Abstract
The synthesis of novel metallophthalocyanines (M = Zn, Mg, and Co) derived from (
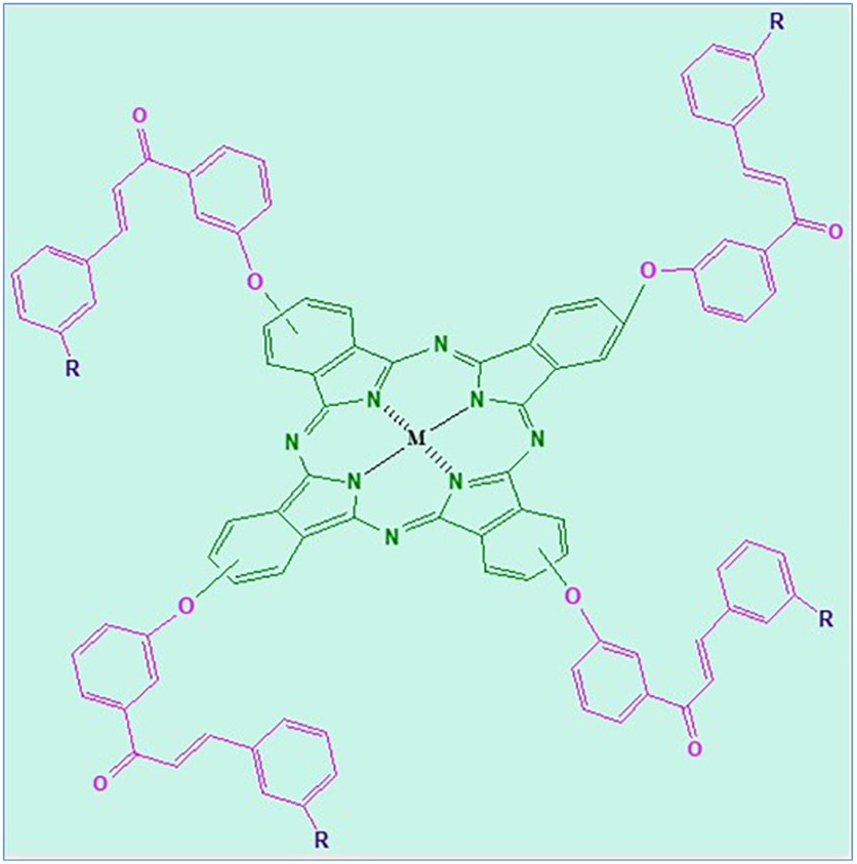
Introduction
Phthalocyanines (PCs) have been examined at an increasing rate since their discovery. 1 This macrocyclic molecule with 18 π-electrons in the conjugated system can be easily synthesized, can be derivatized from peripheral positions to tetra, and can also be derivatized from non-peripheral positions.2–5 This allows for the formation of a rich pool of molecules and to the design of PCs that can be applied in many different applications. Because of their macrocyclic aromatic structure, simple derivatization and easy electron exchange, PCs are frequently used in areas such as catalysis, 6 organic photovoltaic devices and solar cells, 7 electrophotography, 8 Langmuir–Blodgett films, 9 photosensitizers in photodynamic therapy, 10 electrochromic displays, 11 liquid crystals, 12 and optical disks. 13
The solubilization of PCs is necessary for their use in various applications and to improve their low solubility. 14 Two methods are applied for this purpose. The first is the choice of the central metal ion to increase the interaction with solvent molecules. The second is substitution with small moieties. The latter method is also expected to give additional properties to the PC derivative.15,16
Recently, the sensor and biological activities of chalcones have been studied and diverse biological activities have been found, for example, antioxidant, cytotoxic, antiviral, tyrosinase inhibitory, antimalarial, antibacterial, and anti-inflammatory.17–19 In addition, chalcones are used as stating materials for synthesizing many compounds such as flavones, thiadiazines, isoxazoles, quinolinones, benzodiazepines, benzothiazepines, and benzofuranones. 20 Derivation of PCs with chalcones and the applications of these new compounds are quite new. With PCs derived from chalcones, applications as metal sensors, 21 catalysts, 22 photovoltaic devices, and solar cells 23 have been described. Both colorimetric and fluorometric techniques have been studied in metal titrations of PCs derived with chalcones. 24
In this study, the novel phthalonitrile derivatives
Results and discussion
Synthesis
The synthesis of zinc(II), magnesium(II), and cobalt(II) PCs with chalcones bearing halogen atoms was performed by the procedure described in Scheme 1. Chalcone compounds

The synthetic route to the metallophthalocyanines.
