Abstract
Novel 2,6-dihydroxyacetophenone[4]arene derivatives with an electron-withdrawing group at the ortho-position are developed. The 2,6-dihydroxyacetophenone[4]arene derivatives are condensed with benzylamine in chloroform to give the Schiff base derivatives of resorcin[4]arene. The acetyl group of the 2,6-dihydroxyacetophenone[4]arenes is condensed with primary amine groups to form imines. The multifunctional target molecules are purified and isolated in good yields. The Schiff base derivatives are characterized by elemental analysis, 1H and 13C nuclear magnetic resonance spectroscopy, infrared spectroscopy, and mass spectrometry. All the Schiff base compounds are successfully used for the extraction of toxic metals including Ni2+, Mn2+, Hg2+, Co2+, and Na+ using a liquid–liquid solvent extraction process. Among these tested metals, the percentage extraction of Hg2+ is the highest. As sodium dichromate has greater oxidative stability to transfer the HCr2O7− anion from an aqueous a protonated ligand solution, an anionic extraction study of dichromate is tested at various pH values.

Introduction
The synthesis of calix[4]resorcinarenes by acid-catalyzed cyclocondensation of resorcinol with aliphatic or aromatic aldehydes has received increasing attention in recent times.1–3 Calix[4]resorcinarenes, having unique sizes and bowl-like cavities, can hold certain ions and neutral molecules by host–guest interactions and have been used in supramolecular chemistry applications in numerous fields. 4 The field of tetra-aminoresorcin[4]arenes has been studied extensively and these derivatives can be used to obtain advanced functionalized resorcin[4]arene cavities with specific receptor properties. 5 The synthesis and other receptor properties of hetero-functionalized derivatives have been described by Glushko et al. 6 The reactions of tetraformyl resorcin[4]arene with amines to obtain imines have been developed. 7 Tetraformyl resorcin[4]arene imines, containing free OH groups, can serve as important intermediates for the construction of supramolecular assemblies and have been shown to have great potential in the formation of new types of inherently chiral cavity–containing compounds. 7 Schiff bases are very suitable for the formation of metal complexes and in coordination chemistry where they are involved in the formation of chelates.8–15 New calix[4]arene Schiff bases have been synthesized and their transition temperatures and thermal behavior studies have been described in the literature by polarizing optical microscopy (POM) and differential scanning calorimetry (DSC) techniques. 16
Heavy metals create many issues worldwide because of their high toxicity effects on humans and animals. Some toxic heavy metals, such as cadmium, lead, mercury, and chromium, are found in industrial wastewaters produced from metal-plating, smelting, battery manufacturing, tanneries, petroleum refining, paints, pesticides, pigment manufacturing, and so on.17–20 Recently, the molecular recognition of anionic guests through electron-lacking neutral abiotic receptor molecules has been studied. The significance of favorable hydrogen-bonding interactions of amines, amides, or imides (–NH2/OC–NH/OC=N) for anion binding has been exploited in recent times for the design of calix[4]arene anion receptors. Calix[4]arene-based chelating units can be used for numerous studies on anion coordination. 21 In addition, there are a number of articles reporting the use of Schiff base ligands for the detection of cations.22–28 Benzimidazole-based receptors show anion binding with some anions as detected by UV-Vis and fluorescence techniques.29,30 Liquid–liquid extraction methods were used to extract Hg2+ ions by chromogen ionophore calix[4]arene-aza-crown ether derivatives 31 and by the aza-ligating site of p-tert-butylcalix[4]arene aza crown ethers. 32 Studies of dichromate anion extraction by spectrophotometric method with p-tert-butylcalix[4]arene β-ketamine derivatives have been published. 33 Furthermore, the solid–liquid extraction study of calix[4]arene Schiff bases for chromate ions have been performed at different pH values. 34
In this paper, resorcin[4]arenes with an acetyl group at the ortho-position have been synthesized in excellent yields using sodium methoxide as the base under reflux conditions. Benzylamine was used to generate novel Schiff base derivatives of the resorcin[4]arene. These Schiff bases have been applied for the extraction studies of anionic/cationic toxic metals.
Results and discussion
The 2,6-dihydroxyacetophenone[4]arene derivatives (

