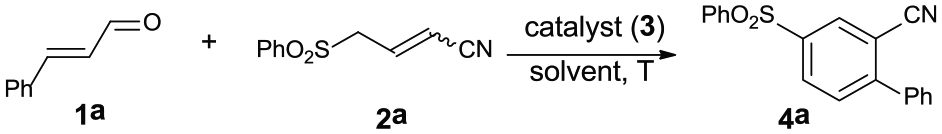
Abstract
A straightforward synthesis of benzonitriles is achieved via amino-catalyzed [3+3] benzannulation of α,β-unsaturated aldehydes and 4-arylsulfonyl-2-butenenitriles. Using pyrrolidine as an organocatalyst via iminium activation, a series of substituted benzonitriles were obtained in good to high yields in a regioselective manner. This reaction can proceed smoothly under mild reaction conditions and without the aid of any metals, additional oxidants, or strong bases, thus making this an efficient and environmentally friendly method to access benzonitriles.
Keywords
Introduction
Benzonitriles are privileged structural elements found in a number of pharmaceuticals, agrochemicals, and natural products. 1 Diverse biological activities render this kind of molecule therapeutically important agents in medicinal chemistry. For example, citalopram has been used as an antidepressant in clinical practice (Figure 1). 2 Letrozole has been ratified as a marked drug to treat breast cancer. 3 Bicalutamide, as a nonsteroidal antiandrogen, has been approved by Food and Drug Administration (FDA) for the treatment of prostate cancer. 4 Cromakalim, which could open the potassium channel, is a potential drug used in therapy of high blood pressure. 5 Apart from these, the cyano group in benzonitriles also plays as an important functional group in organic synthesis due to the multiple possibilities of transforming into other functional groups such as carboxylic acids, amide, and amine. 6

Examples of pharmaceutically important benzonitriles.
Owing to the importance and noteworthy utility of aromatic nitriles, numerous efforts are devoted for their synthesis. One of the most representative methods is the traditional SNAr reactions, in which aromatics such as sodium benzenesulfonates, benzenediazonium chlorides, and bromobenzenes react with cyanides via a C–C bond formation process. 7 Although SNAr approach has long been proved to be efficient in incorporating a cyano group into aromatic rings, it suffers from the common drawbacks of employing toxic cyanide sources and harsh reaction conditions and generating a stoichiometric amount of heavy metal waste. On the other hand, transition-metal-catalyzed cross-coupling reactions with a variety of cyanating reactants have been established as well. 8 However, requiring expensive reagents and contamination with heavy metal residues restrict their practical applicability.
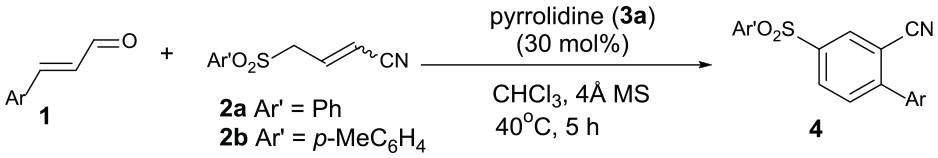
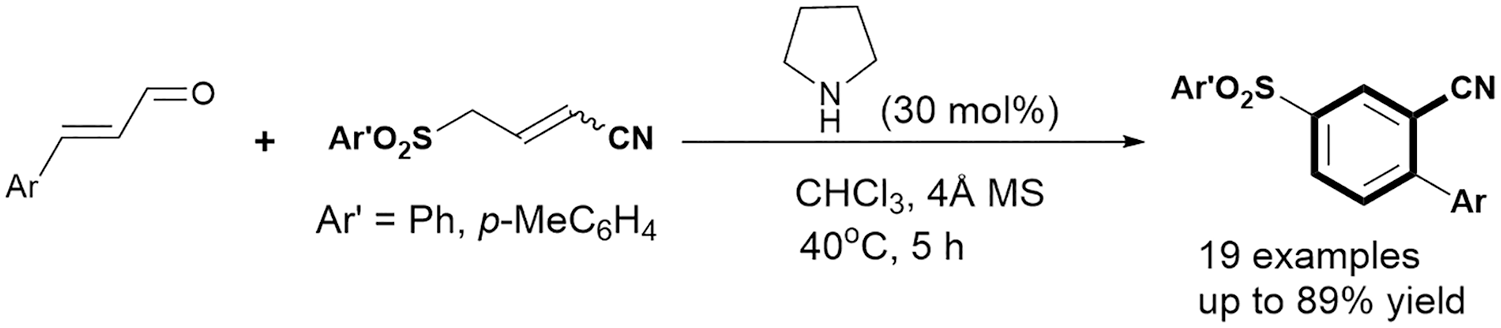
In recent years, benzannulation reaction, an assembly of readily available acyclic precursors into polysubstituted arenes, has received much attention due to its high efficiency, functional flexibility, and superior control of regiochemical outcome.9,10 In this area, formation of benzonitrile cores relies largely on the rational integration of acyclic building blocks which bear one or more cyano groups into aromatic scaffolds. Hence, synthesis of benzannulated benzonitriles to a great extent avoids introducing toxic cyanide sources and provides much broader substrate scope. Despite being more in line with the requirements of environmental concern, the examples of building benzonitrile backbones via metal-free benzannulation are still relatively few compared to those by transition-metal catalysis. 