Abstract
The utility of Cu(OTf)2 as the catalyst for the synthesis of a series of N-tert-butyl amides in excellent isolated yields via the reaction of nitriles (alkyl, aryl, benzyl, and furyl nitriles) with di-tert-butyl dicarbonate is described. Cu(OTf)2 is a highly stable and efficient catalyst for the present Ritter reaction under solvent-free conditions at room temperature.
A convenient synthesis of N-tert-butyl amides by the reaction of di-tert-butyl dicarbonate and nitriles catalyzed by Cu(OTf)2.

Introduction
The N-tert-butyl amide group is found in many drugs such as finasteride, 1 nelfinavir, 2 and CPI-1189. 3 Finasteride is used to treat benign prostatic hyperplasia. As a protease inhibitor, nelfinavir has been developed as a component in the treatment of HIV. CPI-1189, with antioxidant properties, can be applied as a drug candidate for neuroprotective therapy to treat HIV-associated central nervous system (CNS) disease.
Usually, N-tert-butyl amides are synthesized by the condensation of carboxylic acids with tert-butyl amines, 4 the oxidative amidation of alcohols, 5 the amidation of aryl halides, 6 or via the Ritter reaction.
Initially, the synthesis of N-tert-butyl amides via the reaction of tert-butyl amines with benzoic acid or its derivatives was developed using 4-dimethylaminopyridine (DMAP), 7 N,N-diisopropylethylamine (DIPEA), 8 or hydroxybenzotriazole (HOBt) 7 as condensing agents. This route has several drawbacks including difficult product separation and the addition of condensation agents (Scheme 1a). The reaction of tert-butyl bromide and amides with heavy metal catalysts (such as Mn2(CO)10) or alkali produced a wide variety of N-tert-butyl amides at high temperature (Scheme 1b). 9 In 2016, Xu et al. 10 reported the reaction of an aromatic aldehyde with tert-butyl amine to prepare N-tert-butyl benzamide at 100 °C (Scheme 1c), however, this method could only be applied to aromatic compounds. In the Ritter reaction (Scheme 1d–h), tert-butanol,11,12 tert-butyl bromide,13,14 tert-butyl acetate,15–18 and tert-butyl benzoate 19 were employed with nitriles to produce N-tert-butyl amides. In addition to the above methods, methyl tert-butyl ethers,20,21 methyl ethers, 22 and di-tert-butyl dicarbonate 23 can also be used to synthesize N-tert-butyl amides. In 2011, Kalkhambkar et al. 14 produced N-tert-butyl amides from tert-butyl bromide and nitriles using 1-butyl-3-methylimidazolium hexafluorophosphate (BMIM[PF6])/NOPF6 as the catalytic system (Scheme 1d). Tamaddon’s group 21 demonstrated that ZnCl2/SiO2 (15 mol%) could be applied as a catalyst in the conversion of tert-butyl ethers and nitriles into the corresponding amides at 100 °C (Scheme 1f). In 2012, Khaksar et al. 18 developed pentafluorophenylammonium triflate (PFPAT) as an organocatalyst to produce a range of amides via the reaction of tert-butyl acetates with nitriles at 90 °C (Scheme 1e). Indalkar et al., 11 in 2017, reported the reaction of nitriles with tert-butanol in the presence of sulfated polyborate under solvent-free conditions at 100 °C (Scheme 1g). In 2019, Feng et al. 19 reacted nitriles with tert-butyl benzoate in the presence of Zn(ClO4)2·6H2O to synthesize N-tert-butyl amides at 50 °C (Scheme 1e). This procedure suffered from several drawbacks such as complicated post-treatment and environmental hazards. In the reactions listed above, only the reactions of nitriles with di-tert-butyl decarbonate were carried out at room temperature and excellent isolated yields of the products were obtained. In 2018, Feng et al. 23 employed di-tert-butyl dicarbonate with nitriles in the presence of Fe(ClO4)3·xH2O to form a series of N-tert-butyl amides (Scheme 1h), but the catalyst was unstable and explosive. Therefore, it is necessary to find a mild and efficient catalyst for the synthesis of N-tert-butyl amides.

Methods for the preparation of N-tert-butyl amides.
