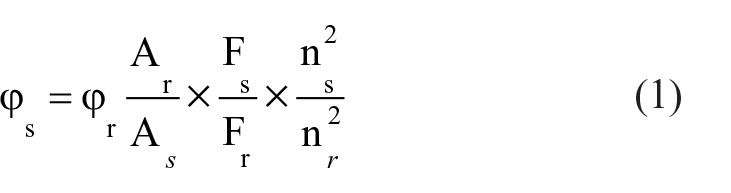Abstract
Taking advantages of both the well-known α,β-unsaturated structure and the special nucleophilicity of organic amines toward its acceptor moieties, intramolecular charge transfer as a signaling mechanism is used to design and synthesize a new ratiometric chromophoric fluorescent probe (BI-CA-ID) with large emission shifts toward organic amines. This probe is employed for the detection of organic amines with high selectivity and sensitivity and a “naked-eye” color change (from red to colorless). Ultraviolet–visible and fluorescence spectrometric measurements are used to determine detection limits as low as 0.024 and 0.43 μM. Furthermore, nucleophilic addition of the amine on the α,β-unsaturated of BI-CA-ID indicated that the sensing mechanism occurs via interruption of the π-conjugation.
A probe with an α,β-unsaturated structure and large emission shift was developed to detect organic amines. The spectral properties of the probe toward organic amines were studied. The results show that the probe can selectively and effectively monitor organic amines with naked-eye color change, and the detection limits is 0.43 μM.

Introduction
The highly toxic nature of organic amines with adverse effects to both human health and the environment has greatly increased the need to develop methods to identify their presence.1,2 Organic amines are present in a wide range of fertilizers, herbicides, pharmaceuticals, surfactants, latex rubber, biological buffer substances and colorants and occur also as industrial, agricultural, and manufacturing pollutants. Such amines are also toxic to human skin and many systems in the human anatomy (i.e. the nervous system, the urinary system, hematopoietic systems, and respiratory systems).1,3 Although a number of traditional methods have worked well for the detection of organic amines, such as gas chromatography–mass spectrometry (GC-MS), 4 high-performance liquid chromatography (HPLC), 5 and electrochemical systems,6,7 there are still some drawbacks, for instance, the requirement of expensive instrumentation and complex and time-consuming procedures. Fluorescent probes, however, have a degree of excellence in detection that can avoid these disadvantages.
The high selectivity, sensitivity, and real-time imaging capabilities have made fluorescent probes the subject of substantial research.8–20 For example, to elucidate the selectivity of interactions among primary, secondary, and tertiary amines, Sriramulu and Valiyaveettil 21 explored the fluorescence quenching behavior of perylene derivatives in the presence of various organic amines in solution. Similarly, Yao et al. synthesized fluorescent trifluoroacetyl-substituted aromatic compounds for the quick, sensitive, and reversible detection of organic amine vapors with detection limits (DLs) as low as 0.17 ppb for diethylamine and 0.23 ppb for aniline. 16 Next, using the Knoevenagel condensation and showing reactivity toward secondary and aromatic amines with a DL of 75 μM, Kumpf et al. 22 synthesized eight acceptor-substituted distyrylbenzene derivatives. Despite these outstanding successes, several problems must be resolved to refine these novel organic amine fluorescent probes. First, the complicated experimental procedure comprising probe design and synthesis must be streamlined. 23 Second, the small emission shifts (typically less than 150 nm) of this current generation of probes must be enhanced to make them desirable for the determination of fluorescent intensities and ratios. Specifically, the substantial spectral overlap between the probes and their products after responding to the analytes must be reduced.24,25 Finally, most existing probe materials are ineffective in the detection of multistage amines.26,27 Therefore, there is an urgent need to develop novel, ratiometric probes for organic amines with larger emission shifts.
Carbazole, an electron donor with a large rigid planar structure, has excellent photo-conductivity, a relatively intense luminescence, and a high thermal stability and can be prepared and modified easily. 28 Although carbazole-based compounds are available,29–34 the use of strong emission properties for chemosensing has only been recently undertaken.35–39
Herein, we have developed a novel chromophoric fluorescent probe (BI-CA-ID) based on the condensation reaction of an indole and carbazolyl aldehyde. We also extended the π-conjugation via Suzuki–Miyaura coupling to obtain fluorophores with emissions at longer wavelengths. This probe has a fast response to organic amines with high selectivity and sensitivity. As such, dramatic hypsochromic shifts in both the absorption and emission profiles of BI-CA-ID are observed.
