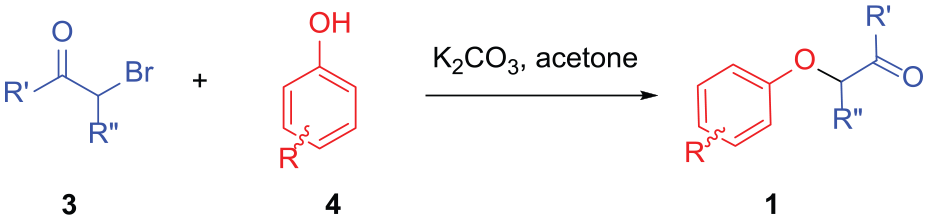
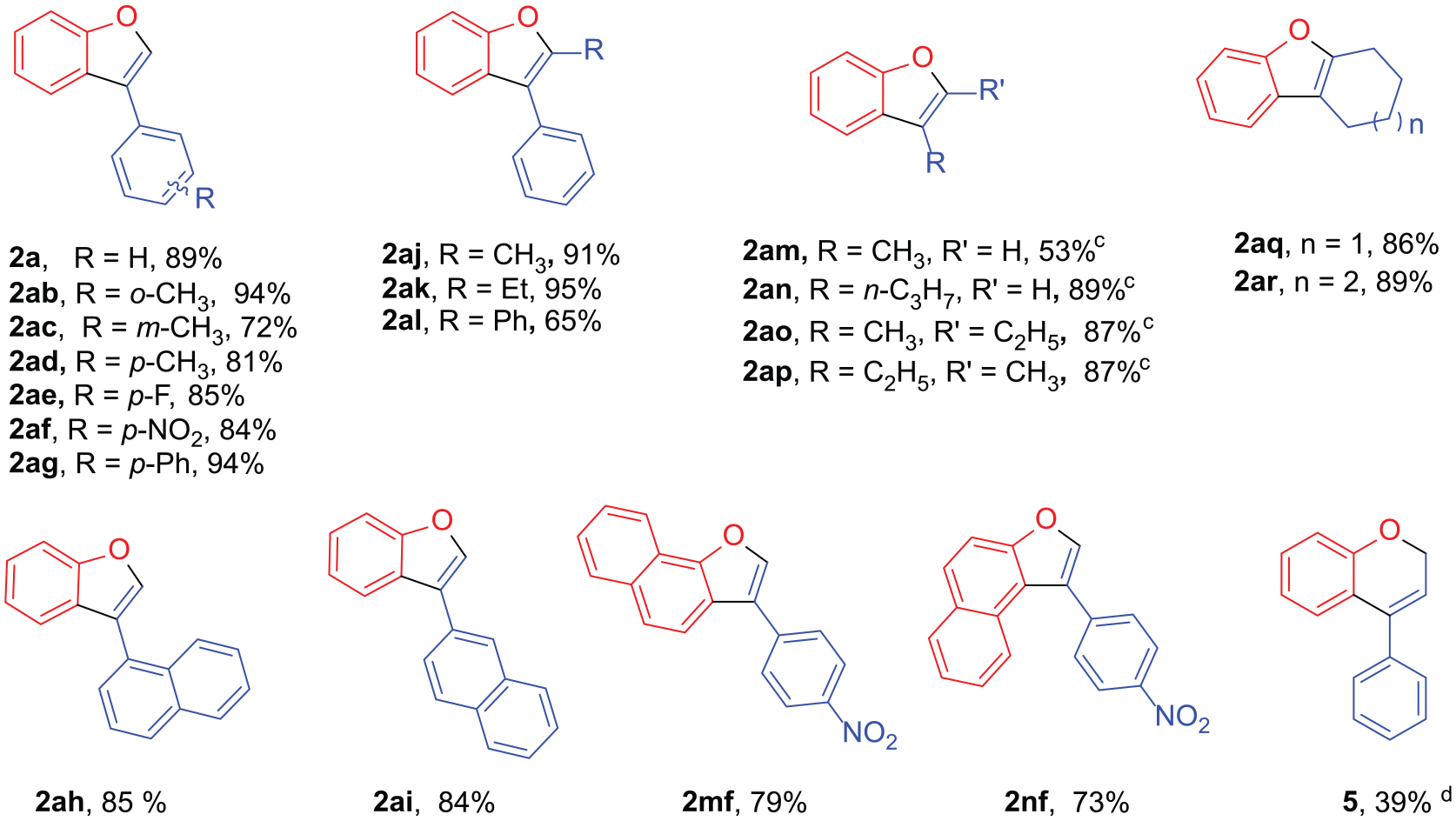
Typical procedure for the synthesis of benzofurans 2a–2q, 2ab–2ar, 2mf, 2nf, and benzopyran 5
To a 10-mL round-bottom flask containing 3 mL Eaton’s reagent in an oil or ethanol bath which was kept in the indicated reaction temperature, the α-phenoxy ketone (1 mmol) was added in one portion after being pretreated to the same temperature. The mixture was stirred at this temperature with TLC monitoring. After the starting material had been consumed, the resulting dark green mixture was poured into 100 mL of ice water and extracted with diethyl ether (3 × 15 mL). The combined organic layer was washed with a saturated solution of NaHCO3 (10 mL) and water (10 mL), then dried over anhydrous Na2SO4 and concentrated. The crude product was purified by flash chromatography to give benzofurans 2 or benzopyran 5. The reaction details are summarized in Table S1 of Supplemental Material.
3-Phenylbenzofuran (2a): Colorless oil. The NMR spectra are in good agreement with the literature.
