The phosphoinositide 3-kinase (PI3K) is a very important node within the PI3K/AKT/mTOR pathway that regulates a variety of cellular processes including cell proliferation, differentiation, survival, and migration.
1
Aberrant activation of PI3K signaling has been implicated in multiple cancer types including colorectal, breast, leukemia, and so on.
2
Therefore, PI3K is considered as a promising target for solid and hematologic tumor therapy.
3
During the past 20 years, a great number of small molecule PI3K inhibitors have been investigated. Some early agents such as wortmannin and LY294002 did not enter clinical development due to their narrow therapeutic window and severe toxicity.
4
Fortunately, the selective second generation PI3K or PI3K/mTOR inhibitors, such as idelalisib (CAL-101, 1),
5
copanlisib (BAY80-6946, 2),
6
apitolisib (GDC-0980, 3),
7
omipalisib (GSK2126458, 4),
8
dactolisib (NVP-BEZ235, 5),
9
buparlisib (NVP-BKM120, 6),
10
and VS-5584 (7)
11
were advanced into clinical development, and some agents showed promising clinical efficacy (Figure 1). Particularly, idelalisib (selective PI3Kδ inhibitor) and copanlisib (pan-PI3K inhibitor) were recently approved by the Food and Drug Administration (FDA) for treatment of hematologic malignancy.5,6 These encouraging facts refueled the discovery and development of new PI3K inhibitors (Figure 1).
Recently, we reported our drug design and synthesis effort on the discovery of new potent PI3K inhibitors. The first strategy was to develop novel selective PI3Kδ inhibitors with distinct chemotypes which may elevate the therapeutic index and provide clinical benefit.12,13 For example, several 6-aryl substituted quinazoline derivatives were synthesized and some showed promising potency against PI3Kδ in vitro.14,15 The other strategy was to develop pan-PI3K or PI3K/mTOR inhibitors, exemplified by the preparation of several series of derivatives such as quinazolin-4(3H)-ones,16,17 [1,2,4]triazolo[1,5-a]pyridines,
18
benzo[d]thiazoles,19,20 4-morpholinoquinazolines,
21
and 4-(morpholin-4-yl)-1,3,5-triazines.
22
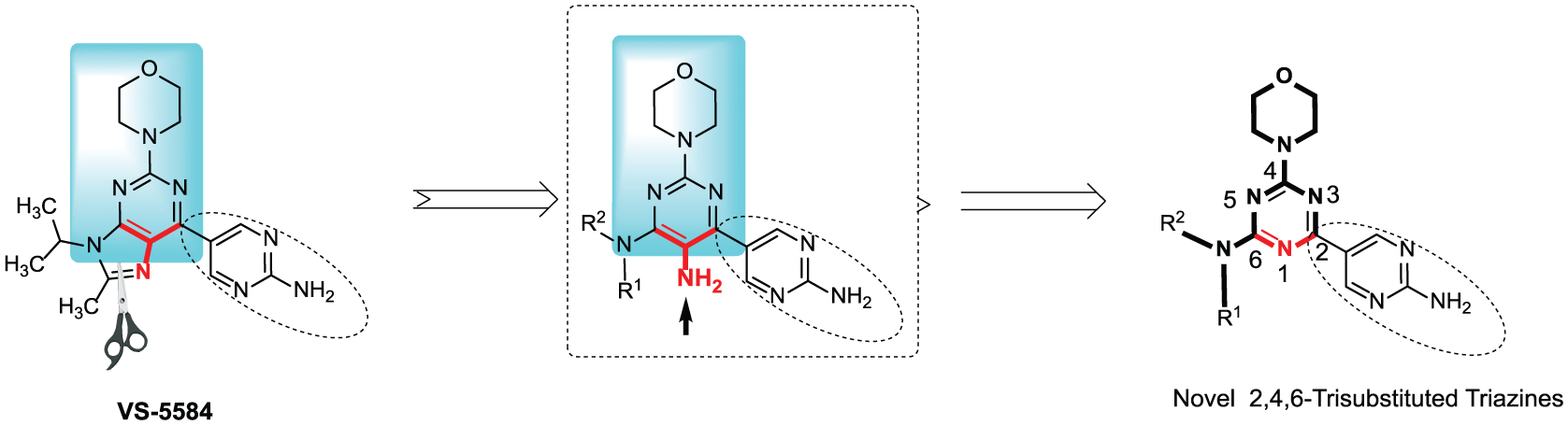
In particular, in the series of 4-(morpholin-4-yl)-1,3,5-triazines, representative derivatives displayed potent antitumor effects in vitro and in vivo. Thus, in an attempt to develop new anticancer agents and based on our previous reports and the chemical structure of VS-5584, we intend to open the purine ring of VS-5584 and replace it with a triazine core so as to design a series of 1,3,5-triazines as new PI3K inhibitors (indicted in Figure 2). Certainly, the 4-(morpholin-4-yl)-1,3,5-triazine is facile to be constructed and its derivatives were reported to show good antitumor effects. Furthermore, the 4-(morpholin-4-yl)-1,3,5-triazine scaffold was a good mimic of the main pharmacophore of VS-5584 which generated several key H-bond interactions with PI3Kα. Therefore, in this paper we design and synthesize a series of 4-(morpholin-4-yl)-1,3,5-triazines by introducing a variety of aliphatic amino groups at the 6-position as potential PI3K inhibitors, and investigate the primary structure–activity relationship (Figure 2).
