and the calculated value is 1.942 Å. The Co1–N1 distance was 2.146 Å, while the calculated value is 1.962 Å. The Co1–O4 distance was found experimentally to be 2.149 Å and calculated theoretically as 2.274 Å, while the O1–C1 distance is the same experimentally and theoretically as 1.250 Å. The O2–C1 bond distance was reported experimentally as 1.256 Å and calculated theoretically as 1.285 Å. Co1–O2 and O2–C1 bonds are rotatable; however, the Co1–N1, Co1–O4, and O1–C1 bonds are not rotatable.
The experimental angle value of O2–Co1–N1 is 89.48° and the computational value is 89.77°. The experimental and the theoretically calculated values for O2–Co1–O4 are 88.99° and 85.07°, respectively. The experimental and the theoretically calculated values for N1–Co1–O4 are 90.88° and 86.38°, respectively, while O1–C1–O2 is 124.4° experimentally and 126.69° theoretically.
All the molecular orbital energies (highest occupied molecular orbital (HOMO), lowest unoccupied molecular orbital (LUMO), and energy gaps (EGAP = EHOMO − ELUMO)) of the complex were studied in dimethyl sulfoxide (DMSO). The energy gap value (eV) of the complex was calculated as 3.998 eV. This value is 3.763 eV in the gas phase. The energy gap, ionization potential, polarizability, dipole moment, electronegativity, electrophilicity index, electron affinity, and softness and hardness values of the complex are given in Table 5.
The electron density of the synthesized complex was investigated in DMSO. The blue region is electropositive, the reddish region is electronegative, and the green region is neutral. The carboxylate group (O2–C1–O1 and opposite) that interacts with water is electronegative. The hydrogen atom in the hydroxy group attached at the third position of the pyridine ring is electropositive and its oxygen atom is slightly electronegative. The other regions in the molecule are neutral (green).
Vibrational frequency analysis was studied for the complex in the gas phase and the calculated FT-IR spectra were compared with the experimental FT-IR spectra. The peak at about 3800 cm−1 in the calculated spectrum is related to the hydroxy group on the pyridine. The large peak for the complex at 3143 cm−1 shows an interaction of the carboxylate group with water. In general, the calculated and theoretical FT-IR spectra values are in agreement with each other (Figure 6).
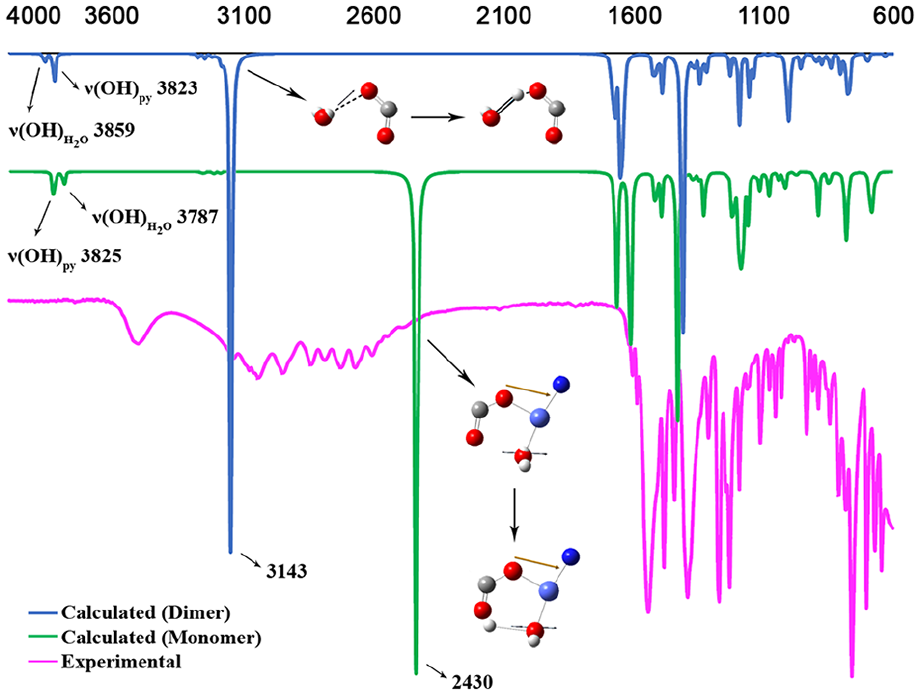
Calculated and experimental vibrational frequency spectra (FT-IR) of the complex.
Biochemical results
Antibacterial potential
In this study, besides the synthesized complex, ampicillin X3261, neomycin X3385, and streptomycin X3385 were used as standard antibiotics. The effects of the complex and standard antibiotics against P. aeruginosa, K. pneumoniae, E. coli, and S. aureus bacteria were investigated and the zone diameters are given in Table 6.