62
1
H NMR (600 MHz, CDCl3): δ = 7.85 (dd, J = 7.7, 0.5 Hz, 1H), 7.80 (s, 1H), 7.68 − 7.63 (m, 2H), 7.56 (d, J = 8.1 Hz, 1H), 7.50 − 7.46 (m, 2H), 7.41 − 7.37 (m, 1H), 7.37 − 7.34 (m, 1H), 7.31 (td, J = 7.7, 1.0 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 156.0, 141.5, 132.2, 129.1, 127.7, 127.6, 126.6, 124.7, 123.2, 122.4, 120.5, 111.9. IR(KBr): υmax = 3033, 1605, 1574, 1489, 1451, 1217, 1107, 1092, 1012, 962, 856, 771, 763, 744, and 694 cm−1. MS (ESI): m/z = 195.07, (100) (M + H)+.
5-Methyl-3-phenylbenzofuran (2b): Pale yellow oil. The NMR spectra are in good agreement with the literature.
63
1
H NMR (600 MHz, CDCl3): δ = 7.73 (s, 1H), 7.64 − 7.60 (m, 3H), 7.48 − 7.43 (m, 2H), 7.41 (d, J = 8.4 Hz, 1H), 7.37 − 7.33 (m, 1H), 7.14 (dd, J = 8.4, 1.4 Hz, 1H), 2.46 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 154.4, 141.6, 132.6, 132.4, 129.1 127.6, 127.5, 126.7, 125.9, 122.2, 120.3, 111.4, 21.6. IR(KBr): υmax = 2921, 2360, 2341, 1467, 1230, 1188, 1107, 1090, 964, 903, 795, 762, 744, and 695 cm−1. MS (ESI): m/z = 209.10, (100) (M + H)+.
7-Methyl-3-phenylbenzofuran (2c): Colorless oil. The NMR spectra are in good agreement with the literature.
64
1
H NMR (600 MHz, CDCl3): δ = 7.85 (s, 1H), 7.74 (d, J = 7.7 Hz, 1H), 7.73 − 7.69 (m, 2H), 7.55 − 7.50 (m, 2H), 7.45 − 7.40 (m, 1H), 7.28 (t, J = 7.5 Hz, 1H), 7.22 (d, J = 7.2 Hz, 1H), 2.63 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.0, 141.2, 132.5, 129.1, 127.7, 127.5, 126.1, 125.6, 123.2, 122.6, 122.2, 118.0, 15.2. IR(KBr): υmax = 3054, 2923, 1608, 1496, 1446, 1416, 1379, 1350, 1223, 1177, 1147, 1116, 1074, 1022, 907, 843, 779, 759, 745, 696, and 549 cm−1. MS (ESI): m/z = 209.06, (100) (M + H)+.
6-Methyl-3-phenylbenzofuran (2d): Yellow solid, m.p. 39°C. 1H NMR (500 MHz, CDCl3): δ = 7.75 (s, 1H), 7.74 (d, J = 8.0 Hz, 1H), 7.69 − 7.65 (m, 2H), 7.49 (t, J = 7.7 Hz, 2H), 7.41 − 7.36 (m, 2H), 7.16 (d, J = 8.0 Hz, 1H), 2.52 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 156.4, 140.9, 135.0, 132.4, 129.1, 127.53, 127.48, 124.6, 124.1, 122.2, 120.0, 112.1, 21.8. IR(KBr): υmax = 3057, 3032, 2920, 2859, 1628, 1606, 1567, 1492, 1447, 1425, 1349, 1326, 1235, 1169, 1142, 1142, 1093, 1072, 1030, 965, 920, 912, 857, 798, 761, 745, 696, 673, and 612 cm−1. MS (ESI): m/z = 247.02, (100) [M + K]+. HRMS (ESI): m/z (M + H)+ calcd for C15H13O+: 209.09609; found: 209.09608.
4-Methyl-3-phenylbenzofuran (2d”): Colorless oil. 1H NMR (500 MHz, CDCl3): δ = 7.53 (s, 1H), 7.46 − 7.37 (m, 6H), 7.21 (dd, J = 8.8, 6.9 Hz, 1H), 6.99 (dd, J = 7.3, 0.8 Hz, 1H), 2.24 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.5, 142.1, 133.1, 132.2, 130.3, 128.2, 127.7, 126.0, 124.55, 124.51, 123.5, 109.4, 19.9. IR(KBr): υmax = 3054, 3028, 2955, 2923, 1605, 1568, 1494, 1444, 1413, 1295, 1223, 1164, 1141, 1106, 1071, 1025, 962, 913, 827, 778, 759, 747, and 700 cm−1. MS (ESI): m/z = 247.09, (100) [M + K]+. HRMS (ESI): m/z (M + H)+ calcd for C15H13O+: 209.09609; found: 209.09569.
5-Methoxy-3-phenylbenzofuran (2e): Colorless oil. The NMR spectra are in good agreement with the literature.36,65 1H NMR (600 MHz, CDCl3): δ = 7.79 (s, 1H), 7.67 (d, J = 7.3 Hz, 2H), 7.53 (t, J = 7.7 Hz, 2H), 7.49 (d, J = 8.9 Hz, 1H), 7.42 (t, J = 7.4 Hz, 1H), 7.33 (d, J = 2.6 Hz, 1H), 7.02 (dd, J = 8.9, 2.6 Hz, 1H), 3.90 (s, 3H). 13C NMR (126 MHz, CDCl3): δ = 156.3, 150.8, 142.3, 132.2, 129.1, 127.6, 127.1, 122.5, 113.4, 112.4, 102.8, 56.1. IR(KBr): υmax = 2929, 2831, 1607, 1560, 1492, 1471, 1451, 1440, 1272, 1259, 1195, 1173, 1136, 1107, 1026, 968, 905, 834, 797, 761, 753, 740, 696, 680, and 646 cm−1. MS (ESI): m/z = 225.03, (100) [M + H]+.
5-(tert-Butyl)-3-phenylbenzofuran (2f): Colorless oil. The NMR spectra are in good agreement with the literature.
