Abstract
7-Bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one is prepared from 6-methoxybenzo[d]thiazol-2-amine and 2-(bromomethyl)-2-butylhexanoic acid as the key starting materials via five simple steps including hydrolysis, substitution, condensation, bromination, and aromatic amidation under microwave conditions. This new route has reduced the reaction time and increased the overall yield to 43%. Moreover, the structure of the target product is also confirmed by X-ray crystal analysis, and further studies indicate that the existence of an intramolecular C–H···Cg1 non-classical hydrogen bond is effective in stabilization of the crystal structure.
Introduction
Constipation is a common digestive disease having a prevalence rate of about 20% in China. It occurs more frequently in women than in men, and the prevalence rate is higher in older people. 1 Prucalopride succinate is now on the market for the treatment of constipation in women.2,3 In addition, stool softeners increase water absorption by the faeces by lowering the surface tension of the feces, a representative example being docusate. 4 However, these stimulant laxatives and stool softeners only provide temporary effects.
Recent medical research has demonstrated that the transfer of bile acids is one of the leading causes of constipation. Benzothiazepine derivatives inhibit bile acid transport that regulates bile acid reabsorption,5–7 thus increasing bile acid flow to the colon, thereby promoting the intestinal tract to secrete more water and facilitate defecation, so as to naturally improve natural defecation in patients. On 19 April 2018, EA Pharma and Mochida launched the World’s first bile acid transport inhibitor in Japan. Its generic name is Elobixibat hydrate. 8
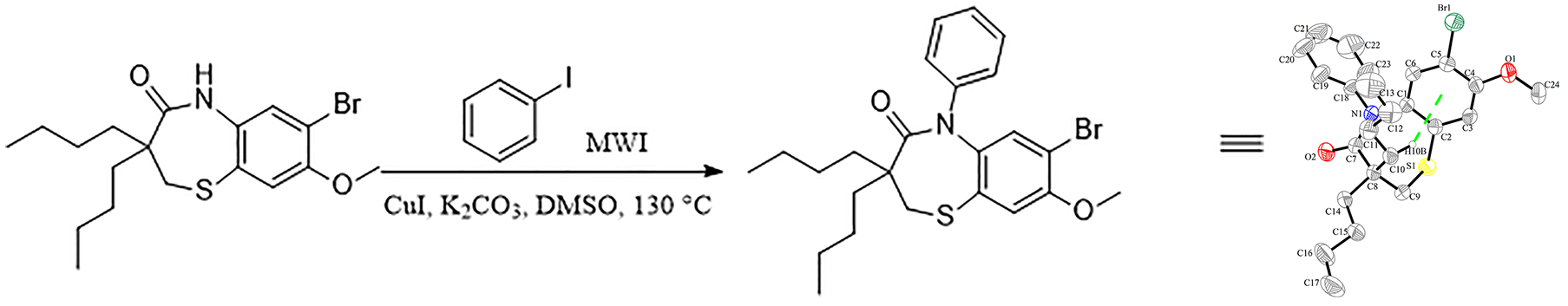
7-Bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one is a key intermediate of Elobixibat hydrate and is the main bioactive group. However, there are only a few studies reported that discuss the synthesis of the title compound in the literature.9,10 Herein, we report an efficient route to prepare the title compound. Compared with the original research patent, we have shortened the reaction time of the last step (from 17 h to 30 min) and reduced the reaction temperature (from 190 °C to 130 °C). In the aromatic amidation step,11–13 we have reduced the amount of amine ligand, which is more conducive to large-scale production (Scheme 1). This synthetic route afforded a 43% overall yield. Furthermore, a single crystal of the title compound was obtained and the molecular structure was determined by X-ray diffraction.
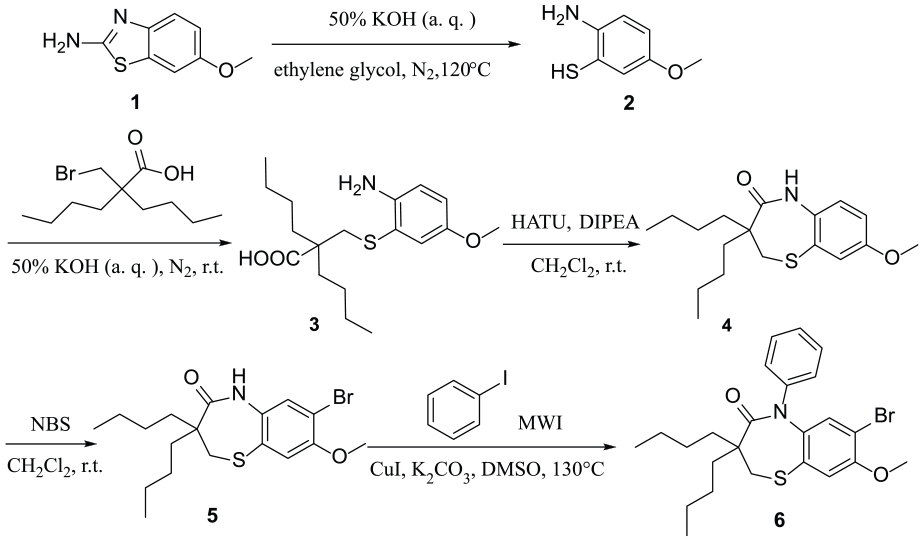
The synthetic route toward 7-bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one.
Results and discussion
In order to prepare the target product, we first investigated the original research patent.
9
When compounds
Optimization of the reaction conditions (ligand, temperature, mode of heating, and duration of reaction) for the preparation of the title compound.