The reaction yields for compounds
Solubility is an important factor for electrochemical- and metal-sensing applications and our synthesized MPcs are highly soluble in common organic solvents such as chloroform, tetrahydrofuran (THF), dimethylformamide (DMF), and dimethyl sulphoxide (DMSO). The MPcs
Structure elucidation
Spectroscopic techniques were used to identify the structures of the phthalonitriles. In the FTIR spectra of phthalonitrile compounds
In the 1H NMR spectra of the phthalonitrile compounds (Figure 1), the OH group at 12.8 ppm had disappeared and aromatic protons appeared at around 8.00–7.15 ppm. The 13C NMR spectra of compounds
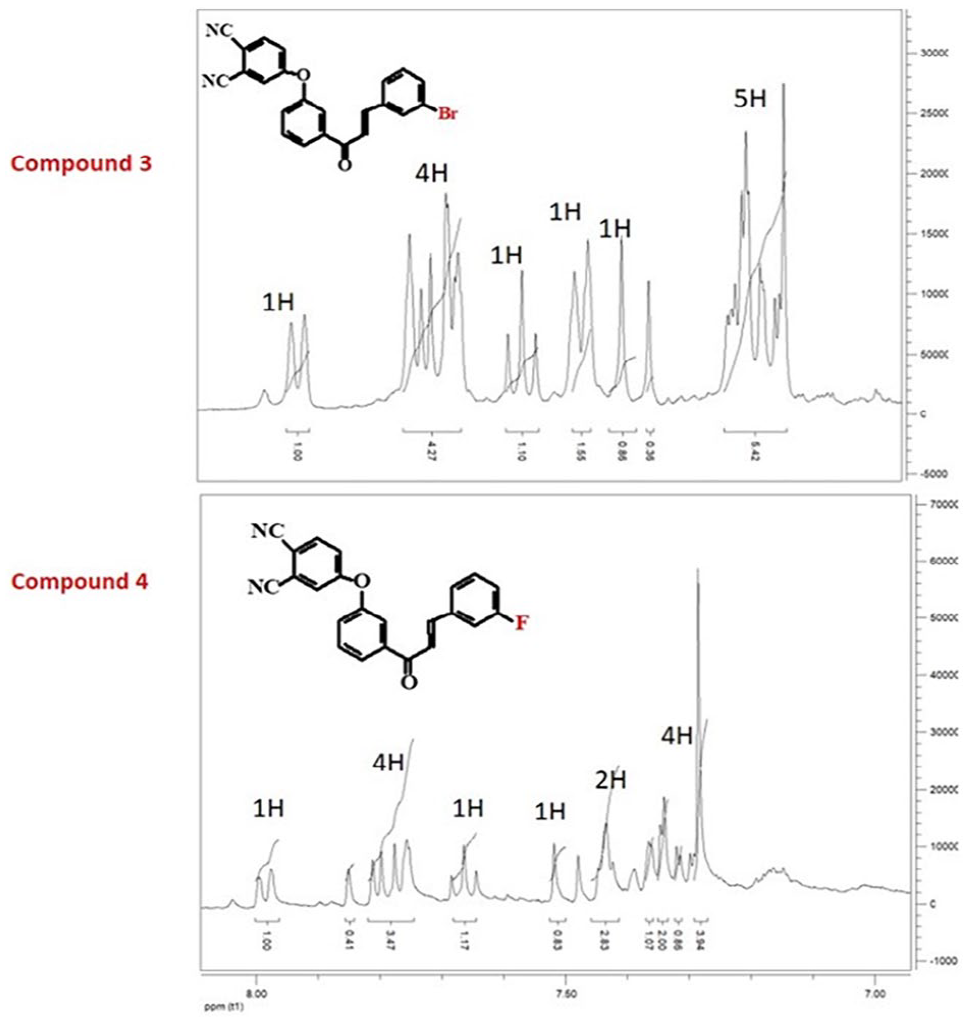
1H NMR spectra of phthalonitrile compounds
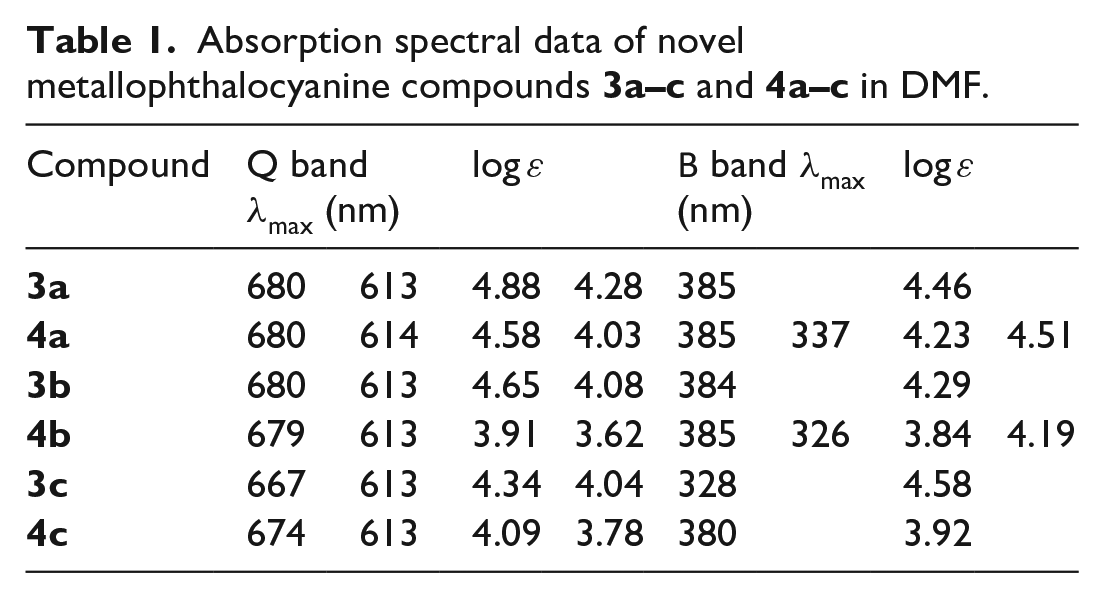
Absorption spectral data of novel metallophthalocyanine compounds
The disappearance of the C≡N vibration band at 2232 cm−1 is considered as evidence of the formation of zinc(II) PCs