11 In 2007, the Deng group reported the base-mediated [4+2] benzannulation of vinyl malononitriles and nitroolefins. 12 Since then, various types of benzannulation such as [3+2+1],13,14 [3+3],15–17 and [4+2] 18 have been uncovered. As an alternative, Wang 19 and Ye 20 groups independently reported a benzannulation route by N-heterocyclic carbene catalysis. However, we notice that most of these metal-free cases require the attendance of added strong bases for nucleophilic activation in mechanism. In view of the synthetic value and inspired by the growing demand for developing new benzannulation strategies to diversify substituted benzonitriles, we report here the synthesis of substituted benzonitriles via amino-catalyzed regioselective [3+3] benzannulation of enals and 4-arylsulfonyl-2-butenenitriles.
Results and discussion
To obtain the optimized benzannulation conditions for substituted benzonitriles, we selected (E)-cinnamaldehyde
Optimization of reaction conditions. a

DMAP: dimethylaminopyridine; DCE: dichloroethane; THF: tetrahydrofuran.
Reaction conditions: unless otherwise noted, [3+3] benzannulation was conducted with (E)-cinnamaldehyde
Yield of isolated product.
20 mol% of catalyst.
10 mol% of catalyst.
2 mL of solvent.
0.03 mmol of AcOH was added.
50 mg 4Å MS was added.
With the optimized reaction conditions in hand, we evaluated the substrate scope of this amino-catalyzed [3+3] benzannulation reaction. As depicted in Table 2, through reaction with 4-arylsulfonyl-2-butenenitrile
Substrate scope of the [3+3] benzannulation. a

Reaction conditions: all reactions were carried out with
Yield of isolated product.
A gram-scale reaction of
The structure of benzannulated benzonitrile
On the basis of the above results and earlier report,
22
a possible mechanism for this amino-catalyzed [3+3] benzannulation reaction is proposed in Scheme 1. Upon treatment of catalytic amount of pyrrolidine

Proposed reaction mechanism.
Conclusion
In summary, we have described an efficient method for the regioselective synthesis of multi-substituted benzonitriles via [3+3] benzannulation of α,β-unsaturated aldehydes with 4-arylsulfonyl-2-butenenitriles. Promoted by iminium activation of nucleophilic amonicatalyst, the benzannulation reaction proceeds with absolute regioselectivity and displays good substrate scope, and generates a series of benzonitriles bearing a sulfonyl group rapidly in good to high yields under the optimized reaction conditions. Compared with previous reports, this methodology involves no transition-metal catalysts or any added strong bases. Thus, it has the advantages of environmentally benign, mild reaction conditions and convenient in application. Further studies on organocatalytic benzannulation reactions are currently underway.
Experiment
1H NMR and 13C NMR spectra were recorded on a Bruker AV 400 MHz spectrometer with tetramethylsilane (TMS) as internal reference. High-resolution electrospray ionization mass spectrometry (ESI HRMS) was recorded on a Waters SYNAPT G2 mass spectrometer. Melting points were determined in open capillary tubes and were uncorrected. Flash column chromatography was performed on silica gel (200–300 mesh). All solvents were dried and redistilled before use. Chemical reagents were used as commercially available and without further purification, unless otherwise stated.