Experimental
All starting materials, reagents, and solvents were commercially available, unless specified otherwise. Reactions were monitored by thin-layer chromatography on silica gel plates (GF-254) and visualized with UV light. The melting points were determined on a Shanghai micro melting-point apparatus (model: SGW® X-4B) and are uncorrected. NMR spectra were recorded on a 400 Bruker NMR spectrometer with tetramethylsilane (TMS) as an internal reference. Chemical shifts are reported in parts per million (ppm). Mass measurements were performed using electrospray ionization (positive mode) on a quadrupole time-of-flight (QTOF) mass spectrometer (Skyray Instrument).
General procedure for the synthesis of intermediates a1–a6
To a solution of 4-(4,6-dichloro-1,3,5-triazin-2-yl)morpholine (0.10 g, 0.43 mmol) in dry dichloromethane (10 mL) was added dropwise a mixture of the substituted aliphatic amine (0.43 mmol) and triethylamine (178 μL) at 0 °C. The reaction mixture was stirred at room temperature until all the starting materials have been consumed. The solvent was removed under reduced pressure. Water (30 mL) was added to the residue and the mixture was stirred at room temperature for 10 min. The mixture was extracted with ethyl acetate (15 mL × 3). The organic extracts were combined and washed with water. After drying with anhydrous Na2SO4, the organic extracts were evaporated to provide compounds a1–a6.
4-Chloro-N-ethyl-N-isopropyl-6-morpholino-1,3,5-triazin-2-amine (a1):
23
colorless oil, yield 88%. ESI-MS m/z: 286.2 [M+ H]+.
4-Chloro-N,N-diisopropyl-6-morpholino-1,3,5-triazin-2-amine (a2):
23
Colorless oil, yield 83%. ESI-MS m/z: 300.2 [M + H]+.
4-(4-Chloro-6-(4-methylpiperazin-1-yl)-1,3,5-triazin-2-yl)morpholine (a3):
23
colorless oil, yield 80%. ESI-MS m/z: 299.2 [M + H]+.
(1-(4-Chloro-6-morpholino-1,3,5-triazin-2-yl)piperidin-4-yl)(pyrrolidin-1-yl)methanone (a4): colorless oil, yield 71%. 1H NMR (CDCl3) δ 4.75–4.70 (m, 2H), 3.73–3.70 (m, 8H), 3.58–3.40 (m, 4H), 2.95–2.90 (m, 2H), 2.61 (d, J = 5.4 Hz, 1H), 1.98 (d, J = 6.1 Hz, 2H), 1.88–1.78 (m, 6H). ESI-MS m/z: 381.2 [M + H]+.
(1-(4-Chloro-6-morpholino-1,3,5-triazin-2-yl)piperidin-4-yl)(piperidin-1-yl)methanone (a5): colorless oil, yield 60%. 1H NMR (CDCl3) δ 4.72–4.68 (m, 2H), 3.62–3.56 (m, 8H), 3.50–3.41 (m, 4H), 2.99–2.81 (m, 2H), 2.69 (d, J = 5.9 Hz, 1H), 1.68–1.55 (m, 6H), 1.50–1.45 (m, 4H). ESI-MS m/z: 395.2 [M + H]+.
(1-(4-Chloro-6-morpholino-1,3,5-triazin-2-yl)piperidin-4-yl)(morpholino) methanone (a6): colorless oil, yield 65%. 1H NMR (CDCl3) δ 4.62–4.52 (m, 2H), 3.62–3.56 (m, 8H), 3.57–3.55 (m, 3H), 3.52–3.38 (m, 4H), 2.93–2.76 (m, 2H), 2.70–2.63 (m, 2H), 1.66–1.60 (m, 4H). ESI-MS m/z: 397.2 [M + H]+.
General procedure for the synthesis of compound A1–A6
The mixture of 5-bromopyrimidin-2-amine (65 mg, 0.37 mmol), bis(pinacolato)diboron (113 mg, 0.44 mmol), potassium acetate (109 mg, 1.11 mmol), PdCl2(dppf) (21 mg, 0.02 mmol), and 1,4-dioxane (10 mL) was heated under reflux under Ar atmosphere for 2.5 h, then concentrated under reduced pressure. To the resulting residue was added b1 (98 mg, 0.25 mmol), K2CO3 (154 mg, 1.11 mmol), PdCl2(dppf) (21 mg, 0.02 mmol), 1,4-dioxane (10 mL), and water (4 mL). The mixture obtained was heated under reflux in Ar atmosphere for 4 h. The volatile material was removed under reduced pressure and the residue was purified by a column chromatography on silica gel with CH2Cl2 / MeOH (v: v = 50:1) as eluent to produce A1 as an off-white solid.
4-(2-Aminopyrimidin-5-yl)-N-ethyl-N-isopropyl-6-morpholino-1,3,5-triazin-2-amine (A1): off-white solid, yield 86%. mp 216–218 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.41 (s, 2H), 5.22–4.86 (m, 1H), 3.90–3.85 (m, 4H), 3.80–3.68 (m, 4H), 3.60–3.39 (m, 2H), 1.22 (d, J = 5.2 Hz, 9H). 13C NMR (DMSO-d6): δ 165.2, 164.7 (2C), 164.1, 158.9 (2C), 119.4, 66.5 (2C), 46.0, 45.9, 43.7 (2C), 20.7, 20.5, 15.0. ESI-MS m/z: 345.2 [M + H]+.