Synthesis of the resorcin[4]arene Schiff bases.
Physical properties of compounds

In the 1H nuclear magnetic resonance (NMR) spectra of the imines, the eight hydroxyl groups of the parent ring are observed at about δ 12 ppm. The protons of the mono-substituted benzene rings are obtained at around δ 7.28 ppm and the signals of the –CH2– protons of benzyl groups are found at about δ 4.7 ppm. The presence of these two peaks confirms the formation of the Schiff bases with benzylamine. The characteristic signals of the bridging protons of the parent ring occurred between 4 and 4.5 ppm as a triplet. All the Schiff base compounds were effective for dichromate anion and cation extraction by liquid–liquid solvent extraction. Highly toxic chromate and dichromate anions are present in soil and water. Chromium (VI) is a human and animal carcinogen.
21
Cationic toxic metals also affected on human health.
17
Therefore, we focused on liquid–liquid extraction studies to remove the toxic anions and cations by Schiff base derivatives. Solvent extraction experiments have been performed to remove alkali metal and transition metal cations such as Co2+, Hg2+, Mn2+, Ni2+, and Na+ and dichromate anions from the aqueous phase into the organic phase using compound

Plot of the percentage extraction of metal cations with ligand

Plot of the percentage extraction of Hg2+ cations with ligands

Plot of the percentage extraction of dichromate anion versus ligands (
Extraction of metal cations with ligand

Concentration of metal cation = 1 × 10−4 M. Concentration of ligands in CH2Cl2 = 1 × 10−3 M.
We found that compound
Percentage extraction of Hg2+ with ligands

Concentration of metal cation = 1 × 10−4 M. Concentration of ligands in CH2Cl2 = 1 × 10−3 M.
Schiff bases show greater extraction capacity than 2,6-dihydroxyacetophenone[4]arene for cations because of the imine group. The imine group binds easily and provides good extraction ability toward transition metals like Hg2+. The percentage extraction of Hg2+ found in the reported literature was nearly about 50%.34,36,37 The best results are obtained with mercury due to its high electronegativity compared to the other tested metals.
The dichromate ion, which includes the oxide moiety of HCr2O7−/Cr2O72−, has potential sites to create hydrogen bonds with ligands.
34
The extraction of dichromate ions with ligands
Percentage extraction of dichromate anions with ligands

Concentration of metal anion = 1 × 10−5 M. Concentration of ligand in CH2Cl2 = 1 × 10−3 M.
At the lower pH values of aqueous solutions of dichromate, the anion will exist in the protonated form HCr2O7−. This mono-anion of HCr2O7− will have less free energy of hydration than the dianionic system Cr2O72−. 33 When HCr2O7− is transferred from the aqueous phase into the dichloromethane phase, there is a slight loss in hydration energy. 33 At lower pH values, the percentage extraction increases because of protonation of the imine nitrogen. 21 Another reason for improved extraction of dichromate anions at lower pH values is that both the development of NaHCr2O7 and the protonation of the imine nitrogen support the extraction into dichloromethane. 21 In the aqueous solution at the low pH, the free energy of hydration for alkylammonium ions is less than that of the sodium ion, hence chromate ions are extracted effectively by the ligands. 33 The maximum percentage extraction of dichromate anions occurred at pH 1.00. At more acidic pH values, the dichromate anion exists as HCr2O7−/Cr2O72−, and due to the highly acidic conditions, the HCr2O7− and Cr2O72− dimerize and become the dominate Cr(IV) form. 33 Upon addition of the NaOH solution into aqueous solution, these ligands do not extract dichromate ions because there is no protonation of the Schiff base in dichloromethane solution. 34 Another factor is that hydrophilic nature of OH improves the extraction ability at the water–dichloromethane interface, which might deliver the proton interchange for transferring Na+ ions. Under highly acidic conditions, Na2Cr2O7 is converted into HCr2O7− and then ionization in aqueous solution exists in Cr2O72− by the given ionization process 34

Extraction of an anion An− by the receptor LHn+ is assumed by the following equilibrium 33
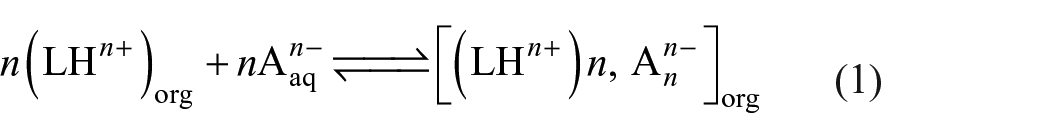
Thus, Kex is defined by equation (2) and the calculated value of Kex leads to log Kex of 5.95 for a 10−3 M concentration of ligand