As an efficient Lewis acid catalyst, Cu(OTf)2 can be used to catalyze a series of organic reactions such as the Friedel–Crafts reaction, 24 dehydrogenative cross-coupling reactions, 25 and reductive ring-opening reactions. 26 In addition, Cu(OTf)2 is stable to moisture and can catalyze reactions under wet conditions.
In this context, we report a mild and practical method for the formation of N-tert-butyl amides via the reaction of nitriles with di-tert-butyl dicarbonate in the presence of Cu(OTf)2 under solvent-free conditions. This method can be extended to aryl nitriles, benzyl nitriles, alkyl nitriles, and furyl nitriles with excellent isolated yields of products being obtained at room temperature (Scheme 1i).
Results and discussion
Initially, the reaction of benzonitrile and di-tert-butyl dicarbonate to form N-tert-butyl benzamide was used as a model reaction. To achieve an excellent isolated yield, the reaction parameters were optimized and the results are summarized in Table 1. We screened a series of Cu catalysts including CuCl2, CuCl, CuI, CuO, Cu2O, Cu(OTf)2, CuBr, CuBr2, Cu, CuSO4·5H2O, Cu(OAc)2·H2O, and Cu(NO3)2·3H2O, of which Cu(OTf)2 was found to be the most suitable catalyst to promote the reaction in an excellent yield. However, CuCl2 afforded N-tert-butyl benzamide in only 36% yield at 50 °C (Table 1, entries 1–11). It was found that CuCl, CuI, CuO, Cu2O, CuBr, Cu, CuSO4·5H2O, and Cu(NO3)2·3H2O were all ineffective at room temperature. On the contrary, the perchlorate salts Zn(ClO4)2·6H2O and Cu(ClO4)2·6H2O produced N-tert-butyl benzamide in a moderate yield (Table 1, entries 12 and 13). Cu(OTf)2 as the most suitable catalyst produced the desired product with an excellent yield of 87% (Table 1, entry 16).
Optimization of the reaction conditions a .

RT: room temperature; NR: no reaction.
All reactions were performed with benzonitrile (5 mmol) and di-tert-butyl dicarbonate (7.5 mmol).
Isolated yield. Bold Values: Although the yield of N-tert-butyl benzamide by employment of 10 mol% catalyst was slightly increasing, 5 mol% catalyst was thought to be enough for this transformation.
Next, we investigated the influence of the temperature on this transformation (Table 1, entries 19 and 20). When the reaction temperature was increased to 40 and 60 °C, the yields were slightly improved compared to those at room temperature. We subsequently investigated the amount of the catalyst and the reaction time. The yield increased dramatically to 87% by prolonging the reaction time to 5 h (Table 1, entry 16). When the catalyst loading was 1 or 2 mol%, the yield of the target product decreased significantly to 16% and 25%, respectively (Table 1, entries 14 and 15). With the catalyst loading increased to 10 mol%, the yield of N-tert-butyl benzamide was 89% (Table 1, entry 17). It was found that 5 mol% catalyst loading was sufficient for this transformation.
To investigate the scope of this transformation, a wide range of nitriles was examined for the formation of N-tert-butyl amides under the optimized reaction conditions, and the substrate scope of the nitriles is summarized in Table 2. The reaction was compatible with a wide variety of substrates including aryl nitriles, benzyl nitriles, alkyl nitriles, and furyl nitriles, affording the corresponding N-tert-butyl amides in good to excellent yields (Table 2, products
The Ritter reaction of nitriles and di-tert-butyl dicarbonate a .