64
1
H NMR (600 MHz, CDCl3): δ = 7.87 (d, J = 1.9 Hz, 1H), 7.79 (s, 1H), 7.71 − 7.68 (m, 2H), 7.55 − 7.52 (m, 3H), 7.47 (dd, J = 8.7, 1.9 Hz, 1H), 7.45 − 7.41 (m, 1H), 1.45 (s, 9H). 13C NMR (151 MHz, CDCl3): δ = 154.2, 146.3, 141.7, 132.5, 129.1, 127.7, 127.5, 126.2, 122.7, 122.6, 116.4, 111.2, 35.0, 32.1. IR(KBr): υmax = 3059, 2961, 2905, 2868, 1606, 1476, 1364, 1258, 1218, 1111, 1075, 970, 895, 879, 828, 804, 789, 763, 748, 724, 696, 664, and 508 cm−1. MS (ESI): m/z = 251.16, (100) (M + H)+.
3,5-Diphenylbenzofuran (2g): Colorless oil. 1H NMR (600 MHz, CDCl3): δ = 8.01 (d, J = 1.4 Hz, 1H), 7.83 (s, 1H), 7.70 − 7.67 (m, 2H), 7.65 − 7.63 (m, 2H), 7.62 (d, J = 8.5 Hz, 1H), 7.59 (dd, J = 8.5, 1.7 Hz, 1H), 7.50 (t, J = 7.7 Hz, 2H), 7.46 (t, J = 7.7 Hz, 2H), 7.40 (t, J = 7.4 Hz, 1H), 7.36 (t, J = 7.4 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 155.6, 142.1, 141.9, 137.0, 132.1, 129.2, 128.9, 127.8, 127.71, 127.70, 127.2, 127.1, 124.5, 122.7, 119.1, 112.0. IR(KBr): υmax = 3058, 3031, 2923, 1600, 1462, 1454, 1426, 1341, 1260, 1211, 1107, 1093, 1029, 965, 878, 807, 789, 754, 694, 656, 624, and 537 cm–1. MS (ESI): m/z = 271.27, (100) [M + H]+. HRMS (ESI): m/z (M + H)+ calcd for C20H15O+: 271.1117; found: 271.1119.
3,7-Diphenylbenzofuran (2h): White solid, m.p. 92°C–94°C (Lit
66
96°C–97°C) (PE/Acetone, 4:1, v/v). The NMR spectra are in good agreement with the literature.
66
1
H NMR (600 MHz, CDCl3): δ = 7.90 − 7.87 (m, 2H), 7.85 (s, 1H), 7.82 (dd, J = 7.8, 1.0 Hz, 1H), 7.70 − 7.66 (m, 2H), 7.55 − 7.48 (m, 5H), 7.44 − 7.38 (m, 3H). 13C NMR (151 MHz, CDCl3): δ = 153.2, 141.6, 136.6, 132.2, 129.1, 128.9, 128.8, 127.9, 127.8, 127.7, 127.4, 126.1, 124.4, 123.7, 122.6, 119.7. IR(KBr): υmax = 3052, 2923, 1605, 1572, 1491, 1444, 1408, 1344, 1218, 1205, 1156, 1122, 1085, 1022, 1073, 969, 877, 834, 798, 760, 746, 698, 644, 586, and 555 cm−1. MS (ESI): m/z = 271.15, (100) (M + H)+.
5-Fluoro-3-phenylbenzofuran (2i): Colorless oil. The NMR spectra are in good agreement with the literature.
36
1
H NMR (600 MHz, CDCl3): δ = 7.82 (s, 1H), 7.60 (dt, J = 7.9, 1.5 Hz, 2H), 7.52 − 7.45 (m, 4H), 7.42 − 7.37 (m, 1H), 7.08 (td, J = 9.0, 2.6 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 159.6 (d, J = 238.5 Hz), 152.1, 143.1, 131.7, 129.2, 127.8, 127.5, 127.4, 122.7 (d, J = 4.0 Hz), 112.6, 112.5 (d, J = 15.8 Hz), 106.2 (d, J = 25.5 Hz). 19F NMR (565 MHz, CDCl3): δ = −120.52. IR(KBr): υmax = 3058, 2925, 1746, 1625, 1595, 1493, 1466, 1450, 1338, 1270, 1250, 1164, 1127, 1104, 1087, 974, 916, 857, 800, 766, 756, 740, 696, 682, 639, 611, and 557 cm−1. MS (ESI): m/z = 212.88, (100) (M + H)+.
5-Chloro-3-phenylbenzofuran (2j): Colorless oil. The NMR spectra are in good agreement with the literature.
64
1
H NMR (600 MHz, CDCl3): δ = 7.80 (d, J = 2.5 Hz, 1H), 7.79 (s, 1H), 7.61 (t, J = 1.6 Hz, 1H), 7.60 (t, J = 1.4 Hz, 1H), 7.51 − 7.46 (m, 3H), 7.42 − 7.38 (m, 1H), 7.32 (dd, J = 8.7, 2.2 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 154.3, 142.7, 131.5, 129.2, 128.9, 128.1, 127.9, 127.6, 125.0, 122.3, 120.3, 112.9. IR(KBr): υmax = 3064, 2923, 2853, 1608, 1484, 1451, 1440, 1368, 1319, 1257, 1219, 1163, 1142, 1102, 1065, 1029, 1010, 968, 912, 866, 826, 801, 760, 742, 722, 690, 660, 644, and 547 cm−1. MS (ESI): m/z = 229.01, (100) (M + H)+.
5-Bromo-3-phenylbenzofuran (2k): Colorless oil. The NMR spectra are in good agreement with the literature.