Synthesis of 4-arylsulfonyl-2-butenenitrile (2a , 2b ); general procedure
Synthetic procedure was based on the literature with little modification.23,24 To a solution of 3-butenenitrile (5 g, 74.5 mmol) in CCl4 (50 mL) was added bromine (4.1 mL, 78.2 mmol) in CCl4 (20 mL) dropwise with stirring at 0 °C over 10 min. Stirring was continued at room temperature for 1 h. After the completion of the reaction, the reaction was quenched with saturated aqueous Na2S2O3 and then extracted with CH2Cl2 (25 mL × 3). The combined organic phase was washed sequentially with water, saturated brine and dried over anhydrous Na2SO4 and concentrated in vacuum. The crude adduct, 3,4-dibromobutanenitrile (16.1 g, 95%), was used without further purification.
To a solution of 3,4-dibromobutanenitrile (16.1 g, 70.8 mmol) in dry THF (80 mL) was added Et3N (10.3 mL, 74.3 mmol) dropwise in a period of 0.5 h at 0 °C. Then the reaction was stirred at the same temperature for 0.5 h. After the completion of the reaction, the reaction was quenched with saturated aqueous NH4Cl and extracted with ethyl acetate (50 mL × 3). The combined organic phase was washed sequentially with water, saturated brine, and dried over anhydrous Na2SO4 and concentrated in vacuum to yield crude 4-bromobut-2-enenitrile (8.5 g, 82%), which was used without further purification.
Sodium sulfinate (69.7 mmol) was added in one portion to a solution of 4-bromobut-2-enenitrile (8.5 g, 58.1 mmol) in ethanol (100 mL). The reaction mixture was reflux for 4 h. After consumption of the 4-bromobut-2-enenitrile monitored by thin-layer chromatography (TLC) analysis, the solvent was removed in vacuum. The mixture was added water and extracted with CH2Cl2 (25 mL × 3). The combined organic phase was washed sequentially with water, saturated brine and dried over anhydrous Na2SO4 and concentrated in vacuum. The residue was recrystallized from ethanol to give the desired 4-arylsulfonyl-2-butenenitrile
4-Phenylsulfonyl-2-butenenitrile (
4-p-(Toluenesulfonyl)-2-butenenitrile (
Synthesis of benzonitriles (4a –s ): general procedure
To a mixture of α,β-unsaturated aldehyde
4-(Phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
4′-Chloro-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
4′-Bromo-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
3′-Chloro-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
3′-Bromo-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
2′-Chloro-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
2′-Bromo-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
4-(Phenylsulfonyl)-4′-(trifluoromethyl)-[1,1′-biphenyl]-2-carbonitrile (
4′-Nitro-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
3′-Nitro-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
3′,4′-Dichloro-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
4′-Methyl-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
3′-Methyl-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
4′-Methoxy-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
3′,4′-Dimethoxy-4-(phenylsulfonyl)-[1,1′-biphenyl]-2-carbonitrile (
2-(Naphthalen-1-yl)-5-(phenylsulfonyl)benzonitrile (
2-(Furan-2-yl)-5-(phenylsulfonyl)benzonitrile (
5-(Phenylsulfonyl)-2-(thiophen-2-yl)benzonitrile (
4-Tosyl-[1,1′-biphenyl]-2-carbonitrile (
Supplemental Material
Lin_Jiang-SUPPLEMENTARY_INFORMATION-200103 – Supplemental material for Regioselective synthesis of benzonitriles via amino-catalyzed [3+3] benzannulation reaction
Supplemental material, Lin_Jiang-SUPPLEMENTARY_INFORMATION-200103 for Regioselective synthesis of benzonitriles via amino-catalyzed [3+3] benzannulation reaction by Lin Jiang, Wen-Fei Jin, Liu-Dong Yu, Ming-Wei Yuan, Hong-Li Li, Deng-Bang Jiang and Ming-Long Yuan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the National Natural Science Foundation of China (21302163).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.