4-(2-Aminopyrimidin-5-yl)-N,N-diisopropyl-6-morpholino-1,3,5-triazin-2-amine (A2): off-white solid, yield 76%. mp 224–226 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.40 (s, 2H), 4.45–4.40 (m, 2H), 3.90–3.85 (m, 4H), 3.79–3.72 (m, 4H), 1.37–1.30 (m, 12H). 13C NMR (DMSO-d6): δ 166.9, 165.2, 164.4, 164.1, 158.8 (2C), 119.5, 66.5 (2C), 45.4 (2C), 43.80 (2C), 20.9 (2C), 20.6 (2C). ESI-MS m/z: 359.8 [M + H]+.
5-(4-(4-Methylpiperazin-1-yl)-6-morpholino-1,3,5-triazin-2-yl)pyrimidin-2-amine (A3): off-white solid, yield 86%. mp >250 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 5.36 (s, 2H), 3.87 (s, 8H), 3.77–3.73 (m, 4H), 2.54–2.42 (m, 4H), 2.36 (s, 3H). 13C NMR (DMSO-d6) δ 167.7, 165.3, 164.7, 164.3, 159.0 (2C), 119.1, 66.5 (2C), 54.9 (2C), 46.2 (2C), 43.7 (2C), 43.1. ESI-MS m/z: 357.2 [M + H]+.
(1-(4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)piperidin-4-yl)(pyrrolidin-1-yl)methanone (A4): off-white solid, yield 87%. mp >250 °C. 1H NMR (CDCl3) δ 9.22 (s, 2H), 5.53 (s, 2H), 4.90–4.75 (m, 2H), 3.87–3.75 (m, 8H), 3.58–3.45 (m, 4H), 2.95–2.90 (m, 2H), 2.67–2.57 (m, 1H), 2.03–1.92 (m, 2H), 1.88–1.72 (m, 6H). 13C NMR (DMSO-d6) δ 172.6, 167.7, 165.3, 164.7, 164.4, 159.0 (2C), 119.1, 66.5 (2C), 63.3, 54.0 (2C), 46.2 (2C), 45.9 (2C), 26.1, 24.3, 18.5, 17.2. ESI-MS m/z: 440.2 [M + H]+.
(1-(4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)piperidin-4-yl)(piperidin-1-yl)methanone (A5): white solid, yield 66%. mp >250 °C. 1H NMR (DMSO-d6) δ 9.03 (s, 2H), 7.25 (s, 2H), 4.90–4.74 (m, 2H), 3.72–3.68 (m, 8H,), 3.48–3.38 (m, 5H), 2.94–2.82 (m, 3H), 1.67–1.58 (m, 4H), 1.48–1.42 (m, 6H). 13C NMR (DMSO-d6) δ 172.4, 167.7, 165.2, 164.8, 164.4, 159.0 (2C), 119.4, 66.5 (2C), 55.4 (2C), 46.2 (2C), 42.5 (2C), 37.64, 28.7, 27.0, 25.8, 24.6 (2C). ESI-MS m/z: 454.2 [M + H]+.
(1-(4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)piperidin-4-yl)(morpholino)methanone (A6): white solid, yield 76%. mp >250 °C. 1H NMR (DMSO-d6) δ 9.04 (s, 2H), 7.25 (s, 2H), 4.90–4.74 (m, 2H), 3.90–3.62 (m, 8H), 3.57–3.50 (m, 6H), 3.49–3.39 (m, 2H), 3.01–2.91 (m, 3H), 1.75–1.62 (m, 2H), 1.50–1.42 (m, 2H). 13C NMR (DMSO-d6) δ 172.9, 167.7, 165.2, 164.5, 164.4, 159.0 (2C), 119.1, 66.8, 66.7, 66.5 (2C), 56.5 (2C), 45.9 (2C), 42.0 (2C), 37.5, 28.5, 19.1. ESI-MS m/z: 456.2 [M + H]+.
Tert-butyl 4-((4-chloro-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino) piperidine-1-carboxylate (b1): To a solution of 4-(4,6-dichloro-1,3,5-triazin-2-yl)morpholine (4.00 g,17.0 mmol) in dry dichloromethane (20 mL) was added dropwise the mixture of tert-butyl 4-(methylamino)piperidine-1-carboxylate (3.65 g, 17.0 mmol) and triethylamine (4.71 mL) at 0 °C. The reaction mixture was stirred at room temperature until all starting materials consumed. The solvent was removed under reduced pressure. To the residue was added ethanol (4 mL) and water (30 mL), and the mixture was stirred at room temperature for 10 min. The solid was filtered, washed with water (5 mL× 3), and dried to afford compound b1 (4.32 g) with a yield of 62%. mp 117–119 °C. 1H NMR (CDCl3) δ 4.11–3.80 (m, 8H), 3.76 (s, CH3), 3.10–2.82 (m, 5H), 1.75–1.64 (m, 4H), 1.48 (s, 9H). ESI-MS m/z: 413.2 [M + H]+.