64
1
H NMR (600 MHz, CDCl3): δ = 7.95 (d, J = 1.7 Hz, 1H), 7.78 (s, 1H), 7.60 (t, J = 1.5 Hz, 1H), 7.59 (d, J = 1.0 Hz, 1H), 7.51 − 7.47 (m, 2H), 7.45 (dd, J = 8.7, 1.8 Hz, 1H), 7.44 − 7.41 (m, 1H), 7.41 − 7.37 (m, 1H). 13C NMR (151 MHz, CDCl3): δ = 154.7, 142.5, 131.4, 129.2, 128.7, 128.0, 127.7, 127.6, 123.3, 122.1, 116.4, 113.4. IR(KBr): υmax = 3059, 2925, 1560, 1491, 1447, 1327, 1292, 1268, 1249, 1217, 1143, 1108, 1098, 1052, 1028, 965, 866, 798, 783, 759, 741, 712, 695, 648, and 539 cm−1. MS (ESI): m/z = 273.11, (100) (M + H)+.
3,7-Diphenylbenzo[1,2-b:4,5-b']difuran (2l): White solid, m.p. 170°C–172°C (PE/Acetone, 4:1, v/v). 1H NMR (600 MHz, CDCl3): δ = 7.64 (s, 2H), 7.56 (s, 2H), 7.10 − 7.05 (m, 4H), 6.99 − 6.95 (m, 2H), 6.87 (t, J = 7.6 Hz, 4H). 13C NMR (151 MHz, CDCl3): δ = 153.1, 142.8, 132.9, 128.5, 127.7, 127.1, 124.0, 119.7, 108.8. IR(KBr): υmax = 3128, 3027, 1605, 1557, 1488, 1446, 1416, 1358, 1261, 1228, 1204, 1151, 1127, 1109, 1072, 1033, 1016, 929, 843, 807, 787, 753, 725, 696, 678, 601, 592, 569, and 493 cm−1. MS (ESI): m/z = 311.21, (100) [M + H]+. HRMS (ESI): m/z (M + H)+ calcd for C22H15O2+: 311.10666; found: 311.10660.
3-Phenylnaphtho[1,2-b]furan (2m): White solid, m.p. 108°C–110°C (Lit
67
111°C–113°C) (PE/Acetone, 4:1, v/v). The NMR spectra are in good agreement with the literature.
67
1
H NMR (600 MHz, CDCl3): δ = 8.38 (d, J = 8.2 Hz, 1H), 7.98 (d, J = 8.2 Hz, 1H), 7.95 (s, 1H), 7.91 (d, J = 8.6 Hz, 1H), 7.76 − 7.70 (m, 3H), 7.66 − 7.62 (m, 1H), 7.54 (m, 3H), 7.42 (t, J = 7.4 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 151.6, 140.7, 132.4, 131.7, 129.2, 128.4, 127.8, 127.6, 126.6, 125.5, 123.8, 123.7, 122.1, 121.8, 120.3, 118.9. IR(KBr): υmax = 3053, 1603, 1561, 1518, 1444, 1386, 1246, 1218, 1127, 1068, 1026, 918, 867, 810, 790, 757, 742, 695, 631, and 565 cm−1. MS (ESI): m/z = 245.08, (100) (M + H)+.
1-Phenylnaphtho[2,1-b]furan (2n): Colorless oil. The NMR spectra are in good agreement with the literature.
68
1
H NMR (600 MHz, CDCl3): δ = 8.07 (d, J = 8.4 Hz, 1H), 7.99 (d, J = 8.1 Hz, 1H), 7.81 (d, J = 9.0 Hz, 1H), 7.77 − 7.72 (m, 2H), 7.66 (dt, J = 3.3, 1.9 Hz, 2H), 7.59 − 7.54 (m, 2H), 7.54 − 7.50 (m, 1H), 7.48 (ddd, J = 8.1, 7.0, 1.2 Hz, 1H), 7.41 (ddd, J = 8.2, 6.9, 1.3 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 153.3, 141.8, 133.3, 131.0, 130.0, 129.1, 128.7, 128.5, 128.0, 126.1, 126.1, 124.6, 124.5, 123.5, 120.9, 112.8. IR(KBr): υmax = 3051, 1524, 1488, 1443, 1384, 1294, 1251, 1224, 1110, 993, 947, 858, 803, 754, 700, 634, 611, 559, and 517 cm−1. MS (ESI): m/z = 245.07, (100) (M + H)+.
3-Phenyl-4H-furo[3,2-c]chromen-4-one (2o): White crystal, m.p. 162°C–164°C (Lit
69
177°C) (PE/EA, 4:1, v/v). The NMR spectra are in good agreement with the literature.
69
1
H NMR (600 MHz, CDCl3): δ = 7.92 (dd, J = 7.8, 1.5 Hz, 1H), 7.78 (s, 1H), 7.76 (d, J = 1.6 Hz, 2H), 7.57 − 7.51 (m, 1H), 7.46 (t, J = 7.6 Hz, 3H), 7.42 − 7.34 (m, 2H). 13C NMR (151 MHz, CDCl3): δ = 159.0, 158.0, 152.8, 141.4, 131.1, 129.2, 128.8, 128.7, 128.5, 126.9, 124.6, 121.1, 117.3, 113.0, 108.7. IR(KBr): υmax = 3150, 3052, 1740, 1629, 1594, 1543, 1497, 1487, 1449, 1414, 1366, 1317, 1286, 1249, 1213, 1155, 1101, 1072, 1045, 966, 927, 888, 793, 753, 722, 695, 632, 612, and 523 cm−1. MS (ESI): m/z = 263.06, (100) (M + H)+.