Antibacterial zone diameters of complexes (mm).

Enzyme results
The development of CAIs as therapeutic agents must take into consideration the fact that their effects on non-target isoenzymes must be minimal. CAIs have been developed for the treatment of a variety of diseases, since CA plays a role in several important physiological functions, such as transport of CO2 and bicarbonate, lipogenesis, gluconeogenesis, calcification, bone resorption, and tumorigenicity, to name but a few.37,38 The CA isoenzymes involved in these physiological processes are, therefore, potential therapeutic targets for treating various disorders such as cancer, epilepsy, glaucoma, acute mountain sickness, pain and osteoporosis by inhibition of target CA isozymes. The use of CAIs for the treatment of glaucoma has been well established for decades, as such drugs lower intraocular pressure.39,40 CAI molecules have recently been demonstrated to have ability as anticancer drugs.39,41 For the hCA I isoform, the Ki value was determined to be 317.26 ± 23.25 µM. In comparison, the Ki for the standard CA inhibitor acetazolamide (AZA), a definitive hCA I inhibitor, was 433.22 ± 55.30 µmol/L against hCA I (Table 7). Hence, novel complex have effective inhibition effects than that of AZA. As shown in Table 7, the IC50 value is 274.37 µM toward hCA I and for hCA II is 204.33 µM. The complex significantly inhibited hCA II with Ki in the low micromolar range. Ki value was calculated between 255.41 ± 48.05 µM (Table 7). In comparison, the Ki for the standard CA inhibitor AZA, a definitive hCA II inhibitor, was 384.14 ± 84.90 µmol/L against hCA II.
The enzyme inhibition results of novel complex against human carbonic anhydrase isoenzymes I and II (hCA I and II).

Acetazolamide (AZA) was used as a control for hCA I and II.
Conclusion
In this study, a new complex, [Co(DFB)2(3-Pyr)2(H2O)2], has been synthesized. The CoII atom is coordinated in a monodentate fashion with the oxygen atoms of the DFB anions, the nitrogen atoms of the 3-Pyr ligands, and the oxygen atoms of coordinated water molecules. The ratio of cobalt/DFB/3-Pyr ligands was determined as 1:2:2. The N2–O4 bonding sets around the metal center formed a distorted octahedral geometry. Geometry optimization, vibrational frequency analyses, and the molecular energy values (molecular orbitals) of the complex were studied using the B3LYP/6-31G(d,p) basis set in DFT/TD-DFT calculations. The experimental and theoretical values were compared with each other. The HOMO and LUMO energy values are negative, indicating that the structure is stable. The theoretical results obtained in this study are useful to obtain new complex derivatives as antimicrobial agents. The antimicrobial activity of the complex was investigated against P. aeruginosa (ATCC 27853), K. pneumoniae (ATCC 4352), E. coli (ATCC 25922), and Gram-positive S. aureus (ATCC) 6538. In adittion, the complex had similar effects to those of commercially available neomycin X3385 and streptomycin X3385, but was less effective than ampicillin X3261. This novel complex would further studied for potential benefit from therapy of diseases such as epilepsy, gastric and duodenal ulcers, mountain sickness, glaucoma, osteoporosis, and neurological disorders.
Experimental
3,5-Difluorobenzoic acid (Alfa Aesar™), 3-pyridinol (Merck), sodium bicarbonate (Merck), and cobalt(II) sulfate heptahydrate (Merck) were used without any further purification. The C, H, and N percentages were measured using a LECO CHNS-932 elemental analyzer. FT-IR spectra were recorded on a Perkin Elmer Frontier™ FT-IR spectrometer from solid samples using a Diamond ATR accessory in the range of 4000–600 cm−1. The crystal structure of the complex was determined on a Bruker SMART BREEZE CCD diffractometer.
Synthesis of the complex
To obtain sodium DFB, 3,5-difluorobenzoic acid (1.58 g, 10 mmol) and sodium bicarbonate (0.84 g, 10 mmol) were stirred at 60 °C in 100 mL of distilled water until complete removal of CO2 gas. Next, CoSO4.7H2O (1.42 g, 5 mmol) and 3-pyridinol (1.22 g, 10 mmol) were dissolved in water (50 mL), and the obtained solution was added to the cobalt sulfate solution. The resulting mixture was allowed to crystallize. After 6–7 days, pink single crystals were obtained; crystals were filtered and washed with distilled water and then dried at room temperature. Anal. Calcd (%) for C24H20CoF4N2O8 (molecular weight (MW) = 599.35): C, 48.09; H, 3.36; N, 4.67. Found (%): C, 48.42; H, 3.55; N, 4.72; Selected IR bands (cm−1): ν(OH)H2O 3504, ν(C–N)py. 1052, ν(COO−)as 1541, ν(COO−)s 1390, (Δν 151), ν(Co–O) 643.