Tert-Butyl 4-((4-chloro-6-morpholino-1,3,5-triazin-2-yl)(cyclopropyl) amino) piperidine-1-carboxylate (b2): To a solution of 4-(4,6-dichloro-1,3,5-triazin-2-yl)morpholine (1.76 g, 7.52 mmol) in dry tetrahydrofuran (20 mL) were added the mixture of tert-butyl 4-(cyclopropylamino)piperidine-1-carboxylate (2.16 g, 9.02 mmol), 4-methy lbenzenesulfonic acid (0.20 g, 1.16 mmol), and N,N-diisopropylethylamine (3.95 mL). The reaction mixture was heated at reflux for 4 h. The solvent was removed under reduced pressure. The residue was suspended in water (50 mL) and extracted with dichloromethane (20 mL × 3). The organic layer was washed by saturated sodium chloride, dried, filtered, and concentrated to afford compound b2 (1.30 g). Yield: 71%. 1H NMR (CDCl3) δ 4.34–4.00 (m, 2H), 3.85–3.59 (m, 8H), 2.72–2.37 (m, 4H), 2.04–1.72 (s, 2H), 1.43 (s, 9H), 0.86–0.80 (m, 2H), 0.75–0.63 (m, 2H). ESI-MS m/z: 439.2 [M + H]+.
General procedure for the synthesis of compound B1 and B2
The mixture of 5-bromopyrimidin-2-amine (242 mg, 1.38 mmol), bis(pinacolato)diboron (386 mg, 1.52 mmol), KOAc (541 mg, 4.14 mmol), PdCl2(dppf) (76 mg, 0.10 mmol), and 1,4-dioxane (20 mL) was heated at reflux under Ar atmosphere for 2.5 h, then concentrated in vacuum. To the resulted residue was added b1 (400 mg, 0.97 mmol), K2CO3 (116 mg, 0.84 mmol), PdCl2(dppf) (76 mg, 0.10 mmol), 1,4-dioxane (20 mL), and water (4 mL). The obtained mixture was heated at reflux under Ar atmosphere for 4 h. The volatile was removed under vacuum and the residue was purified through a column chromatography on silica gel with CH2Cl2 / MeOH (v:v = 50:1) as eluent to produce B1 or B2.
Tert-Butyl-4-((4-(2-aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl) (methyl)amino) piperidine-1-carboxylate (B1): white solid, yield 74%. mp 227–228 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 5.45 (s, 2H), 4.11–3.80 (m, 8H), 3.77 (s, CH3), 3.10–2.82 (m, 5H), 1.75–1.64 (m, 4H), 1.48 (s, 9H). 13C NMR (CDCl3) δ 164.8, 164.1, 159.1 (3C), 154.7, 121.3, 79.7, 66.9 (2C), 52.3 (2C), 43.6 (2C), 28.3 (7C). ESI-MS m/z: 472.3 [M + H]+.
Tert-Butyl-4-((4-(2-aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl) (cyclopropyl)amino)piperidine-1-carboxylate (B2): off-white solid, yield 45%. mp 222–225 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.48 (s, 2H), 4.34–4.00 (m, 2H), 3.85–3.59 (m, 8H), 2.72–2.37 (m, 4H), 2.04–1.72 (s, 2H), 1.43 (s, 9H), 0.86–0.80 (m, 2H), 0.75–0.63 (m, 2H). 13C NMR (CDCl3) δ 167.1, 167.0, 164.6, 164.2, 159.1 (2C), 154.8, 121.4, 79.6, 66.8 (2C), 57.2, 43.6, 30.4, 28.5 (7C), 26.8, 9.2 (2C). ESI-MS m/z: 498.3 [M + H]+.
General procedure for the synthesis of compound B3 and B4
To a solution of B1 or B2 (300 mg, 0.63 mmol) in dichloromethane (10 mL), TFA (2 mL) was added. The resulting solution was stirred at room temperature for 2 h, concentrated in vacuum. The residue was suspended in saturated sodium carbonate solution (5.0 mL) and extracted with dichloromethane (20 mL × 3). The organic layer was washed by saturated sodium chloride, dried, filtered, and concentrated to afford compound B3 or B4 as a white solid.
4-(2-Aminopyrimidin-5-yl)-N-methyl-6-morpholino-N-(piperidin-4-yl)-1,3,5-triazin-2-amine (B3): white solid, yield 65%. mp >250 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.38 (s, 2H), 4.00–3.75 (m, 8H), 4.87–4.65 (m, 1H), 3.71 (s, CH3), 3.25–3.03 (m, 5H), 2.80–2.60 (m, 2H), 2.00–1.71 (m, 4H). 13C NMR (CDCl3) δ 164.8 (2C), 164.0, 159.2 (3C), 121.5, 66.9 (2C), 52.4, 46.4 (3C), 43.6, 30.4, 30.2, 28.3. ESI-MS m/z: 372.2 [M + H]+.
