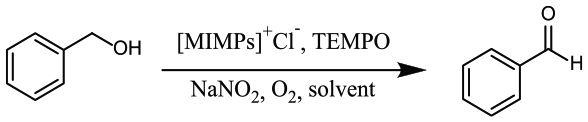Abstract
The ionic liquid 1-methyl-3-(3-sulfopropyl)imidazolium chloride ([MIMPs]+Cl-) in combination with 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) and sodium nitrite (NaNO2) as a catalytic system demonstrates high efficiency in the one-pot two-step aerobic oxidative condensation of benzyl alcohols with 1,2-phenylenediamines to give benzimidazoles. Various benzimidazoles are obtained in good to excellent yields by this strategy.
Keywords

Introduction
Benzimidazole is an important structural motif found in a wide range of natural and bioactive compounds. The benzimidazole nucleus has been employed as a privileged scaffold to synthesize various therapeutic drugs such as anti-HIV, antifungal, antitumor, anti-ulcer, anticancer, and anti-inflammatory agents.1–11 Therefore, many protocols have been developed to synthesize benzimidazoles. The traditional methods to synthesize benzimidazoles include the condensation of o-phenylenediamines with aldehydes, carboxylic acids, amides, acid chlorides, esters, and nitriles in strong acid mediums at high temperature.12–20 Benzimidazoles can also be obtained by the oxidative condensation of alcohols or amines with o-phenylenediamines.21–27 Compared to other protocols, the oxidative condensation of alcohols with o-phenylenediamines has the advantages of easy availability, low toxicity, low cost of the starting materials, and mild reaction conditions. However, transition metals, especially noble metals, are generally required in the reported oxidative condensation of alcohols with o-phenylenediamines to give benzimidazoles,22–25,28–31 which represents a big obstacle in large-scale applications. At the same time, metal-based oxidative condensation processes may leave traces of poisonous heavy metals in drugs, which are harmful to patients. Therefore, it is still necessary to develop efficient, transition metal-free, economic, and environmentally friendly protocols for the synthesis of benzimidazoles via oxidative condensation of alcohols with o-phenylenediamines.
As disclosed in the literature,13,30 the oxidative condensation of an alcohol with a o-phenylenediamine to give a benzimidazole is accomplished via three elementary steps, which are catalytic oxidation of the alcohol to an aldehyde, condensation of the aldehyde with o-phenylenediamine to afford an imine being in equilibrium with a dihydrobenzazole, and catalytic oxidative dehydrogenation of the dihydrobenzazole to the benzimidazole. In the transformation, the aerobic oxidation of the alcohol to the aldehyde is an independent step, but the condensation and dehydrogenation cyclization (CDHC) processes are inseparable, being called the second step.
The aerobic oxidation of alcohols to carbonyl compounds has been intensively investigated. Several TEMPO (2,2,6,6-tetramethylpiperidineoxy)-based transition metal-free catalytic systems have proved to be very efficient for the oxidation of alcohols to aldehydes or ketones with molecular oxygen as the terminal oxidant,32–43 among which those consisting of acid, nitrite sources, and TEMPO, such as HCl/sodium nitrite (NaNO2)/TEMPO and [Imim-TEMPO]+X/[Imim-COOH]+X/NaNO2, are the most attractive due to their high efficiency, easy availability, and mild reaction conditions.36,39 Either HCl or [Imim-COOH] is necessary to convert nitrite into nitrous acid in situ in the catalytic cycles. Besides, using [Imim-COOH] instead of HCl has the advantage of avoiding corrosion to equipment caused by chloride in the large-scale practice.
On the contrary, ionic liquids (ILs) are substances with special properties, such as exceptional thermal and chemical stability and negligible vapor pressure. Because of these important features, ILs have been increasingly applied in catalytic synthesis, either as solvents or as catalysts.44–47 In particular, some ILs as catalysts exhibit good performances in the oxidative condensation of aldehydes or benzylamines with o-phenylenediamine to give benzimidazoles.46–49 More particularly, some Brönsted acid ILs as catalysts performed well in condensation reactions to afford benzimidazoles and bis(arylidene)cycloalkanones.50,51
Inspired by the above-reported results that acidic ILs in combination with TEMPO and NaNO2 to form a transition metal-free catalytic system might be effective in one-pot, two-step synthesis of benzimidazoles from alcohols and o-phenylenediamines in an environmentally friendly manner, we prepared several 1-propylsulfonic acid-functionalized ILs. These acidic ILs in combination with TEMPO and NaNO2 were then tested in the one-pot, two-step aerobic oxidative condensation of alcohols with o-phenylenediamines to give benzimidazoles.