Computational details
The Gaussian 09 program 42 was used for theoretical calculations and GaussView 6.0 43 and Avogadro 44 were used to visualize the calculated values. The 6-31G(d,p) basis set of then B3LYP (Becke-3-Lee-Yang-Parr)45,46 functional correlation in Density Functional Theory (DFT) was used for geometry optimization and frequency analyses of the complex. In addition to the calculations in the gas phase, all stages were repeated in the DMSO as the solvent in order to investigate the solvent effects. TD-DFT (Time Dependent–Density Functional Theory) calculations were also calculated using the B3LYP/6-31G(d,p) basis set. The excited state properties were calculated as 50 single excited states via TD-DFT. Electron-density surfaces were displayed according to the self-consistent field (SCF) density matrix. All calculations were compared with experimental data.
Antibacterial properties
The antibacterial properties of the synthesized complex were investigated against Gram-positive S. aureus (ATCC) 6538), Gram-negative E. coli (ATCC 25922), P. aeruginosa (ATCC 27853), and K. pneumoniae (ATCC 4352). Microorganisms obtained from microbiological environmental protection laboratories were cultivated in the research laboratories of Kafkas University Faculty of Engineering and Architecture and the obtained bacteria were used in experiments. The antimicrobial effects of the resulting molecules were investigated as biological applications in Mueller–Hinton agar (MHA) medium.
First, the Mueller–Hinton broth (MHB) for activation of the bacterial stock was carried out for 24 h with incubation at 37 °C. Bacteria which were standardized with 0.5 McFarland standard were seeded in sterile prepared petri dishes; 0.05 g of the synthesized complex was dissolved in 5 mL of DMSO and homogeneous solutions were prepared. Samples (50 μL) from the stock solutions were transferred into wells drilled 4 mm in diameter using an automated pipette. The inhibition zone was incubated at 37 °C ± 1 °C for 18–24 ± 2 h to determine the diameters.47–50 All inhibition zones were measured in millimeter.
hCA Isoenzyme purification and inhibition studies
The CA inhibitory effects of the novel Co(II) complex were measured according to Verpoorte et al. 51 described in previous studies52,53 and data were measured at 348 nm spectrophotometrically using p-nitrophenylacetate (PNA) as the substrate. 54
Supplemental Material
checkcif_O3 – Supplemental material for Synthesis, spectroscopic properties, crystal structures, DFT studies, and the antibacterial and enzyme inhibitory properties of a complex of Co(II) 3,5-difluorobenzoate with 3-pyridinol
Supplemental material, checkcif_O3 for Synthesis, spectroscopic properties, crystal structures, DFT studies, and the antibacterial and enzyme inhibitory properties of a complex of Co(II) 3,5-difluorobenzoate with 3-pyridinol by Mustafa Sertçelik in Journal of Chemical Research
Supplemental Material
complex – Supplemental material for Synthesis, spectroscopic properties, crystal structures, DFT studies, and the antibacterial and enzyme inhibitory properties of a complex of Co(II) 3,5-difluorobenzoate with 3-pyridinol
Supplemental material, complex for Synthesis, spectroscopic properties, crystal structures, DFT studies, and the antibacterial and enzyme inhibitory properties of a complex of Co(II) 3,5-difluorobenzoate with 3-pyridinol by Mustafa Sertçelik in Journal of Chemical Research
Supplemental Material
Supplementary_Figures_S1 – Supplemental material for Synthesis, spectroscopic properties, crystal structures, DFT studies, and the antibacterial and enzyme inhibitory properties of a complex of Co(II) 3,5-difluorobenzoate with 3-pyridinol
Supplemental material, Supplementary_Figures_S1 for Synthesis, spectroscopic properties, crystal structures, DFT studies, and the antibacterial and enzyme inhibitory properties of a complex of Co(II) 3,5-difluorobenzoate with 3-pyridinol by Mustafa Sertçelik in Journal of Chemical Research
Footnotes
Acknowledgements
I would like to thank Tuncer Hökelek, who provided support for X-ray analysis, Parham Taslimi, who provided support for the enzyme inhibition studies, and Mücahit Özdemir for his support in the DFT calculations.
Author’s Note
Crystallographic data for complex reported in this article have been deposited with the Cambridge Crystallographic Data Center as Supplementary Publication No. CCDC 1972765. Copies of these data can be obtained free of charge on application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK; fax: (+44) 1223 336033, or online via ![]() , or by emailing
, or by emailing
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