4-(2-Aminopyrimidin-5-yl)-N-cyclopropyl-6-morpholino-N-(piperidin-4-yl)-1,3,5-triazin-2-amine (B4): white solid. yield: 94%. mp: >250 °C. 1H NMR (DMSO-d6) δ 9.09 (s, 2H), 7.26 (s, 2H), 4.40–4.35 (m, 1H), 3.81–3.66 (m, 8H), 3.40–3.25 (m, 2H), 3.10–2.95 (m, 2H), 2.63–2.50 (m, 1H), 2.40–2.25 (m, 2H), 2.00–1.90 (m, 2H), 0.92–0.86 (m, 2H), 0.75–0.63 (m, 2H). 13C NMR (DMSO-d6): δ 167.3, 166.9, 165.2, 164.4, 159.2 (2C), 66.52 (2C), 54.7, 43.7 (4C), 27.9, 27.4 (2C), 9.4 (2C). ESI-MS m/z: 398.3 [M + H]+.
4-(2-Aminopyrimidin-5-yl)-N-(1-(cyclopropylmethyl)piperidin-4-yl)-N-methyl-6-morpholino-1,3,5-triazin-2-amine (B5): To a solution of B3 (50 mg, 0.14 mmol), cyclopropylformaldehyde (10 mg, 11 μL), NaBH(OAc)3 (45 mg, 0.22 mmol), and acetic acid (10 μL) were added. The resulted suspension was stirred overnight, quenched with aqueous sodium hydrogen carbonate and extracted with dichloromethane (10 mL×2). The combined organic phase was washed with brine, and dried over Na2SO4, filtered, concentrated in vacuum. The residue was purified by column chromatography on silica gel to produce B5 (10 mg). Yield 17%. mp >250 °C. 1H NMR (CDCl3) δ 9.17 (s, 2H), 5.50 (s, 2H), 5.09–5.03 (m, 1H), 3.80–3.60 (m, 8H), 3.20–3.04 (m, 3H), 2.90–2.75 (m, 6H), 1.90–1.84 (m, 6H), 1.33–1.20 (m, 1H), 0.82–0.75 (m, 2H), 0.50–0.40 (m, 2H). ESI-MS m/z: 426.3 [M + H]+.
General procedure for the synthesis of compounds B6 and B7
The mixture containing compound B3 (50 mg, 0.14 mmol), benzyl bromide (24 mg, 0.14 mmol), potassium carbonate (58 mg), and dimethylformamide (DMF) (10 mL) was stirred at room temperature overnight, concentrated in vacuum. The residue was purified by column chromatography on silica gel to produce B6 or B7.
4-(2-Aminopyrimidin-5-yl)-N-(1-benzylpiperidin-4-yl)-N-methyl-6-morpholino-1,3,5-triazin-2-amine (B6): white solid, yield 89%. mp 232–234 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 7.35–7.26 (m, 5H), 5.33 (s, 2H), 4.68–4.49 (m, 1H), 3.98–3.71 (m, 8H), 3.56 (s, 2H), 3.06–3.01 (m, 5H), 2.13–2.00 (m, 2H), 1.95–1.75 (m, 2H), 1.70–1.65 (m, 2H). 13C NMR (CDCl3) δ 164.8, 164.0, 159.2 (2C), 129.2 (2C), 128.3 (4C), 127.1 (2C), 121.5, 66.9 (2C), 63.2 (2C), 53.32 (2C), 52.4, 43.6, 29.1, 28.9, 28.3. ESI-MS m/z: 462.3 [M + H]+.
4-(2-Aminopyrimidin-5-yl)-N-methyl-6-morpholino-N-(1-(4-nitrobenzyl) piperidin-4-yl)-1,3,5-triazin-2-amine (B7): white solid, yield 87%. mp >250 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 8.20 (d, J = 8.3 Hz, 2H), 7.53 (d, J = 7.6 Hz, 2H), 5.38 (s, 2H), 4.80–4.69 (m, 1H), 3.97–3.73 (m, 8H), 3.62 (s, 2H), 3.09–2.95 (m, 5H), 2.19–2.04 (m, 2H), 1.87–1.72 (m, 2H), 1.69–1.60 (m, 2H). 13C NMR (CDCl3): δ 164.9, 164.0, 159.2 (2C), 147.1, 146.7, 129.5, 123.6 (4C), 121.9, 121.5, 66.9 (2C), 62.2 (2C), 53.4 (2C), 43.6 (2C), 29.2, 28.8, 28.3. ESI-MS m/z: 507.3 [M + H]+.
General procedure for the synthesis of compound B8 and B10
The mixture containing B3 or B4 (50 mg, 0.14 mmol), cyclopropylsulfonyl chloride (16 mg, 0.14 mmol), 4-dimethylaminopyridine (DMAP) (16 mg, 0.07 mmol), triethylamine (60 μL), and dichloromethane (15 mL) was stirred at room temperature for 2 h, concentrated in vacuum. The residue was purified by column chromatography on silica gel to produce B8 or B10.
4-(2-Aminopyrimidin-5-yl)-N-(1-(cyclopropylsulfonyl)piperidin-4-yl)-N-methyl-6-morpholino-1,3,5-triazin-2-amine (B8): white solid, yield 65%. mp >250 °C. 1H NMR (CDCl3) δ 9.20 (s, 2H), 5.47 (s, 2H), 4.89–4.79 (m, 1H), 3.92–3.69 (m, 8H), 3.09–3.05 (m, 3H), 2.97–2.95 (m, 2H), 2.37–2.26 (m, 2H), 1.99–1.86 (m, 3H), 1.81–1.69 (m, 2H), 1.25–1.16 (m, 2H), 1.10–1.00 (m, 2H). 13C NMR (DMSO-d6) δ 167.5, 165.2, 164.8, 164.6, 159.0 (2C), 119.1, 66.51 (2C), 46.0 (4C), 43.6, 28.7, 28.5, 26.1, 26.0, 4.53 (2C). ESI-MS m/z: 498.2 [M + Na]+.
