Abstract
A new cadinane-type sesquiterpene named cornifronone is isolated from the body surface of a mason bee (Osmia cornifrons)–derived Streptomyces sp. OC1611-8A. Its structure is identified by high-resolution electrospray ionization mass spectrometry data and nuclear magnetic resonance spectroscopic analysis. The absolute configuration of cornifronone was determined by electronic circular dichroism spectra calculations. Cornifronone inhibits hexokinase activity with a mean IC50 of 124.3 μM.
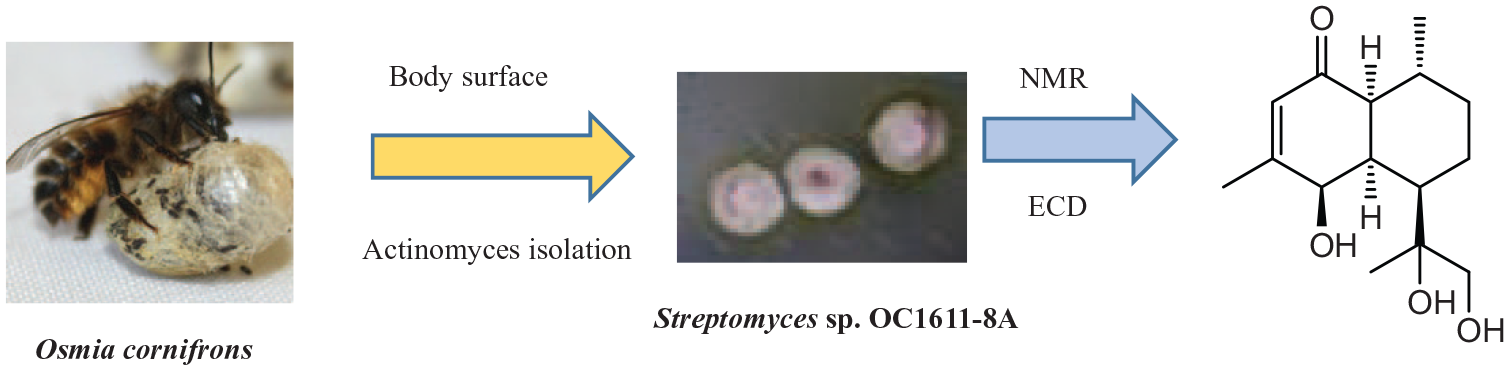
Introduction
Natural products are organic compounds isolated and extracted from natural organisms and are important sources for the development of practical drugs. 1 Microorganisms are the main source of natural products, of which actinomycetes are the largest contributors. 2 These natural products have important effects on human health and life. 3
Studies in recent decades have shown that it is hard to isolate new actinomycetes from soil and to extract new bioactive secondary metabolites. 4 Thus, researchers have turned their studies toward insects. Insects can form special mutually beneficial relationships with their symbiotic microorganisms. Among them, secondary metabolites produced by some insect symbiotic actinomycetes can be used to help host insects protect larvae and cocoons from pathogens;5–10 for example, the European beewolf (Philanthus triangulum) and Streptomyces philanthi; the Streptomyces can be absorbed by the larvae and transferred to the cocoon wall, thus protecting the cocoon from pathogenic fungi and producing a variety of antibiotics to significantly improve the survival rate of the larvae.11,12 Interestingly, the spruce beetle (Dendroctonus rufipennis) also has this symbiotic relationship with the actinomycete Micrococcus luteus. M. luteus has strong inhibitory activity against fungi, protecting beetles from pathogenic fungi, thereby increasing the survival rate of spruce beetles. 13 However, research on insect symbiotic actinomycetes is still in its infancy, and only the functions of a few insect symbiotic actinomycetes and secondary metabolites have been reported. Therefore, further isolation and analysis of symbiotic actinomycetes from insects and identification of related metabolites of actinomycetes will help in the discovery of new natural products with biological activity.