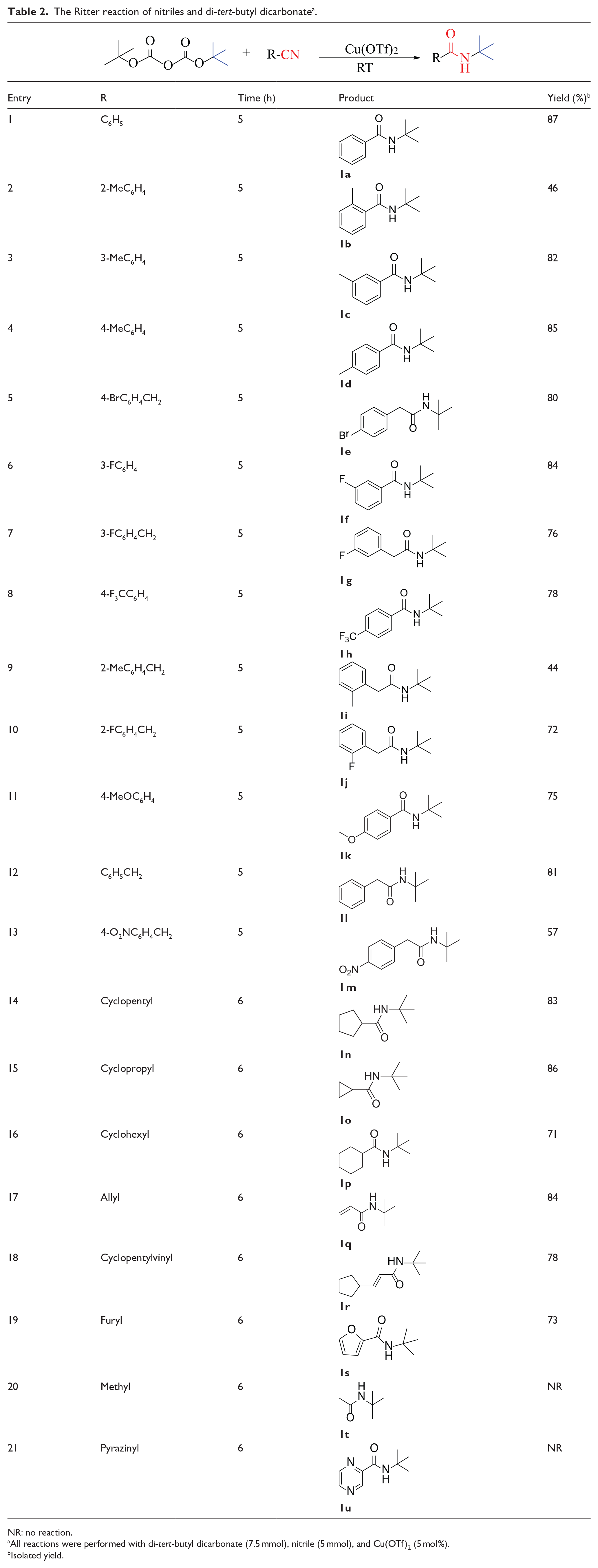
NR: no reaction.
All reactions were performed with di-tert-butyl dicarbonate (7.5 mmol), nitrile (5 mmol), and Cu(OTf)2 (5 mol%).
Isolated yield.
The reaction mechanism of this experiment can be explained by the established Ritter reaction mechanism, 21 as proposed in Scheme 2.
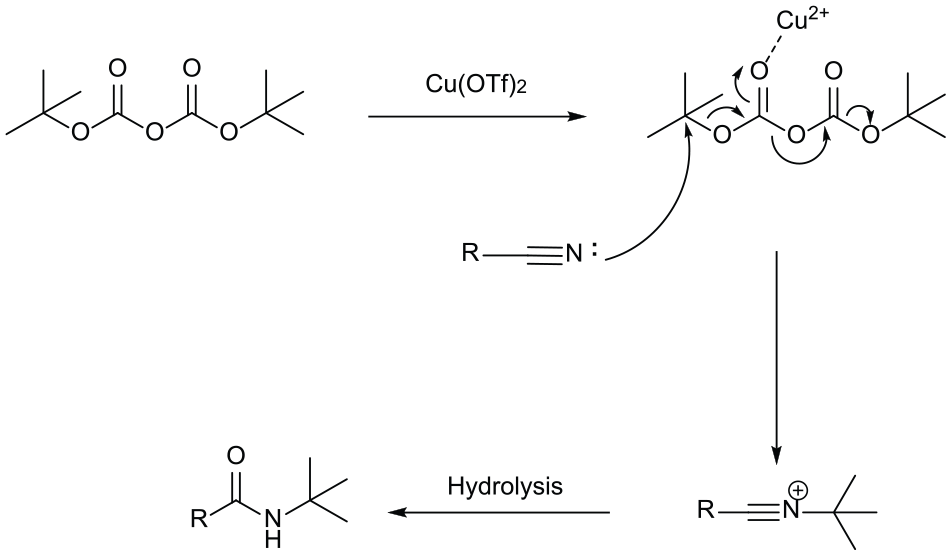
The mechanism of the Ritter reaction catalyzed by Cu(OTf)2.