4-(2-Aminopyrimidin-5-yl)-N-cyclopropyl-N-(1-(cyclopropylsulfonyl) piperidin-4-yl)-6-morpholino-1,3,5-triazin-2-amine (B10): white solid, yield 62%. mp 240–243 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.52 (s, 2H), 4.41–4.38 (m, 1H), 3.95–3.75 (m, 9H), 2.90–2.85 (m, 2H), 2.50 (s, 1H), 2.28–2.19 (m, 4H), 1.95–1.90 (m, 2H), 1.22–1.16 (m, 2H), 1.07–0.99 (m, 2H), 0.95–0.90 (m, 2H), 0.81–0.70 (m, 2H). 13C NMR (CDCl3) δ 167.1 (2C), 164.6, 164.0, 159.1 (2C), 121.2, 66.8 (2C), 58.4, 56.4, 46.6 (2C), 43.6, 30.2 (2C), 26.0, 18.4, 9.2 (2C), 4.4 (2C). ESI-MS m/z: 502.3 [M + H]+.
4-(2-Aminopyrimidin-5-yl)-N-methyl-6-morpholino-N-(1-tosylpiperidin-4-yl)-1,3,5-triazin-2-amine (B9): To a solution of 4-methylbenzenesulfonyl chloride (26 mg, 0.14 mmol) in dry dichloromethane (10 mL) was added dropwise the mixture of B3 (50 mg, 0.14 mmol), and triethylamine (4.71 mL) at 0 °C. The reaction mixture was stirred at room temperature until all starting materials consumed. The solvent was removed under reduced pressure. The residue was purified by column chromatography on silica gel with CH2Cl2 / MeOH (v: v = 50: 1) as eluent to produce B9 as a white solid. Yield 74%. mp >250 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 7.34 (d, J = 7.6 Hz, 2H), 7.22 (d, J = 7.9 Hz, 2H), 5.43 (s, 2H), 4.93–4.80 (m, 1H), 3.99–3.74 (m, 10H), 3.07–3.00 (m, 5H), 2.88–2.80 (m, 2H), 2.38 (s, 3H), 1.83–1.75 (m, 4H). 13C NMR (DMS-d6): δ 164.8, 164.6, 159.0, 143.9, 133.8, 130.4 (3C), 127.9 (4C), 119.1, 66.48 (2C), 56.50 (3C), 46.03 (2C), 43.6, 21.5, 19.0 (2C). ESI-MS m/z: 548.3 [M + Na]+.
General procedure for the synthesis of compound B11–
B19
A mixture of B3 or B4 (50 mg, 0.14 mmol), substituted carboxylic acid (0.14 mmol), HATU (54 mg, 0.14 mmol), N, N-diisopropylethylamine (0.42 mmol) and dichloromethane (15 mL) was stirred at room temperature for 2 h, then concentrated in vacuum. The residue was purified by column chromatography on silica with CH2Cl2 / MeOH (v: v = 50:1) as eluent to produce B11–B19 as a white solid.
1-(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino)piperidin-1-yl)ethan-1-one (B11): white solid, yield 56%. mp >250 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 5.51 (s, 2H), 5.10–4.70 (m, 2H), 4.01–3.67 (m, 9H), 3.20–3.04 (m, 3H), 2.63–2.58 (m, 2H), 2.14 (s, 3H), 1.84–1.62 (m, 4H). 13C NMR (DMSO-d6) δ 168.5, 165.3 (2C), 164.7, 163.8, 159.0 (2C), 119.2, 66.5 (2C), 52.4, 49.1 (4C), 45.8, 43.7, 21.71 (2C). ESI-MS m/z: 414.3 [M + H]+.
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino)piperidin-1-yl)(cyclopropyl)methanone (B12): white solid, yield 79%. mp:>250 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.52 (s, 2H), 5.20–5.05 (m, 1H), 4.90–4.40 (m, 2H), 4.00–3.75 (m, 8H), 3.22–3.03 (m, 4H), 2.67–2.58 (m, 1H), 1.84–1.63 (m, 4H), 1.24 (t, J = 7.0 Hz, 1H), 1.00–0.90 (m, 2H), 0.80–0.75 (m, 2H). 13C NMR (DMSO-d6): δ 171.3 (2C), 165.2 (2C), 164.6, 159.1, 159.0, 119.1, 66.5 (2C), 56.5, 55.4, 52.6 (2C), 44.9, 43.7, 28.7, 19.0, 10.8, 7.3 (2C). ESI-MS m/z: 462.3 [M + Na]+.