Results and discussions
Synthesis of the probe BI-CA-ID
As known, organic amines are nucleophilic that can react with α,β-unsaturated compounds. Thus, based on the nucleophilic addition reaction which can cause a rapid change in the fluorescence signal, the probe BI-CA-ID was synthesized. Through Knoevenagel reaction of an indole and carbazolyl, the reaction site was developed. To enlarge the conjugation of the probe, the biphenyl fragment was introduced by Suzuki reaction. The Synthetic route is shown in Scheme 1. The 1H NMR, 13C NMR, and high-resolution mass spectrometry (HRMS) characterization of BI-CA-ID are shown in the supporting information.

Synthetic route toward BI-CA-ID.
Optical response of BI-CA-ID toward the organic amine, n-BuNH2
Absorption and fluorescence spectroscopy were used to examine the sensing behaviors of BI-CA-ID toward n-BuNH2 (Figure 1(a)). Two distinct absorption bands at 292 and 501 nm occur with BI-CA-ID, with the first attributed to a π-π* transition and the second to intramolecular charge transfer (ICT) transition. An increase in the level of n-BuNH2 (0–2.5 equiv.) added to BI-CA-ID (40 μM) led to a gradual decrease of the absorption peak at 501 nm and a progressive increase of the absorption band at around 292 nm. The result was a very noticeable color change from red to colorless, indicating the inhibition of both the π-conjugation and the ICT progress of BI-CA-ID on nucleophilic addition of -NH2 to the α,β-unsaturated structure. Note that the occurrence of a well-defined isosbestic point at 346 nm implied the formation of only a single new species during the sensing process. The DL of the probe was 0.024 μM, calculated from the plot of the ratio (A292/A501) and concentration of n-BuNH2.

(a) Absorption spectra of BI-CA-ID (40 μM) with increased n-BuNH2 concentrations (0–2.5 equiv.) in solution (CH3CN:water = 1:1). (b) The titration curve plotted from the absorbance ratio of BI-CA-ID (A292/A501). (c) The BI-CA-ID (40 μM) fluorescence spectra with increased n-BuNH2 concentrations (0–2.5 equiv.). (d) The titration curve plotted from the BI-CA-ID fluorescence ratio (I382/I601). Excitation wavelength: 500 nm, excitation slit: 5 nm, and emission slit: 20.0 nm.
The above studies provided the basis for examining the changes in the fluorescence spectrum under the same conditions (Figure 1(c)). The excitation of the probe at 500 nm produced a dual emission with peaks at 382 and 601 nm. The band at 601 nm is attributed to the ICT emission band, and the band at 382 nm is ascribed to the biphenyl carbazole moiety. The addition of n-BuNH2 gradually quenches the fluorescence intensity of the solution at 601 nm and gradually increases the intensity at 382 nm, yielding a bright blue emission that is clearly visible by the naked eye on UV-lamp irradiation at 365 nm. Simultaneously, a clear iso-emissive point at 523 nm indicated the formation of the BI-CA-ID-NH-adduct. The distinct wavelength difference between the two emission bands is almost 220 nm, which makes the probe favorable for ratiometric fluorescent detection of organic amines. A good linear relationship between the intensity ratio (I382/I601) and the concentration of n-BuNH2 could be obtained in the range of 2.0 × 10−6 to 8.0 × 10−6 M (R2 = 0.9924). This ratiometric fluorescence change is potentially useful for the quantitative determination of organic amines, with the DL of BI-CA-ID being 0.43 μM. The quantum yield of the probe was calculated to be 0.077.