Conclusion
In conclusion, we have developed a mild and practical protocol for the preparation of N-tert-butyl amides from nitriles with di-tert-butyl dicarbonate in the presence of Cu(OTf)2 under solvent-free conditions. The approach was compatible with a series of nitriles and the corresponding amides were obtained in good to excellent yields at room temperature. Besides, this procedure has numerous practical advantages such as simple post-treatment, mild reaction conditions and wide substrate scope.
Experiment
Materials and instruments
The starting materials and reagents were obtained from commercial sources without further purification unless otherwise stated. All reactions were monitored by thin-layer chromatography (TLC) with visualization of samples under UV light at 254 nm. Column chromatography was performed over aluminum oxide (neutral, 200–300 mesh, Shanghai Titan Scientific Co., Ltd, China). The melting points were determined on a YRT-3 melting point apparatus and are uncorrected. Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker Avance DPX-500/600 MHz instrument ( 1 H NMR at 500 MHz, 13C NMR at 126 MHz, 19F NMR at 565 MHz) in CDCl3 with tetramethylsilane (TMS) as the internal standard and the chemical shifts (δ) are given in parts per million (ppm). The 13C NMR spectra are decoupled from 1H.
Typical experimental procedure for the reactions of nitriles with di-tert-butyl dicarbonate
To a solution of di-tert-butyl dicarbonate (7.5 mmol) and Cu(OTf)2 (5 mol%), was slowly added dropwise the nitrile (5 mmol) at room temperature. The extent of the reaction was monitored by TLC. After quenching with water (10 mL), the solution was extracted with ethyl acetate 3 times. Finally the organic phases were collected, washed with saturated NaCl solution, dried over anhydrous sodium sulfate, and concentrated in vacuo. The residue was purified by column chromatography over aluminum oxide (200–300 mesh) using petroleum ether/ethyl acetate (10:1–5:1) as eluent.
Di-tert-butyl dicarbonate is a colorless liquid or a white crystal line solid. The melting point is 22–24 °C. The white crystal line solid at low temperature could be melted by heating. The reaction of di-tert-butyl dicarbonate with solid nitriles afforded the corresponding amides under solvent-free conditions.
Analytical data for amides 1a–s
N-(tert-butyl)benzamide
N-(tert-butyl)-2-methylbenzamide
N-(tert-butyl)-3-methylbenzamide
N-(tert-butyl)-4-methylbenzamide
2-(4-bromophenyl)-N-(tert-butyl)acetamide
N-(tert-butyl)-3-fluorobenzamide
N-(tert-butyl)-2-(3-fluorophenyl)acetamide
N-(tert-butyl)-4-(trifluoromethyl)benzamide
N-(tert-butyl)-2-(o-tolyl)acetamide
N-(tert-butyl)-2-(2-fluorophenyl)acetamide
N-(tert-butyl)-4-methoxybenzamide
N-(tert-butyl)-2-phenylacetamide (
N-(tert-butyl)-2-(4-nitrophenyl)acetamide (
N-(tert-butyl)cyclopentanecarboxamide
N-(tert-butyl)cyclopropanecarboxamide
N-(tert-butyl)cyclohexanecarboxamide
N-(tert-butyl)acrylamide (
N-(tert-butyl)-3-cyclopentylacrylamide
N-(tert-butyl)furan-2-carboxamide
Supplemental Material
supporting_information – Supplemental material for A convenient synthesis of N-tert-butyl amides by the reaction of di-tert-butyl dicarbonate and nitriles catalyzed by Cu(OTf)2
Supplemental material, supporting_information for A convenient synthesis of N-tert-butyl amides by the reaction of di-tert-butyl dicarbonate and nitriles catalyzed by Cu(OTf)2 by Wanfeng Yang, Chengliang Feng, Yuqi Tang, Min Ji and Junqing Chen in Journal of Chemical Research
Footnotes
Authors’ Note
Caution! Because some perchlorates are unstable and explosive, reactions involving perchlorates should be performed carefully behind a safety shield.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Fundamental Research Funds for the Central Universities (No. 2242014R30019).
Supplemental material
The spectra data for compounds 1a-s, 1H NMR, 13C NMR and 19F NMR spectra are available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