Results and discussion
Several ILs, as shown in Scheme 1, were synthesized and characterized. The results were in agreement with those in the literature.52–54

The structures of the ILs.
First, [MIMPs]+Cl−/NaNO2/TEMPO was evaluated in the aerobic oxidation of benzyl alcohol to benzaldehyde in a CH3CN/H2O mixture with a volume ratio of 10:1 and a temperature range of 30 °C–50 °C. As shown in Table 1, the reaction proceeded slowly at 30 °C, and the conversion of benzyl alcohol was only 11% in 7 h (Table 1, entry 1). When the reaction temperature was raised from 30 °C to 45 °C, the conversion of benzyl alcohol increased gradually, and the reaction time was also reduced (Table 1, entries 1–4). When the reaction was conducted at 45 °C for 5 h, the benzyl alcohol was converted into benzaldehyde quantitatively. A further increase in the reaction temperature to 50 °C led to a decrease in the selectivity for benzaldehyde due to over oxidation of benzaldehyde to benzoic acid (Table 1, entry 5). Next, the reaction was also performed in other solvent/water mixtures; however, poor results were obtained in all the cases (Table 1, entries 6–11). The influence of the amount of water on the whole reaction was also investigated, and the results are given in Supporting Information (Table S4). At lower water loading, longer reaction time was required to finish the oxidation of benzyl alcohol to give benzaldehyde, and the yield of 2-phenylbenzimidazole was lower. With the increase in CH3CN/H2O ratio to 2.0 mL/0.2 mL, the oxidation rate increased and the yield of 2-phenylbenzimidazole reached a maximum of 95%. A further increase in the amount of water did not affect the oxidation rate, but led to low yield of 2-phenylbenzimidazole. It is assumed that water is beneficial for [MIMPs]+Cl− to dissolve in acetonitrile. However, the high concentration of water will hinder the dehydration in the condensation reaction.
Reaction conditions for the oxidation of benzyl alcohol to benzaldehyde. a

EtOAc: ethyl acetate; DMF: dimethylformamide; DCM: dichloromethane; DME: dimethyl ether.
Unless noted otherwise, the reaction conditions are as follows: benzyl alcohol (2 mmol), [MIMPs]+Cl− (20 mol%), TEMPO (5 mol%), NaNO2 (8 mol%), CH3CN/H2O (2.0 mL/0.2 mL), and atmospheric O2.
T1: reaction temperature during oxidation of the alcohol to the aldehyde; t1: reaction time for the oxidation of the alcohol to the aldehyde.
Conversions and selectivity were based on the gas chromatography (GC) analysis with area normalization.
The other acidic ILs were then compared with [MIMPs]+Cl− in the aerobic oxidation of benzyl alcohol to benzaldehyde under the same conditions (step 1), followed by evaluation in the condensation of benzaldehyde generated in situ with o-phenylenediamine to give 2-phenylbenzimidazole. As shown in conversion and selectivity columns in Table 2, the catalytic systems from all the ILs in combination with TEMPO and NaNO2 were efficient in the aerobic oxidation of benzyl alcohol to benzaldehyde, and benzaldehyde was achieved quantitatively except for the cases of
Acidic ILs catalyzed aerobic oxidative condensation of benzyl alcohol with o-phenylenediamine to give 2-phenylbenzimidazole. a

IL: ionic liquid.
Unless noted otherwise, the reaction conditions are as follows: benzyl alcohol (2 mmol), o-phenylenediamine (2 mmol), IL (20 mol%), TEMPO (5 mol%), NaNO2 (8 mol%), CH3CN/H2O (2.0 mL/0.2 mL), T1 = 45 °C, T2 = 45 °C, and atmospheric O2.
t1: reaction time in step 1; t2: reaction time in step 2.