Selectivity studies
To assess the specificity, BI-CA-ID (40 μM) was treated with the following anions and molecules (0.8 mM) under identical conditions: Cl−, Br−, I−,
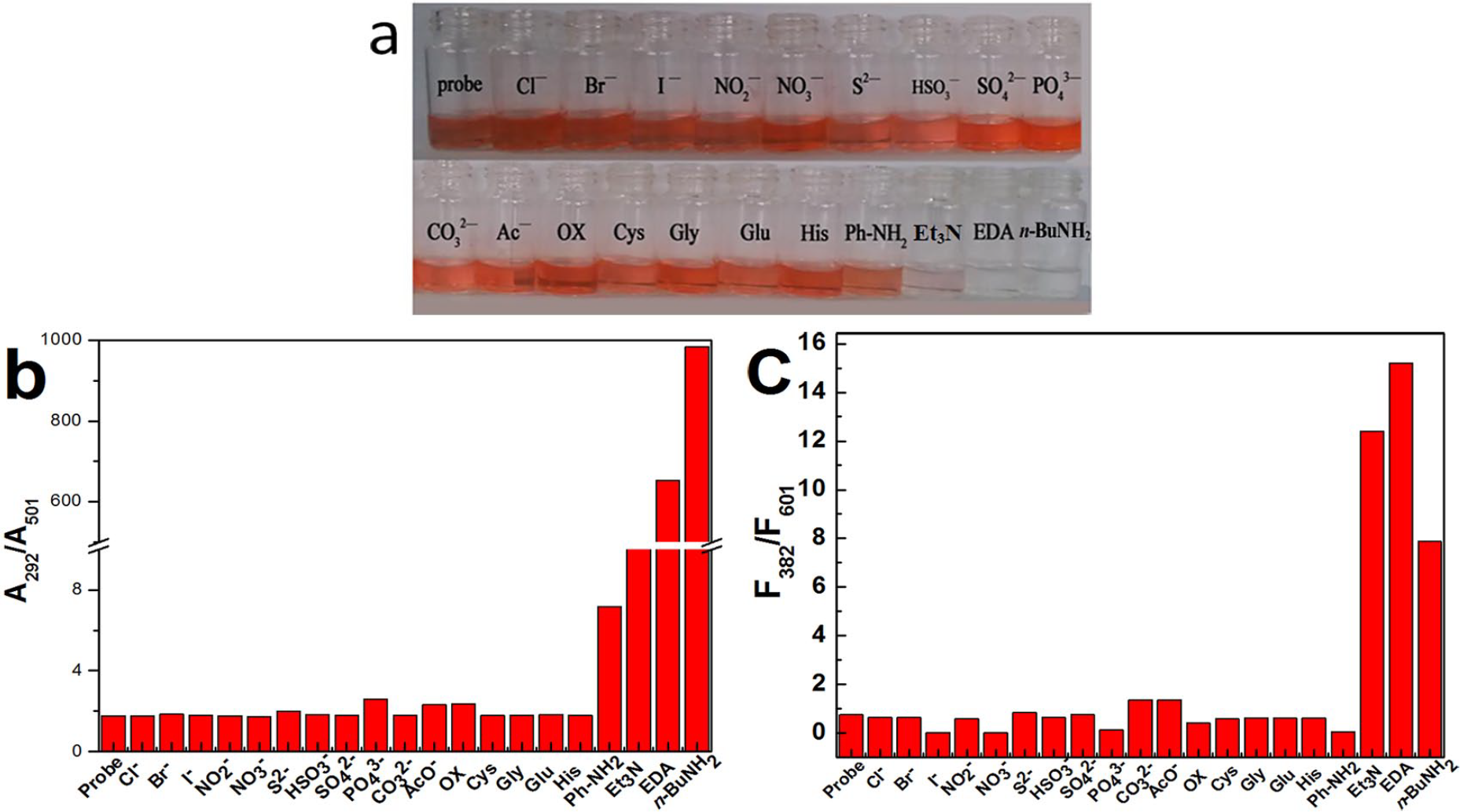
(a) Response of BI-CA-ID to various anions (EDA is ethylenediamine) and molecules. (b) and (c) Absorption and fluorescence response of BI-CA-ID (40 μM) in the presence of different anions and molecules (0.8 mM). Excited at 500 nm, excitation slit: 2.5 nm, and emission slit: 20.0 nm.