Conclusion
The 2,6-dihydroxyacetophenone[4]arene derivatives (
Materials and methods
Materials
All reagents and solvents used were of L.R. grade without further purification, and the aldehydes were purchased from Sigma-Aldrich.
Instruments
Thin-layer chromatography (TLC) was run on aluminum pre-coated TLC silica gel 60 F254 plates (Merck, Germany) and visualization was performed using iodine or UV light. Melting points were recorded on a programmable Veego melting point apparatus. 1H NMR (CDCl3) and 13C NMR (CDCl3) spectra were recorded using a Bruker Avance spectrometer (400 MHz). IR spectroscopic data were recorded using a Perkin Elmer FTIR 377 spectrometer using the KBr powder pellet technique. Mass spectra were obtained using an XEVO-G2S QTOF instrument. Elemental analysis were recorded on a EURO VECTOR EA3000 CHNS-O Analyzer. The UV absorption spectra of all compounds were measured on a Jasco V-630 UV-Vis spectrophotometer.
Preparation of 2,6-dihydroxyacetophenone[4]arene derivatives (1a–h )
The derivatives of 2,6-dihydroxyacetophenone[4]arene were synthesized by following our group’s published procedure. 35 2,6-Dihydroxyacetophenone (0.500 g, 3.289 mmol) and the aliphatic aldehyde (3.289 mmol) in tetrahydrofuran (THF; 20 mL) was treated with NaOMe (0.1 g, 1.85 mmol) at room temperature. The reaction mixture was then heated at reflux with constant stirring for 72 h. After completion of the reaction, the mixture was cooled to room temperature and the solvent was evaporated under vacuum. The crude product was dissolved in MeOH (25 mL) and a few drops of AcOH were added until pH 7. A yellow precipitate was formed in the solution and it was stirred at 0–5 °C for 30 min. The product was then filtered, washed with cold methanol, and purified by column chromatography using ethyl acetate and hexane and then dried at 40–45 °C. All the synthesized compounds have been characterized by IR, 1H and 13C NMR, and mass spectrometry.
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetraethyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (2a ); general procedure
A mixture of compound
The other derivatives of the Schiff bases were synthesized using a similar pathway from compounds
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetrapropyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetrabutyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetrapentyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetrahexyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetraheptyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
15-(1-(Benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetraoctyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
15-(1-(benzylimino)ethyl)-35,55,75-tris(1-(benzylimino)ethyl)-2,4,6,8-tetranonyl-1,3,5,7(1,3)-tetra-benzenacyclooctaphan-14,16,34,36,54,56,74,76-octol (
Analytical procedure for the extraction study
All the cations of Ni2+, Mn2+, Hg2+, Co2+, and Na+ were taken directly from the acetate or sulfate salts of the metals. All the cations and sodium dichromate anion (Na2Cr2O7) solutions were prepared in an aqueous medium. Cation and anion salts of the metals were taken at pH 6 in aqueous medium at concentrations of 1 × 10−4 and 1 × 10−5 M, respectively. The anion extraction experiment was performed by changing the pH values of the dichromate ion solution using 0.01 M HCl/0.01 M KOH solution. The pH solutions were kept at 6.0, 5.0, 4.0, 3.0, 2.0, and 1.0 for the dichromate solutions.21,33,34 Ligand solutions of concentration 1 × 10−3 M were prepared in CH2Cl2 as the solvent. A liquid–liquid extraction study was applied to obtain the host and guest interactions. In the analytical experiment, a solution of the cation or anion (10 mL) and the ligand

where A0 is the initial concentration of the cation/anion solution before extraction and A is the final concentration of the cation/anion solution after extraction.
Supplemental Material
CHL915871_Supplemental_Material_CLN – Supplemental material for Resorcin[4]arene Schiff base derivatives: Synthesis, characterization, and extraction studies
Supplemental material, CHL915871_Supplemental_Material_CLN for Resorcin[4]arene Schiff base derivatives: Synthesis, characterization, and extraction studies by Juhi B Upadhyay and Hitesh M Parekh in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are thankful to the Head of the Chemistry Department, Gujarat University for necessary laboratory facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: We are thankful to the financial support by UGC-BSR for research start-up grants.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