MWI: microwave irradiation.
Meanwhile, we successfully grew a single crystal of product 6 from n-hexane, and the structure was confirmed by the X-ray crystal analysis (Figure 1, CCDC 1960860).

ORTEP diagram of compound
Compound
Conclusion
In summary, using 6-methoxybenzo[d]thiazol-2-amine as a starting material, 7-bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one was synthesized. By optimizing the original research route, the overall yield of this five-step sequence was 43%. At the same time, the reaction efficiency was greatly improved by using a microwave, and thus the use of expensive ligands was reduced and production costs were saved. A study of the crystal structure of the title compound may contribute evidence for further studies on drug designs and mechanisms.
Experimental
General information
Unless noted otherwise, all the reagents were commercially available and were used without further purification. The reactions were monitored by thin-layer chromatography (TLC). Microwave irradiation was performed in a MAS-1 microwave reactor apparatus (Shanghai Sineo Microwave Chemistry Technology Co., Ltd). Melting points were measured with an X4-A microscopic melting point apparatus. Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker BioSpin 400 MHz spectrometer with chemical shifts reported in ppm (in dimethyl sulfoxide-d6 (DMSO-d6), with tetramethylsilane (TMS) as the internal standard). High-resolution mass spectrometry (HRMS) was performed using a 6540 Ultra High Definition (UHD) Accurate-Mass Quadrupole Time-of-Flight (Q-TOF) liquid chromatography/mass spectrometry (LC/MS) instrument. X-ray single-crystal diffraction data were recorded on a XtaLAB Synergy, Dualflex, HyPix four-circle diffractometer.
Preparation of 2-{[(2-amino-5-methoxyphenyl)thio]methyl}-2-butylhexanoic acid (3 )
Under a nitrogen atmosphere, 6-methoxybenzo[d]thiazol-2-amine (
Preparation of 3,3-dibutyl-8-methoxy-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one (4 )
Compound
Preparation of 7-bromo-3,3-dibutyl-8-methoxy-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one (5 )
A mixture of compound
Preparation of 7-bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one (6 )
Compound
X-Ray structure determination
A colorless block-like crystal of compound
Crystallographic data for compound

Supplemental Material
Data – Supplemental material for Synthesis and crystal structure of 7-bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one
Supplemental material, Data for Synthesis and crystal structure of 7-bromo-3,3-dibutyl-8-methoxy-5-phenyl-2,3-dihydrobenzo[b][1,4]thiazepin-4(5H)-one by Damin Du and Haijian Wu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nurturing Project from Taizhou University (grant no. 2019PY016).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