To further examine the high selectivity for organic amines of BI-CA-ID, a panel of related alkyl amine compounds was analyzed under identical conditions. The corresponding changes in the fluorescence and absorbance spectra before and after addition of different amines were recorded as shown in Figure 3. As expected, in the presence of alkyl amines, the F382/F601 ratio was enhanced significantly, and a decrease in absorbance at 501 nm occurred, indicating similar response and color changes as depicted in Figure 1. All the results further demonstrated the acquisition of a new ratiometric fluorescent probe for detecting organic amines.
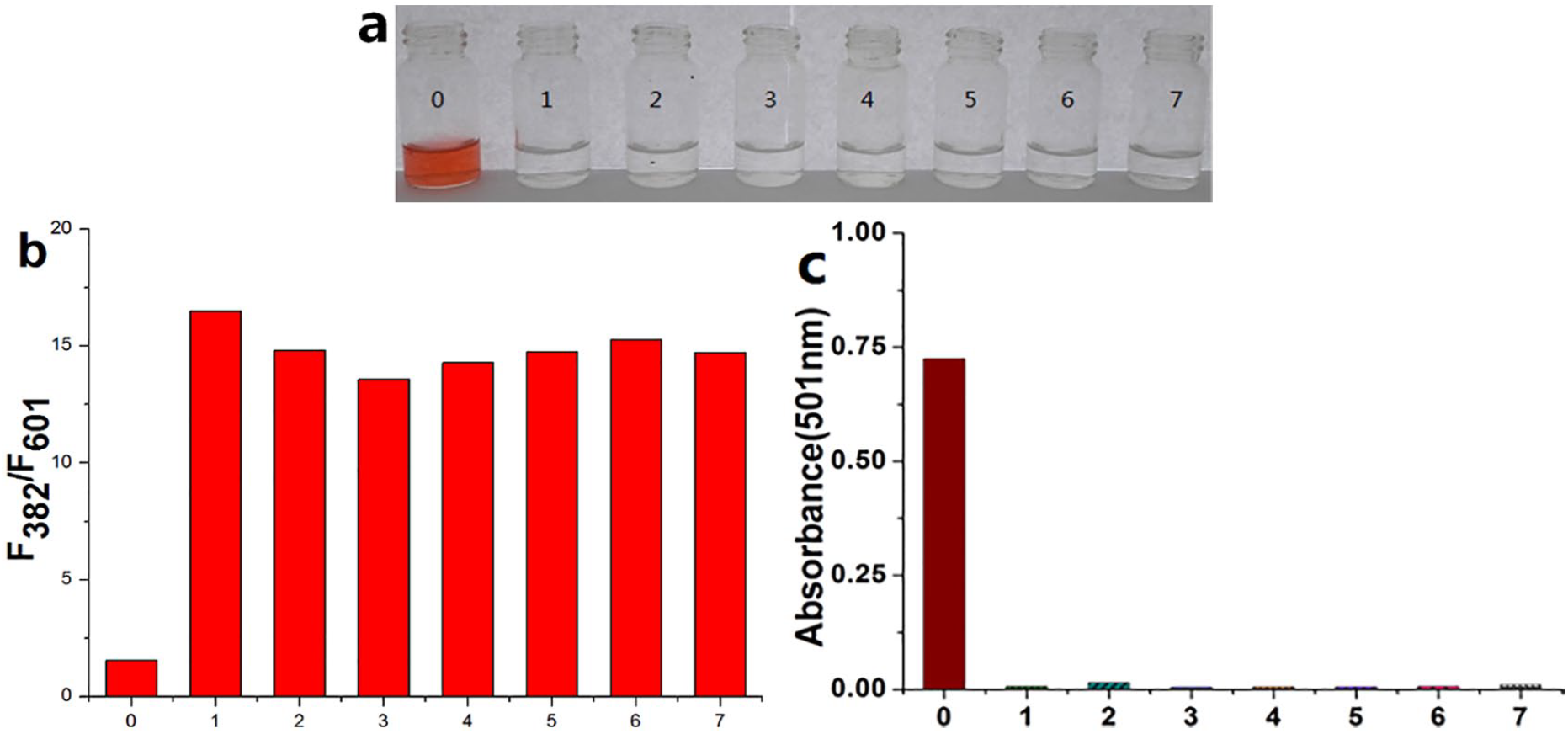
(a) The color changes after adding different amines. (b) and (c) The fluorescence and absorbance response of BI-CA-ID in the presence of different organic amines (400 μM) after 10 min. (a) From left to right: blank solution (0), butylamine (1), isopropylamine (2), ethanediamine (3), dicyclohexylamine (4), diisopropylamine (5), diisopropylethylamine (6), and triethanolamine (7). Excited at 500 nm, excitation slit: 2.5 nm, and emission slit: 20.0 nm.