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino)piperidin-1-yl)(cyclobutyl)methanone (B13): white solid, yield 51%. mp >250 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 5.52 (s, 2H), 5.15–4.66 (m, 2H), 3.99–3.68 (m, 9H), 3.29–3.20 (m, 1H), 3.03–2.95 (m, 4H), 2.70–2.50 (m, 1H), 2.40–2.25 (m, 2H), 2.25–2.17 (m, 2H), 2.09–1.81 (m, 2H), 1.80–1.52 (m, 4H). 13C NMR (DMSO-d6): δ 172 (2C), 165.2 (2C), 164.6, 159.1, 159.0, 119.1, 66.5 (2C), 52.5, 52.4, 44.4, 44.3, 43.7, 41.2, 36.8, 28.6, 25.1 (2C), 17.8 (2C). ESI-MS m/z: 476.3 [M +Na]+.
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino)piperidin-1-yl)(cyclopentyl)methanone (B14): white solid, yield 51%. mp 233–235 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.45 (s, 2H), 5.20–4.75 (m, 2H), 4.12–4.05 (m, 1H), 4.02–3.67 (m, 8H), 3.23–2.92 (m, 5H), 2.63–2.50 (m, 1H), 1.93–1.64 (m, 12H). 13C NMR (DMSO-d6): δ 173.6 (2C), 165.2 (2C), 164.6, 159.1, 158.9, 119.1, 66.51 (2C), 52.6, 52.5, 44.8, 43.7, 41.3, 30.2 (2C), 30.1 (2C), 26.2 (2C), 26.1 (2C). ESI-MS m/z: 468.3 [M + H]+.
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino)piperidin-1-yl)(cyclohexyl)methanone (B15): white solid, yield 53%. mp 181–182 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.55 (s, 2H), 5.05–4.75 (m, 2H), 4.07–4.00 (m, 1H), 3.82–3.70 (m, 8H), 3.29–2.99 (m, 4H), 2.62–2.51 (m, 2H), 1.80–1.75 (m, 8H), 1.51–1.46 (m, 4H), 1.43–1.25 (m, 2H). 13C NMR (DMSO-d6): δ 173.7 (2C), 165.2 (2C), 164.6, 159.1, 159.0, 119.1, 66.51 (2C), 54.0, 52.5, 44.7, 43.7, 42.3, 41.1, 38.7, 29.7, 26.1, 25.7 (2C), 18.5, 17.2, 13.0. ESI-MS m/z: 482.3 [M + H]+.
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(methyl)amino)piperidin-1-yl)(tetrahydro-2H-pyran-4-yl)methanone (B16): white solid, yield 67%. mp 237–239 °C. 1H NMR (CDCl3) δ 9.18 (s, 2H), 5.44 (s, 2H), 5.16–4.70 (m, 2H), 4.04–4.00 (m, 3H), 3.97–3.63 (m, 8H), 3.48–3.44 (m, 2H), 3.18–3.01 (m, 4H), 2.80–2.64 (s, 2H), 2.06–1.79 (m, 4H), 1.70–1.45 (m, 4H, CH2). 13C NMR (DMSO-d6): δ 172.6, 165.2 (2C), 164.6, 159.1, 159.0, 119.1, 66.8 (4C), 66.5 (2C), 52.6 (2C), 44.7, 43.7, 36.8, 29.5, 29.5. ESI-MS m/z: 506.3 [M +Na]+.
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(cyclopropyl)amino)piperidin-1-yl)(cyclopropyl)methanone (B17): white solid, yield 46%. mp: >250 °C. 1H NMR (CDCl3) δ 9.19 (s, 2H), 5.53 (s, 2H), 4.77–4.60 (m, 1H), 4.59–4.25 (m, 2H), 4.00–3.78 (m, 8H), 3.19–3.15 (m, 1H), 2.65–2.53 (m, 2H), 2.19–2.02 (m, 2H), 2.02–1.79 (m, 3H), 1.09–0.85 (m, 4H), 0.80–0.73 (m, 4H). 13C NMR (CDCl3) δ 171.8, 167.1, 167.0, 164.6, 164.0, 159.1 (2C), 121.31, 66.8 (2C), 57.4 (2C), 45.7, 43.6 (2C), 42.4, 11.0 (2C), 9.3, 7.3 (2C). ESI-MS m/z: 466.3 [M + H]+.