Conversions and selectivity, referring to the alcohol oxidation step, were based on the gas chromatography (GC) analysis with area normalization.
Isolated yields of 2-phenylbenzimidazole from benzyl alcohol and o-phenylenediamine.
With these results in hand, the IL/NaNO2/TEMPO catalytic systems were then applied to the one-pot, two-step aerobic oxidative condensation of benzyl alcohol with o-phenylenediamine to give 2-phenylbenzimidazole at 45 °C in both steps. To our delight, an isolated yield of 85% of 2-phenylbenzimidazole was obtained after the CDHC reaction lasting for 8 h under the catalysis of [MIMPs]+Cl−/NaNO2/TEMPO (Table 2, entry 1). However, much lower yields of 2-phenylbenzimidazole were obtained under the catalysis of other IL/NaNO2/TEMPO catalytic systems (Table 2, entries 2–8). The results indicated that the aerobic oxidative condensation of benzyl alcohol with o-phenylenediamine to give 2-phenylbenzimidazole was mainly affected by the second step, that is, the CDHC reaction. Obviously, the CDHC reaction was related to both the cationic and anionic moieties of the ILs. Except for the ILs with an
Next, the effect of the catalyst loading on the whole reaction was investigated, and the results are given in Table S1 (see the Supporting Information). In the case of 15 mol% of [MIMPs]+Cl−, 5 mol% of TEMPO, and 8 mol% of NaNO2, the conversion of benzyl alcohol was only 26%, which was not enough to support the second-step reaction. With an increase in the [MIMPs]+Cl− loading, and keeping the amounts of TEMPO and NaNO2 constant, the time to finish the transformation of benzyl alcohol into benzaldehyde always decreased (Table S1, entries 1–5). However, the yield of benzimidazole reached a maximum of 85% at the [MIMPs]+Cl− loading of 20 mol% (Table S1, entry 3). A further increase in the catalyst loading to 25 mol% led to a decrease in the yield of 2-phenylbenzimidazole to 72%, which might be due to the combination of acidic [MIMPs]+Cl− with basic o-phenylenediamine, preventing the condensation reaction of benzaldehyde with o-phenylenediamine. The optimum loadings of [MIMPs]+Cl−, NaNO2, and TEMPO were determined to be 20, 8, and 5 mol%, respectively.
After determination of the optimum catalytic system, the effect of the molar ratio of benzyl alcohol to o-phenylenediamine was investigated by maintaining the reaction conditions constant. As shown in Table S2 (see the Supporting Information), the yield of 2-phenylbenzimidazole initially increased with an increase in the molar ratio of o-phenylenediamine to benzyl alcohol, and then decreased slightly with a further increase in the molar ratio. The highest yield of 90% was obtained when the molar ratio of o-phenylenediamine to benzyl alcohol was 1:1.1. A higher amount of o-phenylenediamine could suppress the formation of 1-benzyl-2-phenylbenzimidazole, which is a major by-product in the synthesis of 2-phenylbenzimidazole. However, a much higher quantity of o-phenylenediamine will also lead to combination of [MIMPs]+Cl− with the basic o-phenylenediamine, which will decrease the catalysis of [MIMPs]+Cl− in the CDHC of the aldehyde with o-phenylenediamine to form 2-phenylbenzimidazole.
Finally, the effect of temperature on the CDHC reaction was studied by maintaining the other reaction conditions constant. As shown in Table S3 (see the Supporting Information), the yield of 2-phenylbenzimidazole increased gradually with an increase in the reaction temperature and reached a maximum of 95% at 55 °C (Table S3, entries 1–3). A further increase in the reaction temperature led to a decrease in the yield of 2-phenylbenzimidazole due to the preferred formation of 1-benzyl-2-phenylbenzimidazole at high temperature (Table S3, entries 4 and 5).
For further confirmation of the catalysis by the [MIMPs]+Cl−/NaNO2/TEMPO system on the CDHC reaction, the condensation of benzaldehyde with o-phenylenediamine to give 2-phenylbenzimidazole was performed in the absence of [MIMPs]+Cl−/NaNO2/TEMPO. The reaction proceeded very slowly, and 2-phenylbenzimidazole was obtained in a yield of only 7% after reaction for 5 h (Table S3, entry 6).