Effect of pH toward the probe
Next, to examine the influence of the pH value on the probe, we explored its response to different pH values. Initially, the pH titration was carried out in water. As shown in Figure 4, there is little influence upon BI-CA-ID when the Ph = 4–10, which indicates its suitability for near-neutral pH solutions.
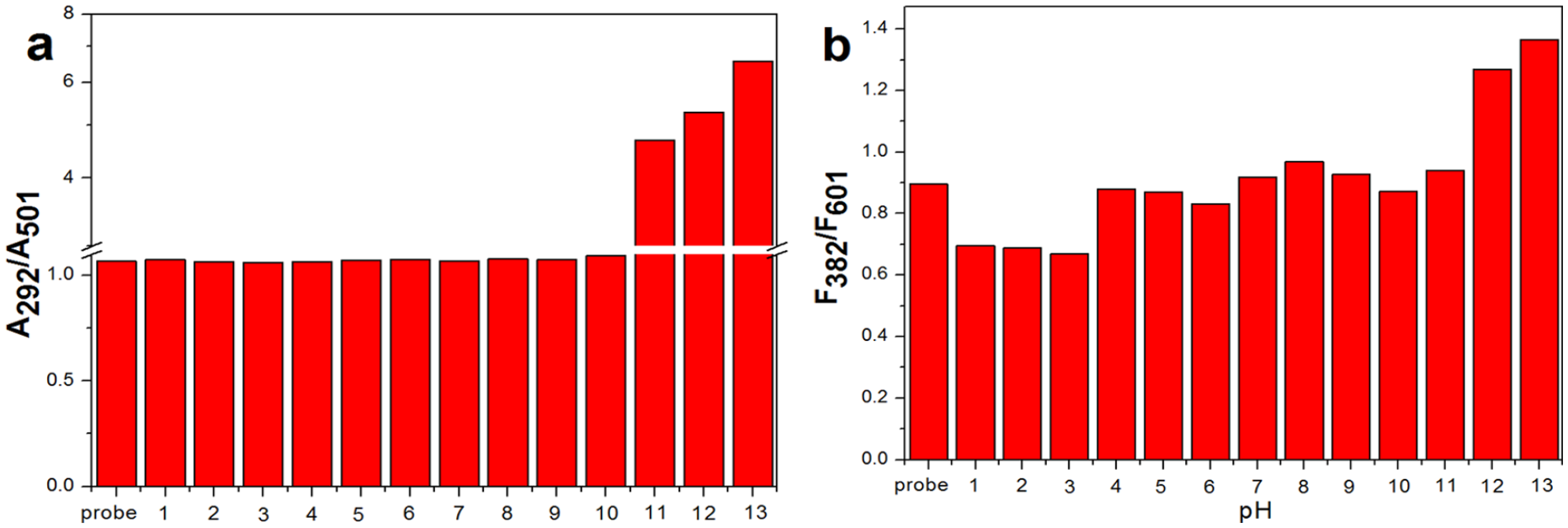
(a) Plot of the absorbance ratio (A292/A501) of BI-CA-ID (40 μM) with increasing value of pH in water. (b) Plot of the intensity ratio (I382/I601) of BI-CA-ID (40 μM) with increasing value of pH in water, excited at 365 nm.
Mechanism study
Scheme 2 contains an illustration of the mechanism for the sensory response of BI-CA-ID to n-BuNH2. We studied and contrasted the 1H NMR spectra of the probe in the absence and presence of n-BuNH2. The addition of an equivalent of n-BuNH2 to a BI-CA-ID solution in CDCl3 at room temperature yielded significant shifts of several proton signals. The vinyl protons at 9.14 (Hc) and 8.82 (Hb) ppm shifted upfield toward 7.91 (Hc) and 6.56 (Hb) ppm, indicating a decrease in the electron-withdrawing effect of the indoline quaternary N atom. A dramatic shift in the methyl protons Hd adjacent to the quaternary nitrogen from 4.37 to 3.30 ppm occurred due to the removal of the electron-withdrawing effect originating from the indole moiety. Besides, Ha of the carbazole N atom shifted slightly from 4.27 to 4.36 ppm, also the appearance of He at 3.30 ppm was observed. All these observations are in accordance with the nucleophilic addition of –NH2 to the indole carbon in BI-CA-ID.