9-Phenyl-2H-furo[2,3-h]chromen-2-one (2p): Pale yellow needles, m.p. 112°C–114°C (PE/Acetone, 4:1, v/v). 1H NMR (500 MHz, CDCl3): δ = 7.80 (d, J = 9.6 Hz, 1H), 7.76 − 7.73 (m, 2H), 7.72 (d, J = 1.1 Hz, 1H), 7.53 − 7.48 (m, 2H), 7.47 (d, J = 8.5 Hz, 1H), 7.45 − 7.39 (m, 2H), 6.37 (d, J = 9.5 Hz, 1H). 13C NMR (126 MHz, CDCl3): δ = 160.3, 158.4, 149.4, 144.6, 142.9, 130.5, 129.3, 128.6, 128.3, 124.5, 123.1, 115.3, 114.4, 114.0, 109.2. IR(KBr): υmax = 3150, 3052, 1740, 1629, 1594, 1543, 1497, 1487, 1449, 1414, 1366, 1317, 1286, 1249, 1213, 1155, 1101, 1072, 1045, 999, 966, 927, 888, 793, 753, 722, 695, 632, 612, and 523 cm−1. MS (ESI): m/z = 285.04, (100) [M + Na]+. HRMS (ESI): m/z (M + H)+ calcd for C17H13O3+: 265.07918; found: 265.07675.
3-Phenylbenzo[b]thiophene (2q): Colorless oil. The NMR spectra are in good agreement with the literature.
70
1
H NMR (600 MHz, CDCl3): δ = 7.98 − 7.93 (m, 2H), 7.63 (t, J = 1.6 Hz, 1H), 7.61 (t, J = 1.5 Hz, 1H), 7.54 − 7.50 (m, 2H), 7.46 − 7.40 (m, 4H). 13C NMR (151 MHz, CDCl3): δ = 140.8, 138.2, 138.1, 136.2, 128.9(2C), 127.7, 124.6, 124.5, 123.5, 123.1(2C). IR(KBr): υmax = 3057, 3027, 1483, 1425, 1347, 833, 762, 731, 697, 636, 573, and 511 cm−1. MS (ESI): m/z = 211.06, (100) (M + H)+.
3-(o-Tolyl)benzofuran (2ab): Pale yellow liquid. The NMR spectra are in good agreement with the literature.
14
1
H NMR (500 MHz, CDCl3): δ = 7.63 (s, 1H), 7.56 (d, J = 8.2 Hz, 1H), 7.47 (d, J = 7.8 Hz, 1H), 7.40 (dd, J = 7.2, 1.5 Hz, 1H), 7.36 − 7.31 (m, 3H), 7.31 − 7.27 (m, 2H), 2.33 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.2, 142.3, 137.0, 131.0, 130.6, 130.5, 128.0, 126.0, 124.5, 122.9, 121.5, 120.8, 111.7, 20.6. IR(KBr): υmax = 3061, 2924, 2853, 1606, 1488, 1452, 1379, 1332, 1219, 1103, 1086, 1008, 962, 857, 774, 747, and 723 cm−1. MS (ESI): m/z = 209.14, (100) (M + H)+.
3-(m-Tolyl)benzofuran (2ac): Colorless oil. The NMR spectra are in good agreement with the literature.
64
1
H NMR (500 MHz, CDCl3): δ = 7.85 (d, J = 7.3 Hz, 1H), 7.78 (s, 1H), 7.55 (d, J = 8.0 Hz, 1H), 7.46 (d, J = 7.1 Hz, 2H), 7.36 (dt, J = 9.2, 4.6 Hz, 2H), 7.31 (td, J = 7.6, 0.9 Hz, 1H), 7.20 (d, J = 7.5 Hz, 1H), 2.44 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.9, 141.4, 138.8, 132.1, 129.0, 128.39, 128.35, 126.7, 124.8, 124.6, 123.1, 122.5, 120.6, 111.9, 21.7. IR(KBr): υmax = 3034, 2920, 1609, 1452, 1336, 1234, 1188, 1108, 1012, 857, 833, 783, 768, 744, 699, and 614 cm−1. MS (ESI): m/z = 209.11, (100) (M + H)+.
3-(p-Tolyl)benzofuran (2ad): Yellow oil. The NMR spectra are in good agreement with the literature.
14
1
H NMR (600 MHz, CDCl3): δ = 7.84 (dd, J = 7.7, 0.6 Hz, 1H), 7.77 (s, 1H), 7.58 − 7.53 (m, 3H), 7.38 − 7.33 (m, 1H), 7.32 − 7.29 (m, 3H), 2.43 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.9, 141.2, 137.4, 129.8, 129.2, 127.5, 126.8, 124.6, 123.0, 122.3, 120.6, 111.9, 21.4. IR(KBr): υmax = 3026, 2920, 1578, 1508, 1452, 1342, 1307, 1220, 1209, 1108, 1092, 1012, 964, 857, 824, 796, 769, 745, 565, 516, and 421 cm−1. MS (ESI): m/z = 209.32, (100) (M + H)+.
3-(4-Fluorophenyl)benzofuran (2ae): Yellow oil. The NMR spectra are in good agreement with the literature.
14
1
H NMR (600 MHz, CDCl3): δ = 7.81 − 7.77 (m, 1H), 7.76 (s, 1H), 7.63 − 7.58 (m, 2H), 7.57 − 7.55 (m, 1H), 7.37 (m, 1H), 7.34 − 7.30 (m, 1H), 7.20 − 7.15 (m, 2H). 13C NMR (151 MHz, CDCl3): δ = 162.4 (d, J = 246.6 Hz), 155.9, 141.3, 129.2 (d, J = 8.0 Hz), 128.2 (d, J = 3.3 Hz), 126.5, 124.8, 123.2, 121.5, 120.3, 116.1 (d, J = 21.5 Hz), 112.0. IR(KBr): υmax = 3056, 1817, 1597, 1570, 1505, 1452, 1341, 1298, 1218, 1159, 1109, 1090, 1013, 964, 932, 904, 857, 839, 813, 770, 745, 657, 578, 563, and 521 cm−1. 19F NMR (565 MHz, CDCl3): δ = −115.10. MS (ESI): m/z = 251.06, (100) (M + H)+.
3-(4-Nitrophenyl)benzofuran (2af): Yellow solid, m.p. 142° C–144° C (Lit
71
143° C) (CHCl3/PE, 4:1, v/v). The NMR spectra are in good agreement with the literature.