Based on the above results, the optimum reaction conditions for the synthesis of 2-phenylbenzimidazole from aerobic oxidative condensation of benzyl alcohol with o-phenylenediamine were obtained, which were as follows: CH3CN/H2O (10:1) as the solvent; the molar ratio of benzyl alcohol to o-phenylenediamine is 1:1.1; the loadings of [MIMPs]+Cl−, TEMPO, and NaNO2 are 20, 5, and 8 mol%, respectively; the reaction temperature and time in the first step are 45 °C and 5 h, respectively; and the reaction temperature and time in the second step are 55 °C and 5 h, respectively. Under these conditions, the yield of 2-phenylbenzimidazole was 95%, which is higher than the results in the literature.28–30
Having optimized the reaction condition, the substrate scope was studied to demonstrate the generality of this strategy. As shown in Table 3, a series of substituted benzyl alcohols could be condensed with o-phenylenediamine to give the corresponding benzimidazoles (
Unless noted otherwise, the reaction conditions are as follows: alcohol (2 mmol), diamine (2.2 mmol), [MIMPs]+Cl− (20 mol%), TEMPO (5 mol%), NaNO2 (8 mol%), CH3CN/H2O (2.0 mL/0.2 mL), T1 = 45 °C, t1 = 5 h, T2 = 55 °C, t2 = 5 h, and atmospheric O2.
Isolated yields.
The reaction time (t1) in the first step is 30 h.
In order to illustrate the usefulness of this reaction in the synthesis of benzimidazoles, a gram-scale experiment was carried out (Scheme 2). This reaction could be readily scaled up to 20 mmol, and the target product

Gram-scale experiment.
The above experimental results manifest the following three facts: (1) all the catalytic systems from the eight acidic ILs can catalyze the aerobic oxidation of benzyl alcohol to benzaldehyde, (2) [MIMPs]+Cl− demonstrates the best catalyst performance in the conversion of benzaldehyde and o-phenylenediamine into 2-phenylbenzimidazole, and (3) almost no 2-phenylbenzimidazole is obtained from benzaldehyde with o-phenylenediamine in the absence of [MIMPs]+Cl−/NaNO2/TEMPO.
Conclusion
In conclusion, a catalytic system, [MIMPs]+Cl−/NaNO2/TEMPO, has been proved to be very efficient for the aerobic oxidative condensation of benzyl alcohols with o-phenylenediamines to give the corresponding benzimidazoles. This strategy tolerates a wide scope of both substituted benzyl alcohols and substituted o-phenylenediamines, and various benzimidazoles can be synthesized in good to excellent yields. The acid IL [MIMPs]+Cl− acts both as an acid to transform NaNO2 into HNO2 and as a catalyst to promote condensation of the aldehyde with the o-phenylenediamine followed by intramolecular cyclization. TEMPO, which can be regenerated in the catalytic cycles, not only oxidizes the alcohol to the aldehyde in the first step but also converts the dihydrobenzazole into the benzimidazole via oxidative dehydrogenation in the second step.
Experimental
Preparation of acidic ILs
The acidic ILs were prepared as described in the literature,52–54 and the procedures are given in the Supporting Information.
General experimental details
The starting materials were obtained from the Macklin Biochemical Technology Co., Ltd or Aladdin Chemical and used as received, without further purification. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AC-P 400 spectrometer (400 MHz for 1H and 101 MHz for 13C) in dimethyl sulfoxide (DMSO)-d6 and CDCl3 with tetramethylsilane (TMS) as the internal standard or D2O. The data are reported as follows: chemical shift, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet), and coupling constant, J, which is reported in hertz. The Fourier-transform infrared spectroscopy (FTIR) spectra were recorded on a Bruker Vector 22 type instrument as KBr pellets. Melting points were measured on a Shanghai Inesa WRS-3 melting-point apparatus without calibration. The thin-layer chromatography (TLC) was performed using 60 mesh silica gel plates visualized with short-wavelength ultraviolet (UV) light (254 nm).