Cornifronone (Figure 1), a new cadinane-type sesquiterpene, was isolated from the body surface of a mason bee (Osmia cornifrons)–derived Streptomyces sp. OC1611-8A. Sesquiterpenoids have a variety of biological activities, such as antitumor, neurotoxicity, antibacterial, and anti-inflammatory, 14 and most are derived from plants. Up to now, only very few sesquiterpenoids have been isolated from actinomycetes, such as 15-hydroxy-T-muurolol, 15 strepsequitriol, 16 and elgonenes.17,18 In the detection of sesquiterpenoids by thin-layer chromatography (TLC) analyses, terpene derivatives can be dyed blue or purple by anisaldehyde/sulfuric acid. However, because unsaturated fatty acid derivatives with similar color reactions masked them, their lipid-like nuclear magnetic resonance (NMR) spectra are usually not obvious; this is the reason for their low isolated yields. 15 Thus, cornifronone enriches the diversity of sesquiterpenoids isolated from actinomycetes.

The structure of cornifronone (
Results and discussion
Structure elucidation
Compound
These facts and four degrees of unsaturation indicate that the compound has a bicyclic ketone structure with a double bond. The planar structure of
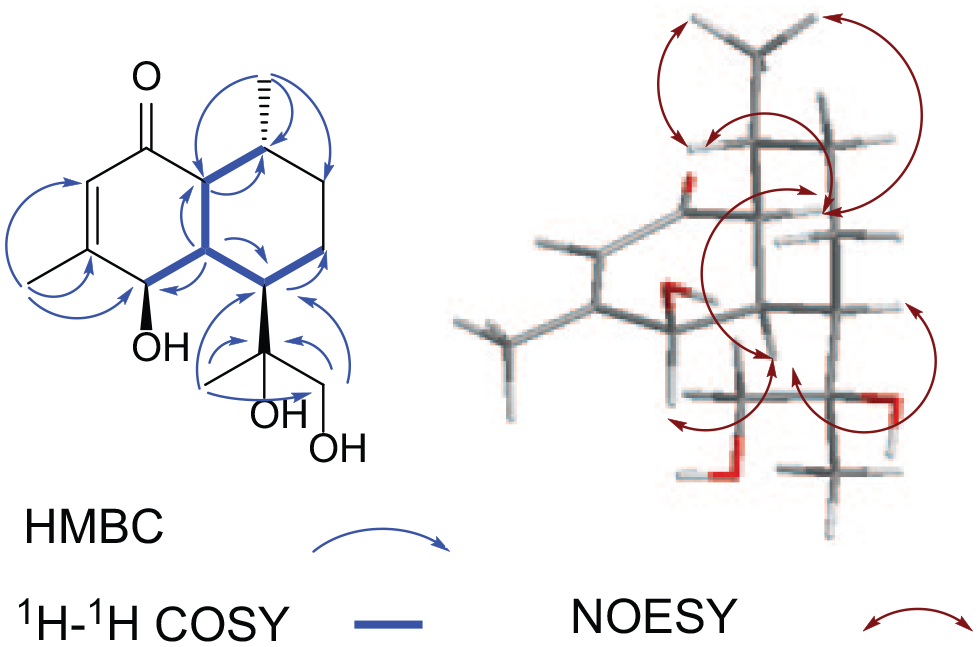
The key HMBC, 1H-1H COSY and NOESY correlations for
The relative configuration of

Experimental and calculated ECD spectra of compound
1H and 13C NMR data for compound
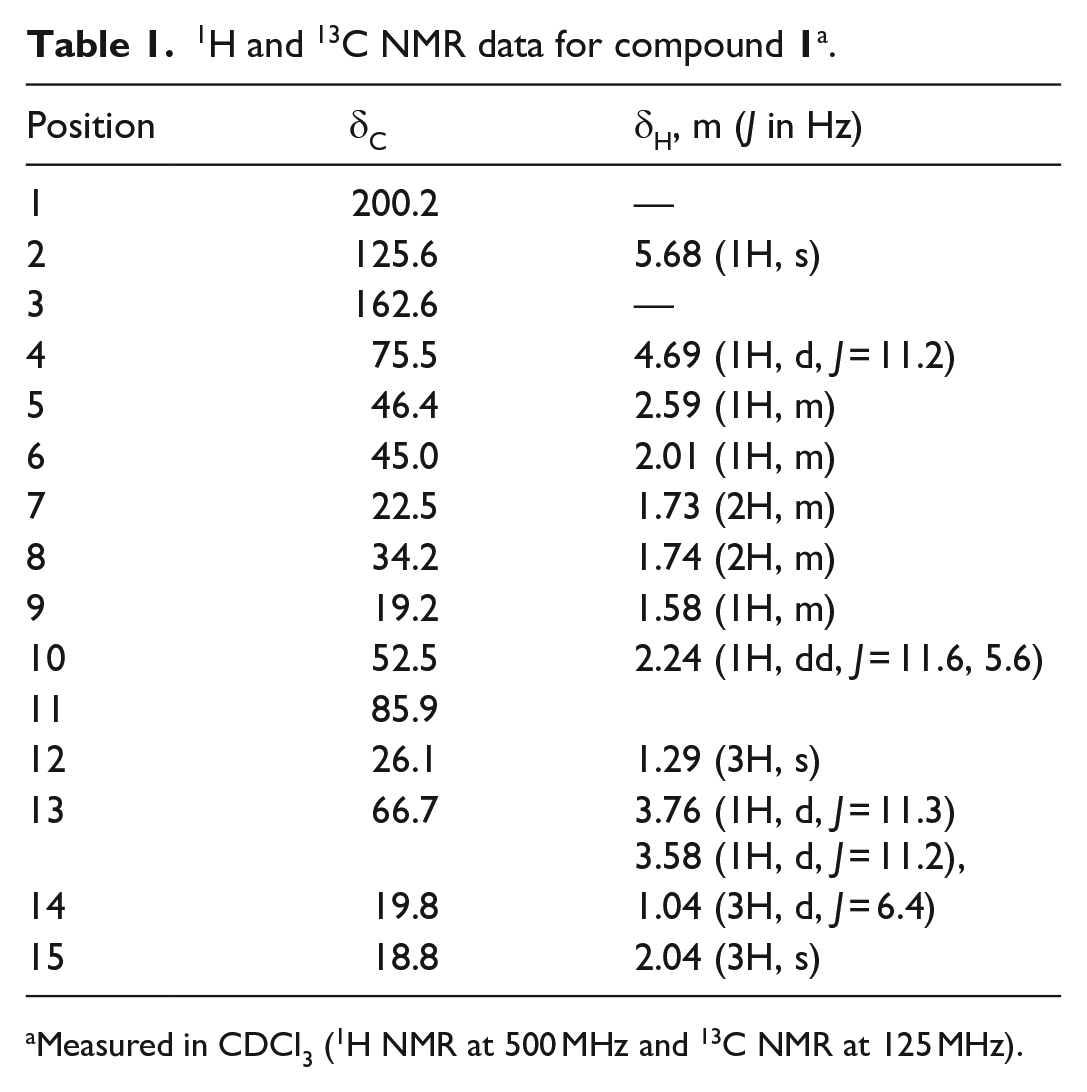
Measured in CDCl3 (1H NMR at 500 MHz and 13C NMR at 125 MHz).
Cytotoxicity and antimicrobial activity
The disk diffusion method was used for testing the antibacterial and antifungal activities of compound
Analysis of the crude extract of the mason bee (O. cornifrons)–derived Streptomyces sp. OC1611-8A led to the isolation and structural characterization of a new cadinane-type sesquiterpene, namely, cornifronone. The compound was characterized based on the NMR, high-resolution mass spectrometry (HRMS), and ECD data. The cornifronone showed weak HK inhibitory activity with a mean IC50 of 124.3 μM.