The sensing mechanism.
Conclusion
A new ratiometric and highly selective chemosensor, BI-CA-ID, for alkyl amines was successfully synthesized. Upon reaction with the –NH2 function as a nucleophile, the probe displays substantial dual changes in both its ratiometric emission and absorption spectra. The significant changes in color, attributed to a restricted ICT, which, due to the formation of a BI-CA-ID-NH-adduct, was observable with the naked eye. The estimated DLs of BI-CA-ID calculated by UV and fluorescence techniques were 0.024 and 0.43 μM, respectively, clearly indicating the promise of this novel dual-fluorescent probe for the rapid detection of alkyl amines and in chemical and environmental applications.
Experimental
General and materials
All reagents were purchased from commercial suppliers and used without further purification unless otherwise specified. Qingdao Hai Yang silica gel (200–300 mesh) was used for flash chromatography and an F-4600 FL spectrophotometer with a 10 mm quartz cuvette was used to measure the fluorescence spectra and relative fluorescence intensity. A Specord Plus instrument was used to capture the ultraviolet–visible (UV-Vis) spectra. A Bruker spectrometer operating at 400 MHz, with tetramethylsilane (TMS) as the internal standard, was used to collect the 1H and 13C NMR spectral data. A Bruker Compact TOF mass spectrometer was used to record the high-resolution mass spectra (HRMS).
Synthesis of the probe BI-CA-ID
9-Dodecyl-9H-carbazole
9-Dodecyl-9H-carbazole-3-carbaldehyde (
6-Bromo-9-dodecyl-9H-carbazole-3-carbaldehyde (
6-([1,1′-biphenyl)-4-yl)-9-dodecyl-9H-carbazole-3-carbaldehyde (
Synthesis of the probe BI-CA-ID
The novel product BI-CA-ID was synthesized based on an analogous procedure.
42
As shown in Scheme 1, under an N2 atmosphere, a mixture of 1-butanol (5 mL) and toluene (5 mL) containing compound
General procedure for the measurement of spectra
Stock solutions of BI-CA-ID (0.1 mmol) were prepared in CH3CN and then diluted to derive the target solution (40 μM). The target solution (1.5 mL) and 1.5 mL of different concentrations of n-BuNH2 in water were combined to obtain the test solution (3 mL), which was then maintained at room temperature (25 °C) for 30 s. The absorption and fluorescence spectra were recorded, and the fluorescence ratio of I382/I601 measured with the excitation wavelength set at 500 nm.
Determination of the DL
The DL was obtained by 3Sb/k, where Sb is the standard deviation of the blank measurements (10 times) and k is the slope of the fitted line.
Quantum yield
The fluorescence quantum yield can be calculated by means of equation (1)
where the subscripts s and r refer to the sample and the reference, respectively, and Ф, F, A and n stand for quantum yield, the integrated emission intensity, the absorbance and refractive index, respectively. Rhodamine B (Φ = 0.97) in ethanol was used as the standard for calculating the fluorescence quantum yields of the probes.
Supplemental Material
Supplementary_file – Supplemental material for Detection of organic amines using a ratiometric chromophoric fluorescent probe with a significant emission shift
Supplemental material, Supplementary_file for Detection of organic amines using a ratiometric chromophoric fluorescent probe with a significant emission shift by Dan Wu, Yi Liu, Fei Zheng, Shi-Qi Rong, Tao Yang, Yan-Kun Zhao, Rui-Wu Yang, Ping Zou and Guang-Tu Wang in Journal of Chemical Research
Footnotes
Acknowledgements
D.W., Y.L., and F.Z. contributed equally to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Foundation of Two-Way Support Programs of Sichuan Agricultural University (Nos 03572832 and 03572227).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.