General catalytic procedures
TEMPO (5 mol%), NaNO2 (8 mol%), and the IL (20 mol%) were added successively into a 5-mL round-bottom flask that was purged with oxygen five times. Subsequently, benzyl alcohol (2 mmol) and CH3CN/H2O (10:1; 2.2 mL) were added in turn via a syringe. Then, the mixture was stirred at 45 °C in an oil bath under atmospheric oxygen. When the benzyl alcohol had been consumed (monitored by TLC), o-phenylenediamine (2.2 mmol) was added. The mixture was further stirred at 55 °C under atmospheric oxygen until completion of the reaction (monitored by TLC). The reaction mixture was extracted with dichloromethane (4 × 15 mL), and the combined extract was evaporated under vacuum. The crude product was purified by column chromatography on silica gel to afford product
2-Phenyl-1H-benzo[d]imidazole (
2-(p-Tolyl)-1H-benzo[d]imidazole (
2-(m-Tolyl)-1H-benzo[d]imidazole (
2-(o-Tolyl)-1H-benzo[d]imidazole (
2-(4-Methoxyphenyl)-1H-benzo[d]imidazole (
2-(3-Methoxyphenyl)-1H-benzo[d]imidazole (
2-(2-Methoxyphenyl)-1H-benzo[d]imidazole (
2-(4-Fluorophenyl)-1H-benzo[d]imidazole (
2-(3-Fluorophenyl)-1H-benzo[d]imidazole (
2-(2-Fluorophenyl)-1H-benzo[d]imidazole (
2-(4-Chlorophenyl)-1H-benzo[d]imidazole (
2-(3-Chlorophenyl)-1H-benzo[d]imidazole (
2-(2-Chlorophenyl)-1H-benzo[d]imidazole (
2-(4-Bromophenyl)-1H-benzo[d]imidazole (
2-(3-Bromophenyl)-1H-benzo[d]imidazole (
2-(2-Bromophenyl)-1H-benzo[d]imidazole (
2-(4-Nitrophenyl)-1H-benzo[d]imidazole (
2-(2-Nitrophenyl)-1H-benzo[d]imidazole (
2-(3,5-Difluorophenyl)-1H-benzo[d]imidazole (
2-(Furan-2-yl)-1H-benzo[d]imidazole (
2-(Thiophen-2-yl)-1H-benzo[d]imidazole (
4-Methyl-2-phenyl-1H-benzo[d]imidazole (
5-Methyl-2-phenyl-1H-benzo[d]imidazole (
2-Phenyl-5-(trifluoromethyl)-1H-benzo[d]imidazole (
5-Fluoro-2-phenyl-1H-benzo[d]imidazole (
5-Chloro-2-phenyl-1H-benzo[d]imidazole (
5-Nitro-2-phenyl-1H-benzo[d]imidazole (
5,6-Dimethyl-2-phenyl-1H-benzo[d]imidazole (
5,6-Difluoro-2-phenyl-1H-benzo[d]imidazole (
5,6-Dichloro-2-phenyl-1H-benzo[d]imidazole (
2-(Naphthalen-2-yl)-1H-benzo[d]imidazole (
2-Cyclohexyl-1H-benzo[d]imidazole (
2-(2,6-Dimethylhepta-1,5-dien-1-yl)-1H-benzo[d]imidazole (
1-Hexyl-2-pentyl-1H-benzo[d]imidazole (
Supplemental Material
Supporting_Information – Supplemental material for A one-pot synthesis of benzimidazoles via aerobic oxidative condensation of benzyl alcohols with o-phenylenediamines catalyzed by [MIMPs]+Cl-/NaNO2/TEMPO
Supplemental material, Supporting_Information for A one-pot synthesis of benzimidazoles via aerobic oxidative condensation of benzyl alcohols with o-phenylenediamines catalyzed by [MIMPs]+Cl-/NaNO2/TEMPO by Zhenzhen Geng, Hong-yu Zhang, Guohui Yin, Yuecheng Zhang and Jiquan Zhao in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors appreciate the financial support from the National Natural Science Foundation of China (grant no. 21776056) and the Natural Science Foundation of Hebei Province (CN) (grant no. B2018202253).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.