Materials and methods
Microorganism material
Aiming to discover new antibiotics from insect symbiotic actinomycetes, the strain OC1611-8A was isolated from the body surface of O. cornifrons, which was collected in Weihai, Shandong Province, China. Its 16S ribosomal RNA (rRNA) nucleotide sequence (GenBank number: MK358137) was compared with the data in GenBank, and it was found to be similar to Streptomyces lavendulae (AY745498) and Streptomyces toxytricini (HQ844491), with similarities of 99.27% and 99.26%, respectively. It can be confirmed from the neighbor-joining analysis that the strain OC1611-8A and the members of the genus Streptomyces are closely related in phylogeny and clustered with Streptomyces toxytricini (Figure 4). Subsequently, strain OC1611-8A was fermented and the fermentation broth was extracted with ethyl acetate to obtain a crude extract. A new cadinane-type sesquiterpene was isolated from the crude extract and named cornifronone (
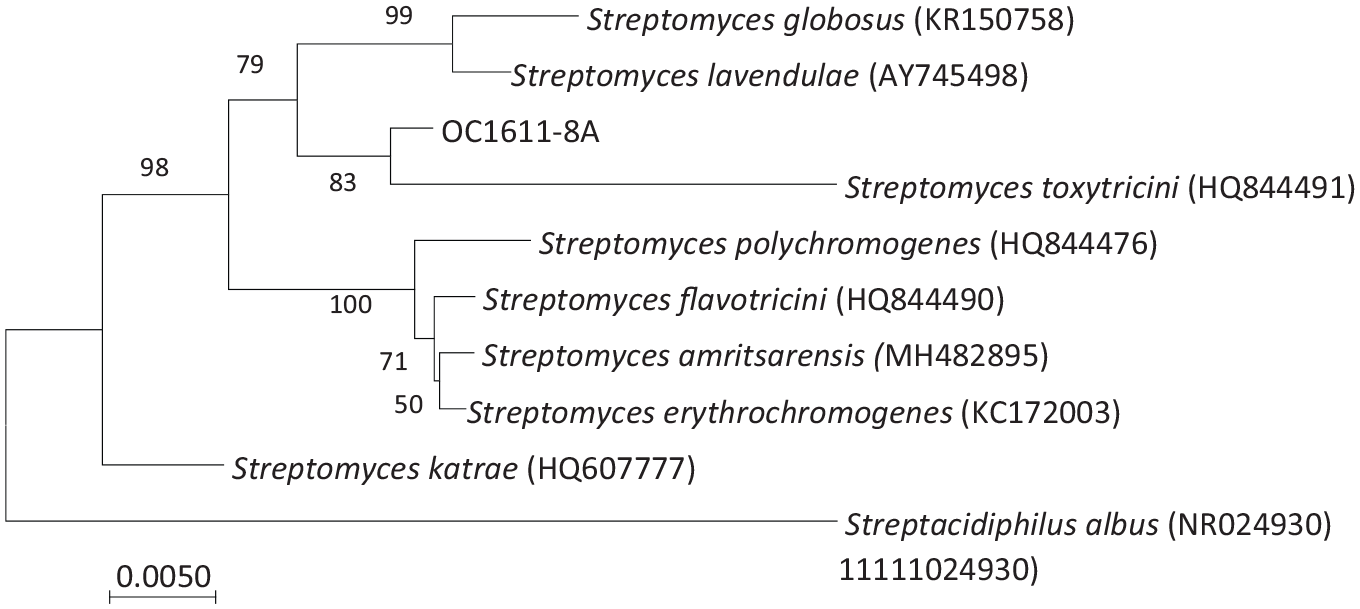
Neighbor-joining tree based on 16S rRNA gene sequences showing the relationships between strains OC1611-8A and related members of the Streptomyces family. Numbers at nodes indicate percentages of 1000 bootstrap re-samplings; only values above 50% are shown. The tree is rooted with Streptacidiphilus albus NBRC 100918T (NR024930). Bar, 0.005 nucleotide substitution per position.
First, the body surface of the mason bee was washed with 2 mL of sterile water to obtain an actinomycete suspension; 200 µL of the suspension was pipetted, transferred to the petri dishes containing Gauze’s medium (20 g starch, 1 g KNO3, 0.5 g NaCl, 0.5 g K2HPO4·3H2O, 0.5 g MgSO4· 7H2O, and 0.01 g FeSO4·7H2O per liter of water) containing nystatin (10,000 units/mL) and cyclohexanimide (5%, w/v) as an antibacterial agent, and the petri dishes were cultured at 35 °C. The actinomycetes colonies were singled out and purified to obtain pure morphologies. Genomic DNA was extracted using a DNA isolation kit (Shanghai Sangon Biotech Co., China), and 16S rRNA was amplified by polymerase chain reaction (PCR) using primers Fd2 (5′-GAGTTTGATCATGGCTCAG-3′) and 16Sr (5′-TTGCGGGACTTAACCCAACAT-3′). Finally, sequencing was performed by Shanghai Sangon Biotech Co., China. The nucleotide sequence was stored in GenBank (Accession number MK358137), and BLASTN software was used to retrieve closely related taxa from the GenBank database. The strain (No. OC1611-8A) was stored in the Laboratory of Natural Products Chemistry, Department of Pharmacy, Shandong University at Weihai.
