Abstract
A facile one-pot synthesis of highly functionalized dialkyl 1-(5-aryl-1,3,4-thiadiazol-2-yl)-4-ethoxy-5-oxo-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate derivatives via the reaction between acetylenic esters, triphenylphosphine, and ethyl 2-[(5-aryl-1,3,4-thiadiazol-2-yl)amino]-2-oxoacetate is developed. The structure of the products is confirmed by spectroscopic methods.
Keywords
One-pot synthesis of 4-ethyl 2,3-dimethyl 1-(5-aryl-1,3,4-thiadiazol-2-yl)-5-oxo-2,5-dihydro-1H-pyrrole-2,3,4-tricarboxylate derivatives via intramolecular Wittig reaction.
Introduction
Exhibiting interesting biological behavior, heterocyclic compounds have found a special place in the pharmaceutical industry.1,2 Many substances containing five-membered heterocycles demonstrate a variety of interesting biological activities. In this category, 1,3,4-thiadiazoles and 2-pyrrolidinones have been used as structural motifs in the construction of substances having multi-target active, such as anti-inflammatory,3,4 antimicrobial,
5
anticonvulsant,6,7 and antihypertensive.8,9 1,3,4-Thiadiazoles have also been frequently used as important building blocks for the synthesis of biologically active heterocyclic molecules. Heterocycles, bearing a 1,3,4-thiadiazole moiety, are reported to demonstrate a broad spectrum of biological activity such as antimicrobial, tranquilizer, antiepilepsy, antiinflamatory, antiviral, anti-coagulant, and antitumor (Figure 1, structures

Biologically active thiadiazole and lactam molecules.
Moreover, 2-pyrrolidinones, as a class of five-membered lactams with a four-carbon heterocyclic ring structure, are of interest to biologists.13,14 2-Pyrrolidinone play a significant role in natural products and pharmaceuticals. Some known biologically important natural products comprising a 2-pyrrolidinone unit are cotinine, a tobacco alkaloid,
15
doxapram, a respiratory stimulatant,
16
and ethosuximide, a succinimide containing anticonvulsant.
17
Moreover, substituted 2-pyrrolidinone derivatives have various therapeutic properties, for example, anti-cancer,
18
antitumor,
19
HIV-1 integrase inhibition,20,21 antimicrobial,
22
antibacterial,
23
and anti-inflammatory.
24
In addition, the 2-pyrrolidinone moiety is used in the drug PI-091, consisting of anti-coagulant properties, as well as in Jatropham, which is an antitumor alkaloid (Figure 1, structures
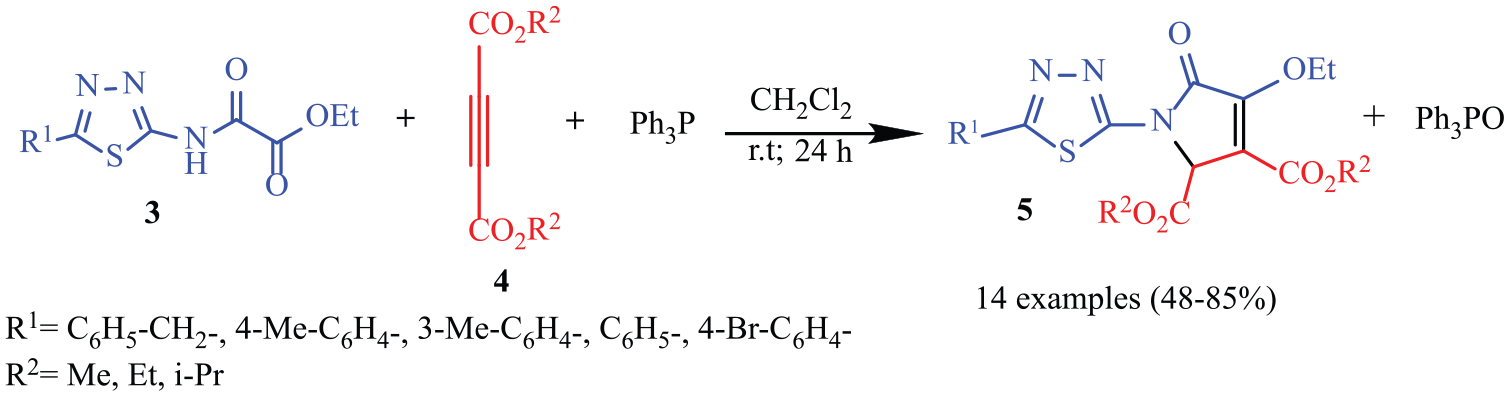
The Wittig reaction of phosphonium ylides with carbonyl compounds has been adopted widely for the construction of C=C double bonds in an inter- or intramolecular approach under mild reaction conditions.37,38 The intramolecular Wittig reaction is one of the most common methods for the synthesis of different five-membered heterocycles through intramolecular cyclization of phosphorus ylides with various carbonyl groups, such as aldehyde, ketone, ester, amide, and imide.39–43 To date, a number of synthetic approaches to pyrrolidinones structures have been developed.44–49 In 2007, Anary-Abbasinejad et al. 50 reported the synthesis of novel highly functionalized pyrrolidinones via intramolecular Wittig reactions. Inspired by the biological profiles of 1,3,4-thiadiazoles and pyrrolidinones and as part of our interest in the synthesis of heterocyclic compounds via intramolecular Wittig reactions,51,52 we sought to synthesize the title compounds. For this purpose, first ethyl chlorooxoacetate was reacted with 2-amino-1,3,4-thiadiazoles for the synthesis of ethyl 2-oxo-2-[(5-aryl-1,3,4-thiadiazol-2-yl)amino]acetate derivatives (Scheme 1). Subsequent reaction with triphenylphosphine and acetylenic esters gave the desired products (Scheme 2).