71
1
H NMR (500 MHz, CDCl3): δ = 8.35 (d, J = 8.9 Hz, 2H), 7.94 (s, 1H), 7.86 − 7.80 (m, 3H), 7.62 − 7.58 (m, 1H), 7.44 − 7.40 (m, 1H), 7.37 (td, J = 7.5, 1.2 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 156.1, 147.1, 143.0, 139.2, 127.9, 125.6, 125.4, 124.5, 123.8, 120.8, 120.2, 112.3. IR(KBr): υmax = 2959, 2924, 1599, 1565, 1508, 1449, 1343, 1316, 1259, 1215, 1185, 1137, 1111, 1091, 1013, 964, 852, 804, 761, 746, and 693 cm−1. HRMS (ESI): m/z (M + Na)+ calcd for C14H9NNaO3+: 262.0475; found: 262.0478.
3-((1,1'-Biphenyl) (2ag): White needles, m.p. 138° C–140° C (PE/EA, 4:1, v/v). The NMR spectra are in good agreement with the literature.
72
1
H NMR (500 MHz, CDCl3): δ = 7.92 − 7.88 (m, 1H), 7.85 (s, 1H), 7.76 − 7.70 (m, 4H), 7.69 − 7.64 (m, 2H), 7.58 (d, J = 7.7 Hz, 1H), 7.51 − 7.45 (m, 2H), 7.38 (td, J = 7.8, 1.4 Hz, 2H), 7.34 (td, J = 7.5, 1.2 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 156.0, 141.5, 140.8, 140.5, 131.2, 129.0, 128.0, 127.8, 127.6, 127.2, 126.6, 124.8, 123.2, 122.0, 120.6, 112.0. IR(KBr): υmax = 3033, 1606, 1569, 1487, 1451, 1347, 1253, 1218, 1167, 1146, 1121, 1095, 960, 929, 857, 838, 805, 767, 734, and 691 cm−1. MS (ESI): m/z = 271.15, (100) (M + H)+.
3-(Naphthalen-1-yl)benzofuran (2ah): Colorless liquid. The NMR spectra are in good agreement with the literature.
73
1
H NMR (500 MHz, CDCl3): δ = 7.98 (d, J = 8.4 Hz, 1H), 7.93 (t, J = 8.0 Hz, 2H), 7.82 (s, 1H), 7.63 − 7.56 (m, 3H), 7.55 − 7.50 (m, 1H), 7.47 − 7.41 (m, 2H), 7.38 (ddd, J = 8.4, 7.3, 1.2 Hz, 1H), 7.23 (dd, J = 7.2, 0.9 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 155.4, 142.9, 134.0, 132.3, 129.4, 128.6, 128.49, 128.48, 127.8, 126.3, 126.2, 126.1, 125.7, 124.7, 123.0, 121.1, 120.9, 111.8. IR(KBr): υmax = 3046, 2924, 2853, 1594, 1507, 1451, 1396, 1347, 1322, 1291, 1255, 1226, 1183, 1147, 1100, 1017, 936, 856, 798, 777, 767, 746, 670, 650, 615, and 583 cm−1. MS (ESI): m/z = 267.23, (100) (M + Na)+.
3-(Naphthalen-2-yl)benzofuran (2ai): White solid, m.p. 76° C–78° C (PE/EA, 4:1, v/v). 1H NMR (500 MHz, CDCl3): δ = 8.16 (d, J = 0.6 Hz, 1H), 8.02 − 7.95 (m, 2H), 7.95 − 7.88 (m, 3H), 7.77 (dd, J = 8.4, 1.7 Hz, 1H), 7.62 (m, 1H), 7.54 (pd, J = 6.9, 1.5 Hz, 2H), 7.41 (dd, J = 7.9, 1.5 Hz, 1H), 7.39 (dd, J = 7.5, 1.3 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 156.0, 141.8, 133.9, 132.8, 129.7, 128.8, 128.1, 127.9, 126.7, 126.6, 126.12, 126.07, 125.9, 124.8, 123.2, 122.4, 120.6, 112.0. IR(KBr): υmax = 3141, 3052, 1629, 1601, 1507, 1450, 1324, 1274, 1208, 1198, 1141, 1116, 1094, 1017, 981, 947, 932, 855, 836, 824, 804, 765, 745, 623, and 564 cm−1. MS (ESI): m/z = 267.19, (100) [M + Na]+. HRMS (ESI): m/z (M + H)+ calcd for C18H13O+: 245.09601; found: 245.09599.
2-Methyl-3-phenylbenzofuran (2aj): Colorless oil. The NMR spectra are in good agreement with the literature.
74
1
H NMR (600 MHz, CDCl3): δ = 7.58 (d, J = 7.9 Hz, 1H), 7.52 − 7.49 (m, 2H), 7.48 (d, J = 7.9 Hz, 1H), 7.46 (d, J = 7.9 Hz, 1H), 7.36 (s, 1H), 7.25 (d, J = 6.8 Hz, 1H), 7.24 − 7.20 (m, 1H), 2.54 (s, 3H). 13C NMR (151 MHz, CDCl3): δ = 154.2, 151. 5, 133.0, 129.1, 128.9, 127.1, 123.7, 122.7, 119.5, 117.0, 110.9, 13.0. IR(KBr): υmax = 3055, 2952, 2919, 2851, 1624, 1610, 1598, 1497, 1473, 1455, 1389, 1361, 1283, 1251, 1207, 1175, 1105, 1075, 1018, 1008, 963, 922, 904, 849, 769, 749, 700, 666, 653, 611, and 561 cm−1. MS (ESI): m/z = 209.09, (100) (M + H)+.
2-Ethyl-3-phenylbenzofuran (2ak): Colorless oil. The NMR spectra are in good agreement with the literature.