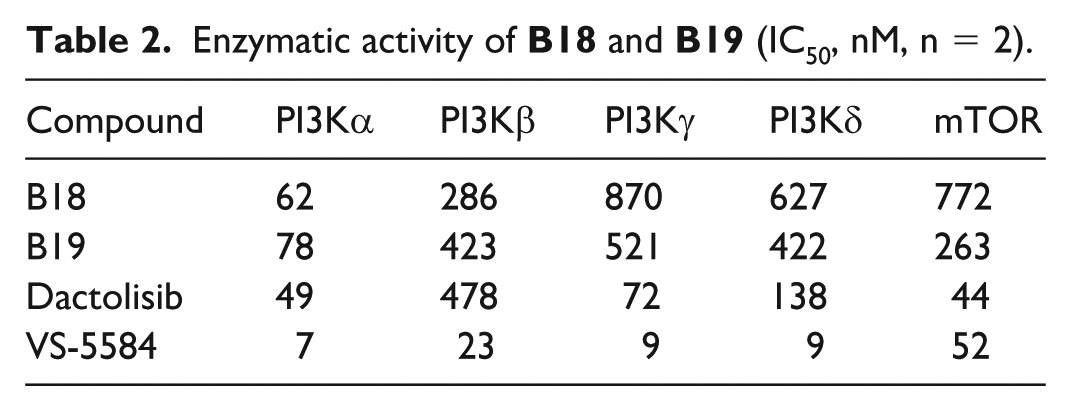
(4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(cyclopropyl)amino)piperidin-1-yl)(cyclopentyl)methanone (B18): white solid, yield 63%. mp 233–236 °C. Anal. calcd for C25H35N9O2: C, 60.83; H, 7.15; N, 25.54; found: C, 60.86; H, 7.13; N, 25.57%; 1H NMR (CDCl3) δ 9.18 (s, 2H), 5.52 (s, 2H), 4.81 (d, J = 8.7 Hz, 1H), 4.43 (s, 1H), 4.09 (d, J = 8.7 Hz, 1H), 3.80–3.73 (m, 8H), 3.11–2.85 (m, 2H), 2.60–2.50 (m, 2H), 2.18–1.97 (m, 2H), 1.95–1.74 (m, 8H), 1.66–1.51 (m, 2H), 0.92 (m, 2H), 0.79–0.66 (m, 2H). 13C NMR (CDCl3): δ 174.4, 167.1, 167.0, 164.6, 164.1, 159.1 (2C), 121.1, 66.8 (2C), 58.3, 57.5, 45.6, 43.6, 42.2, 41.1 (2C), 31.2, 30.3, 30.1, 26.1, 26.0, 18.4, 9.3, 9.2. ESI-MS m/z: 494.4 [M + H]+.
4-((4-(2-Aminopyrimidin-5-yl)-6-morpholino-1,3,5-triazin-2-yl)(cyclopropyl)amino)piperidin-1-yl)(tetrahydro-2H-pyran-4-yl)methanone (B19): white solid, yield 58%. mp 149–151 °C. Anal. calcd for C25H35N9O3: C, 58.92; H, 6.92; N, 24.74; found: C, 58.96; H, 6.89; N, 24.78%; 1H NMR (CDCl3) δ 9.17 (s, 2H), 5.52 (s, 2H), 4.81 (d, J = 8.7 Hz, 1H), 4.43–4.35 (m, 1H), 4.03–4.00 (m, 2H), 3.80–3.75 (m, 8H), 3.48–3.44 (m, 2H), 3.15–3.11 (m, 1H), 2.80–2.74 (m, 1H), 2.63–2.49 (m, 2H), 2.21–1.83 (m, 7H), 1.63–1.50 (m, 2H), 0.95–0.84 (m, 2H), 0.79–0.66 (m, 2H). 13C NMR (CDCl3): δ 172.7, 167.1, 167.0, 164.6, 164.2, 159.1 (2C), 121.1, 67.3 (2C), 66.8, 58.2, 57.4, 45.5, 43.6, 42.1, 37.7 (2C), 29.3, 29.1, 27.3, 18.4 (2C), 9.3 (2C). ESI-MS m/z: 510.3 (M + H)+.
Antiproliferative assays by MTT method
VS-5584 was purchased from Shanghai Biochempartner Company (Purity: 99%, high-performance liquid chromatography), and 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl-2H-tetrazolium bromide (MTT) was purchased from Sigma (St. Louis, MO, USA). The cellular chemosensitivity was determined by using a modified MTT method assay in human cell lines HCT-116 and MCF-7, which was similar to the procedures described.
16
In brief, HCT-116, MCF-7 cells in 200 μL culture medium were seeded into 96-well microplates at 3000–5000 cells/well, respectively, and cultured in Dulbecco’s Modified Eagle Medium with 10% fetal bovine serum (FBS), or RPMI-1640 with 10% calf serum, incubated at 37 °C for 24 h prior to drug exposure. Cell numbers were titrated to keep control cells growing in the exponential phase throughout the 72 h incubation period. Cells were treated with final concentrations of 10.0, 5.0, 1.0, and 0.5 μM of tested compounds simultaneously and incubated for 72 h and then 20 μL of MTT solution (5 mg/mL in PBS) was added to each well at lucifugal condition and incubated for 4 h at 37 °C. The formed purple formazan crystals were pelleted at the bottom of the well, separated from the supernatant, and dissolved in 200 μL of DMSO. The optical density at 570 nm was determined by Varioskan Flash Multimode Reader (Thermo scientific). Three separate experiments with triplicate data were performed to obtain mean cell viability. The IC50 value was calculated according to the inhibition ratios.
PI3K enzymatic activity assay
PI3K and mTOR enzymatic assay was carried out as described in Wang et al.
18
Molecular modeling
The PI3Kα (PDB code: 5UK8) was used in the molecular modeling, and the docking procedure was performed as described in Hei et al.
17
The protein-ligand complex crystal structure was chosen as the template, to elucidate the binding mode of B18. Protein structure was downloaded from Protein Data Bank (PDB 5UK8). The molecular docking procedure was performed within SYBYLX 2.0 software. The EGFR enzyme was defined as a receptor and the site sphere was selected based on the ligand binding location. Ligand was removed and compound B18 was placed, other parameters were set as default. After accomplishment of the molecular docking procedure, 20 docking poses were scored and selected based on calculated energy. The best docking pose was prepared using PyMOL as showed in Figure 3.