Synthesis of ethyl 2-oxo-2-[(5-aryl-1,3,4-thiadiazol-2-yl)amino] acetates.

Synthesis of the final products.
Results and discussion
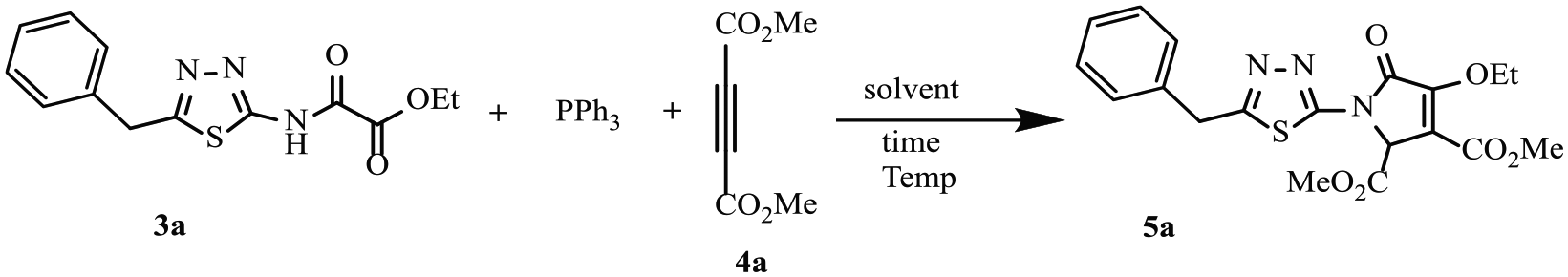
Initially, a model reaction was conducted using triphenylphosphine (1 mmol), ethyl (5-benzyl-1,3,4-thiadiazol-2-ylcarbamoyl)formate (
Optimization of the reaction conditions.