75
1
H NMR (600 MHz, CDCl3): δ = 7.63 (dd, J = 7.7, 0.6 Hz, 1H), 7.58 − 7.51 (m, 5H), 7.45 − 7.40 (m, 1H), 7.35 − 7.30 (m, 1H), 7.30 − 7.26 (m, 1H), 2.95 (q, J = 7.5 Hz, 2H), 1.42 (t, J = 7.6 Hz, 3H). 13C NMR (151 MHz, CDCl3): δ = 156.4, 154.2, 133.0, 129.2, 129.0, 128.9, 127.1, 123.7, 122.7, 119.6, 116.3, 111.0, 20.4, 13.1. IR(KBr): υmax = 3055, 2974, 2935, 2876, 1610, 1455, 1311, 1281, 1240, 1195, 1174, 1106, 1057, 1012, 980, 963, 898, 847, 769, 749, 700, and 611 cm−1. MS (ESI): m/z = 223.15, (100) (M + H)+.
2,3-Diphenylbenzofuran (2al): White solid, m.p. 120° C–122° C (Lit
76
121° C–123° C; Acetone/PE, 4:1, v/v). The NMR spectra are in good agreement with the literature.
76
1
H NMR (600 MHz, CDCl3): δ = 7.72 − 7.67 (m, 2H), 7.59 (d, 1H), 7.56 − 7.52 (m, 3H), 7.50 (td, J = 7.6, 1.7 Hz, 2H), 7.46 − 7.41 (m, 1H), 7.38 − 7.29 (m, 4H), 7.29 − 7.26 (m, 1H). 13C NMR (151 MHz, CDCl3): δ = 154.1, 150.7, 133.0, 130.8, 130.4, 129.9, 129.1, 128.6, 128.5, 127.8, 127.2, 124.8, 123.1, 120.2, 117.7, 111.3. IR(KBr): υmax = 3061, 1601, 1497, 1455, 1440, 1368, 1254, 1203, 1110, 1062, 1027, 1008, 960, 891, 827, 764, 748, 693, 621, and 608 cm−1. MS (ESI): m/z = 271.12, (100) (M + H)+.
3-Methylbenzofuran (2am): Colorless oil. The NMR spectra are in good agreement with the literature.
77
1
H NMR (500 MHz, CDCl3): δ = 7.55 − 7.51 (m, 1H), 7.48 − 7.44 (m, 1H), 7.41 (d, J = 1.3 Hz, 1H), 7.31 − 7.27 (m, 1H), 7.26 − 7.21 (m, 1H), 2.25 (d, J = 1.3 Hz, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.4, 141.5, 129.1, 124.2, 122.3, 119.5, 115.8, 111.4, 8.1. IR(KBr): υmax = 2959, 2929, 2871, 1453, 1185, 1088, and 743 cm−1. MS (ESI): m/z = 133.05, (100) (M + H)+.
3-Propylbenzofuran (2an): Colorless oil. 1H NMR (600 MHz, CDCl3): δ = 7.55 (d, J = 7.5 Hz, 1H), 7.46 (d, J = 8.1 Hz, 1H), 7.40 (s, 1H), 7.30 − 7.26 (m, 1H), 7.24 − 7.20 (m, 1H), 2.65 (t, J = 7.2 Hz, 2H), 1.74 (dq, J = 14.8, 7.3 Hz, 2H), 1.00 (t, J = 7.3 Hz, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.5, 141.2, 128.5, 124.1, 122.3, 120.6, 119.8, 111.5, 25.8, 22.4, 14.2. IR(KBr): υmax = 2959, 2929, 2871, 1453, 1219, 1185, 1089, 856, 772, 743 cm−1. MS (ESI) calcd for C11H13O+(M + H)+ 161.10; found: 161.05. MS (ESI): m/z = 161.05, (100) [M + H]+. HRMS (ESI): m/z (M + Na)+ calcd for C11H12NaO3+: 183.07817; found: 183.07820.
2-Ethyl-3-methylbenzofuran (2ao): Colorless oil. The NMR spectra are in good agreement with the literature.
78
1
H NMR (500 MHz, CDCl3): δ = 7.42 − 7.39 (m, 1H), 7.38 − 7.36 (m, 1H), 7.21 − 7.19 (m, 1H), 7.19 − 7.16 (m, 1H), 2.74 (q, J = 7.6 Hz, 2H), 2.15 (s, 3H), 1.28 (t, J = 7.6 Hz, 3H). 13C NMR (151 MHz, CDCl3): δ = 155.6, 153.9, 130.7, 123.1, 122.1, 118.8, 110.6, 108.9, 19.9, 12.9, 7.9. IR(KBr): υmax = 3036, 2973, 2937, 2876, 1631, 1474, 1455, 1315, 1276, 1238, 1179, 1133, 1107, 875, 828, and 743 cm−1. MS (ESI): m/z = 161.06, (100) (M + H)+.
3-Ethyl-2-methylbenzofuran (2ap): Colorless oil. The NMR spectra are in good agreement with the literature.
31
1
H NMR (500 MHz, CDCl3): δ = 7.52 − 7.48 (m, 1H), 7.42 (m, 1H), 7.25 − 7.20 (m, 2H), 2.67 (q, J = 7.6 Hz, 2H), 2.43 (s, 3H), 1.28 (t, J = 7.6 Hz, 3H). 13C NMR (151 MHz, CDCl3): δ = 154.1, 150.1, 129.7, 123.0, 122.0, 118.9, 116.1, 110.7, 17.1, 14.6, 12.0. IR(KBr): υmax = 3036, 2967, 2930, 2873, 1633, 1474, 1455, 1390, 1278, 1249, 1180, 1150, 1136, 1110, 1063, 1010, 952, 895, 848, and 743 cm−1. MS (ESI): m/z = 161.10, (100) (M + H)+.