Fermentation and separation
Strain Streptomyces sp. OC1611-8A was initially cultured on Gauze’s medium (20 g/L starch, 1 g/L KNO3, 0.5 g/L NaCl, 0.5 g/L K2HPO4·3H2O, 0.5 g/L MgSO4·7H2O, and 0.01 g/L FeSO4·7H2O) for 7 days. Then, a selected single colony was inoculated into 100 mL of Gauze’s liquid medium in Erlenmeyer flasks (250 mL) on a rotary shaker (220 r/min) at 32 °C for 4 days to prepare the seed broth. Subsequently, 10 mL of seed broth was transferred into a sterile 500 mL Erlenmeyer flask containing 150 mL of Gauze’s medium. About 30 L of this fermentation broth was incubated for 14 days at 220 r/min and 32 °C. The fermentation broth was then extracted three times with 30 L of EtOAc. The combined EtOAc solution was evaporated to dryness using a rotary evaporator at 40 °C to give 4.0 g of EtOAc extract. The extract was separated by chromatography on a silica gel (200–300 mesh) column eluting with n-hexane-acetone (10:1, 5:1, 2:1, 1:1, acetone), finally eluting with MeOH. Five fractions (A–E) were collected according to the results of TLC analysis. Fraction B (286 mg) was separated by eluting with silica gel column chromatography with n-hexane-acetone (10: 1, 5: 1, 2: 1) to obtain four subfractions B1–B4. Finally, B4 (51 mg) was purified through a C-18 reverse column (H2O-MeOH, 3:2) to give compound
Cornifronone (
Colorless oil.

Infrared (IR; KBr): 3442, 2929, 1653, 1619, 1440, 1379, 1239, 1072, and 1046 cm−1.
HRESIMS: m/z = 251.1641 ([M + H − H2O]+, calcd for C15H23O3: 251.1642).
1H NMR (500 MHz, CDCl3): δH = 5.68 (1H, s, H-2), 4.69 (1H, d, J = 11.2 Hz, H-4), 3.76 (1H, d, J = 11.3 Hz, H-13), 3.58 (1H, d, J = 11.25 Hz, H-13), 2.59 (1H, m, H-5), 2.24 (1H, dd, J = 11.6 Hz, 5.6 Hz, H-10), 2.04 (3H, s, Me-15), 2.01 (1H, m, H-6), 1.74 (2H, m, H-8), 1.73 (2H, m, H-7), 1.58 (1H, m, H-9), 1.29 (3H, s, Me-12), 1.04 (3H, d, J = 6.45 Hz, Me-14). 13C NMR (125 MHz, CDCl3): δC = 200.2 (C-1), 162.6 (C-3), 125.6 (C-2), 85.9 (C-11), 75.5 (C-4), 66.7 (C-13), 52.5 (C-10), 46.4 (C-5), 45.0 (C-6), 34.2 (C-8), 26.1 (C-12), 22.5 (C-7), 19.8 (C-14), 29.2 (C-9), and 18.8 (C-15).
Supplemental Material
supporting_information_2 – Supplemental material for Cornifronone: A cadinane-type sesquiterpene from a mason bee (Osmia cornifrons)–derived Streptomyces sp
Supplemental material, supporting_information_2 for Cornifronone: A cadinane-type sesquiterpene from a mason bee (Osmia cornifrons)–derived Streptomyces sp by Guo-Li Li, Ze-Yu Xu, Nan Li, Zhe Wang, Tian Tian and Tong Shen in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Collaborative Innovation Team Project of the Department of Education of Gansu Province (2017C-10), the National Natural Science Foundation of China (NFSC No. 21967015), the Science and Technology Major Project of Gansu Province (18ZD2NA005), the Natural Science Foundation for Young Scholars of Gansu Province (17JR5RA086), and the Young Scholars Science Foundation of Lanzhou Jiaotong University (2017006).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