Ultimately, CH2Cl2, room temperature, and 1.5 mmol triphenylphosphine were selected as the optimized conditions. With optimized conditions established, we next tested the scope of various reactants. Ethyl 2-oxo-2-[(5-aryl-1,3,4-thiadiazol-2-yl)amino]acetates
Synthesis of compounds

The structure elucidation of compounds
The reaction mechanism is presented in Scheme 3. The initial step is formation of zwitterion intermediate

The mechanism of reaction.
Conclusion
In summary, we have established that the one-pot reaction between Ph3P, acetylenic esters
Experimental
Melting points were determined on an Electrothermal 9100 apparatus. Infrared (IR) spectra were recorded using a Nicolet Avatar 370 FTIR Therma spectrometer as KBr pellets and are reported in cm−1. 1H NMR and 13C NMR spectra were recorded on a Bruker DPX-300 Avance instrument at 300 and 75.47 MHz, respectively. CDCl3 were used as the solvent and shifts are given in ppm. Elemental analyses were performed using a Thermo Finnegan Flash EA 1112 series instrument. Mass spectra were recorded with a Varian Meth ch-7 at 70 eV. Column chromatography was performed on silica gel (160–200 mesh, E. Merck). Thiadiazole derivatives 3, were prepared according to the literature source. 53
Dimethyl 1-(5-benzyl-1,3,4-thiadiazol-2-yl)-4-ethoxy-2,5-dihydro-5-oxo-1H-pyrrole-2,3-dicarboxylate (5a ); typical procedure
To a stirred solution of ethyl (5-benzyl-1,3,4-thiadiazol-2-ylcarbamoyl)formate (
Pale yellow powder (0.292 g, 70%), m.p. 118 °C–119 °C; IR (KBr) (υmax/cm−1): 1750, 1702, 1641 (3 C=O); 1H NMR (300.13 MHz, CDCl3): δ (ppm) = 1.42 (3H, t,
Dimethyl 4-ethoxy-5-oxo-1-[5-(p-tolyl)-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Dimethyl 4-ethoxy-5-oxo-1-[5-(m-tolyl)-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Dimethyl 4-ethoxy-5-oxo-1-[5-phenyl-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diethyl 1-[5-benzyl-1,3,4-thiadiazol-2-yl]-4-ethoxy-5-oxo-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diethyl 4-ethoxy-5-oxo-1-[5-(p-tolyl)-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diethyl 4-ethoxy-5-oxo-1-[5-(m-tolyl)-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diethyl 4-ethoxy-5-oxo-1-[5-phenyl-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diethyl 1-[5-(4-bromophenyl)-1,3,4-thiadiazol-2-yl]-4-ethoxy-5-oxo-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diisopropyl 1-[5-benzyl-1,3,4-thiadiazol-2-yl]-4-ethoxy-5-oxo-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diisopropyl 4-ethoxy-5-oxo-1-[5-(p-tolyl)-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diisopropyl 4-ethoxy-5-oxo-1-[5-(m-tolyl)-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diisopropyl 4-ethoxy-5-oxo-1-[5-phenyl-1,3,4-thiadiazol-2-yl]-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Diisopropyl 1-[5-(4-bromophenyl)-1,3,4-thiadiazol-2-yl]-4-ethoxy-5-oxo-2,5-dihydro-1H-pyrrole-2,3-dicarboxylate (
Supplemental Material
Supplementary_material – Supplemental material for One-pot synthesis of 4-ethyl 2,3-dimethyl 1-(5-aryl-1,3,4-thiadiazol-2-yl)-5-oxo-2,5-dihydro-1H-pyrrole-2,3,4-tricarboxylate derivatives via intramolecular Wittig reaction
Supplemental material, Supplementary_material for One-pot synthesis of 4-ethyl 2,3-dimethyl 1-(5-aryl-1,3,4-thiadiazol-2-yl)-5-oxo-2,5-dihydro-1H-pyrrole-2,3,4-tricarboxylate derivatives via intramolecular Wittig reaction by Samin Iravani and Abbas Ali Esmaeili in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Research Council of Ferdowsi University of Mashhad is acknowledged for financial support (Grant No. 3/50159).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.