1,2,3,4-Tetrahydrodibenzo[b, d]furan (2aq): Pale red oil. The NMR spectra are in good agreement with the literature.
79
1
H NMR (500 MHz, CDCl3): δ = 7.43 − 7.37 (m, 2H), 7.22 − 7.16 (m, 2H), 2.75 (tt, J = 6.3, 1.9 Hz, 2H), 2.63 (tt, J = 6.0, 1.9 Hz, 2H), 1.97 − 1.91 (m, 2H), 1.85 (tdd, J = 6.3, 4.6, 1.3 Hz, 2H). 13C NMR (151 MHz, CDCl3): δ = 154.4, 154.2, 129.0, 123.1, 122.2, 118.5, 113.0, 110.9, 23.6, 23.1, 22.8, 20.6. IR(KBr): υmax = 3054, 2932, 2845, 1639, 1476, 1453, 1362, 1297, 1274, 1257, 1222, 1189, 1121, 1008, 951, 875, 852, 820, and 742 cm−1. MS (ESI): m/z = 173.13, (100) (M + H)+.
7,8,9,10-Tetrahydro-6H-cyclohepta[b]benzofuran (2ar): Colorless oil. The NMR spectra are in good agreement with the literature.
80
1
H NMR (600 MHz, CDCl3): δ = 7.43 − 7.39 (m, 1H), 7.39 − 7.35 (m, 1H), 7.22 − 7.17 (m, 2H), 2.93 − 2.90 (m, 2H), 2.73 − 2.68 (m, 2H), 1.87 − 1.86 (m, 2H), 1.84 − 1.77 (m, 4H). 13C NMR (151 MHz, CDCl3): δ = 156.5, 153.4, 130.6, 122.9, 122.0, 118.4, 116.0, 110.6, 30.7 29.2, 28.4, 26.5, 23.3. IR(KBr): υmax = 3035, 2923, 2849, 1474, 1455, 1308, 1277, 1239, 1227, 1209, 1170, 1095, 1069, 1008, 814, 742 cm−1. MS (ESI): m/z = 187.11, (100) (M + H)+.
1-(4-Nitrophenyl)naphtho[2,1-b]furan (2mf): Yellow solid, m.p. 138° C–140° C (Lit
81
139° C–140° C) (CHCl3/PE, 4:1, v/v). The NMR spectra are in good agreement with the literature.
81
1
H NMR (600 MHz, CDCl3): δ = 8.42 − 8.35 (m, 2H), 7.98 (d, J = 8.1 Hz, 1H), 7.88 (d, J = 8.3 Hz, 1H), 7.82 (d, J = 9.0 Hz, 1H), 7.81 − 7.79 (m, 2H), 7.76 (s, 1H), 7.72 (d, J = 9.0 Hz, 1H), 7.52 − 7.45 (m, 1H), 7.41 (ddd, J = 8.2, 7.0, 1.2 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 153.7, 147.7, 142.4, 140.5, 131.1, 130.7, 129.4, 128.0, 126.9, 126.6, 124.9, 124.1, 123.1, 122.9, 120.0, 112.8. IR(KBr): υmax = 3071, 2925, 1600, 1515, 1380, 1342, 1250, 1226, 1115, 995, 948, 863, 850, 811, 795, 753, and 697 cm−1. HRMS (ESI): m/z (M + Na)+ calcd for C18H11NNaO3+: 312.0631; found 312.0637.
3-(4-Nitrophenyl)naphtho[1,2-b]furan (2nf): Yellow solid, m.p. 218° C–220° C (CHCl3/PE, 4:1, v/v). 1H NMR (600 MHz, CDCl3): δ = 8.37 (d, J = 8.7 Hz, 2H), 8.36 (d, J = 7.7 Hz, 1H),8.06 (s, 1H), 7.99 (d, J = 8.2 Hz, 1H), 7.87 (d, J = 8.7 Hz, 2H), 7.86 (d, J = 8.6 Hz, 1H), 7.79 (d, J = 8.6 Hz, 1H), 7.66 (t, J = 7.6 Hz, 1H), 7.57 (t, J = 7.5 Hz, 1H). 13C NMR (151 MHz, CDCl3): δ = 152.0, 147.1, 142.0, 139.4, 131.9, 128.5, 128.1, 127.0, 126.0, 124.61, 124.58, 122.1, 121. 7, 121.1, 120.3, 118.1. IR(KBr): υmax = 3133, 3062, 1597, 1571, 1508, 1453, 1379, 1340, 1312, 1294, 1278, 1251, 1221, 1154, 1120, 1106, 1065, 1026, 1009, 963, 919, 859, 851, 821, 811, 766, 755, 699, 687, 637, and 614 cm−1. HRMS (ESI): m/z (M + H)+ calcd for C18H12NO3+: 290.0812; found 290.0814.
4-Phenyl-2H-chromene (5): Pale yellow oil. The NMR spectra are in good agreement with the literature.
82
1
H NMR (500 MHz, CDCl3): δ = 7.43 − 7.32 (m, 5H), 7.18 − 7.13 (m, 1H), 7.01 (dd, J = 7.7, 1.6 Hz, 1H), 6.90 (dd, J = 8.1, 1.1 Hz, 1H), 6.85 (td, J = 7.5, 1.2 Hz, 1H), 5.80 (t, J = 3.9 Hz, 1H), 4.86 (d, J = 3.9 Hz, 2H). 13C NMR (151 MHz, CDCl3): δ = 154.9, 138.4, 137.3, 129.4, 128.8, 128.5, 127.9, 126.0, 123.8, 121.3, 120.1, 116.3, 65.4. IR(KBr): υmax = 3059, 2933, 1739, 1603, 1484, 1448, 1347, 1260, 1223, 1148, 1115, 1065, 1031, 1011, 928, 800, 758, and 700 cm−1. MS (ESI): m/z = 209.13, (100) (M + H)+.