In the mass spectra of the zinc, magnesium, and cobalt Pcs, the presence of molecular ion peaks at
UV-Vis absorption spectra and aggregation properties
PC complexes in the UV-Vis region show two strong absorptions regions; the first one is at 300–500 nm and is called the B band. The second, the more energetic absorption, is known as the Q band, and occurs at near 600–700 nm.
29
The UV-Vis absorption spectra of the synthesized zinc, magnesium, and cobalt PCs

UV-Vis spectra of phthalocyanine compounds

UV-Vis spectra of phthalocyanine compounds
The MPCs
As shown in Figure 3, PC complexes
Aggregation depends on the concentration, solvent, temperature, metal ions, and structures of the substituents. 30 Aggregation of the PC compounds is generally examined by changing the concentration of the PC or by changing the solvent. These factors will affect the shape and position of the Q band when aggregation happens.
In this study, the aggregation behavior of the MPC complexes
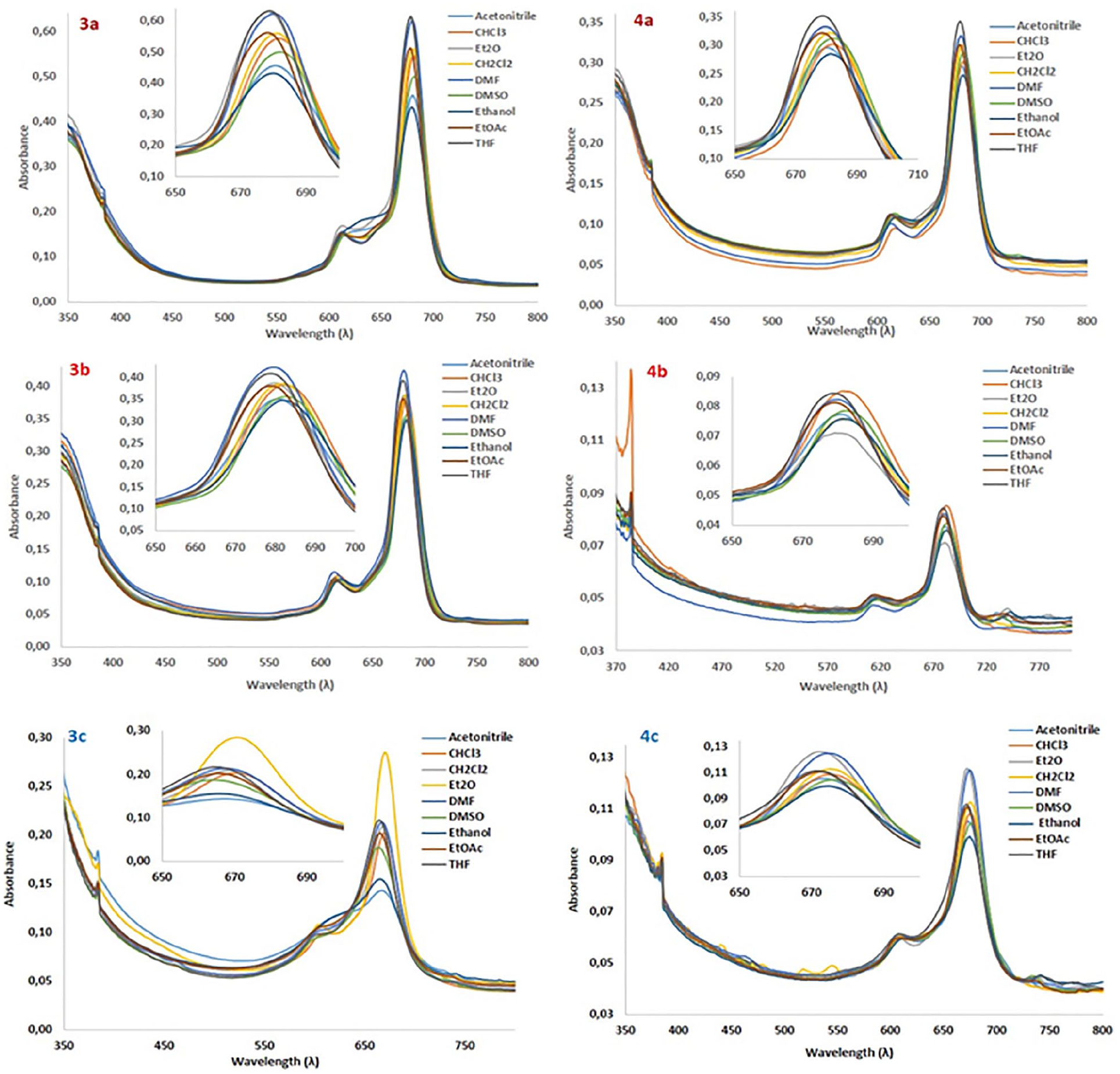
Absorption spectra of ZnPcs

Absorption spectra of ZnPcs
Compound
The UV-Vis absorption spectra of cobalt PCs
The effect of changing the concentration on the aggregation properties of the ZnPcs (
Cyclic voltammetric measurements
The cyclic voltammetric measurements of all the MPcs were recorded on a Parstat 2273 potentiostat/galvanostat. In three-electrode systems, a Pt working electrode, a Pt counter electrode and a glassy carbon electrode, as reference, were used. As the electrolyte pure 0.1 M tetra-
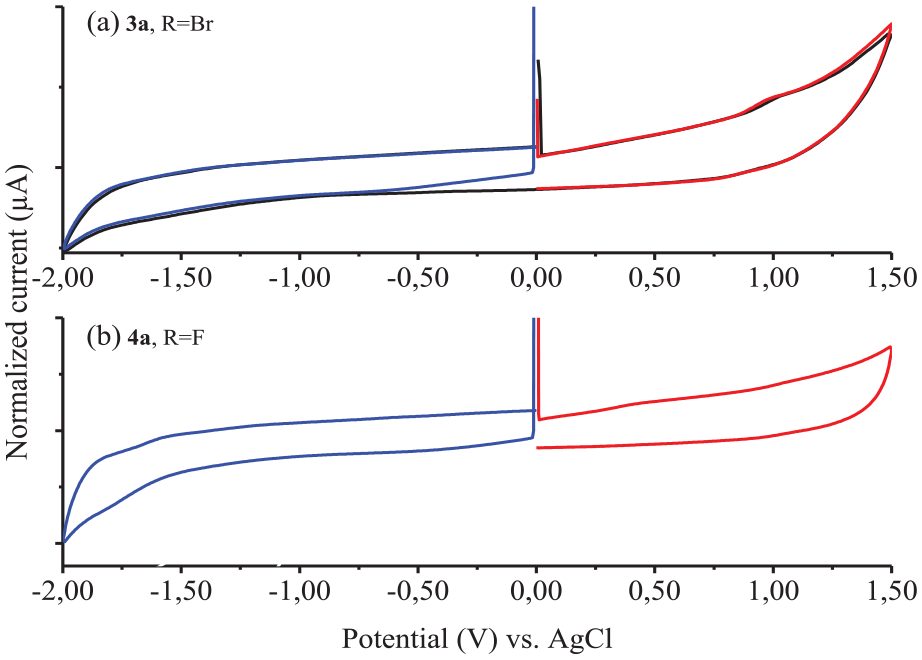
Cyclic voltammograms of zinc phthalocyanines

Cyclic voltammograms of magnesium phthalocyanines

Cyclic voltammograms of cobalt phthalocyanines
Compounds
Fluorometric metal ion titrations of Zn PC compounds 3a and 4a
The fluorescence spectra were recorded on a Hitachi F-7000 fluorescence spectrophotometer. The PC compounds
The fluorescence excitation wavelength of the PC s was determined as 610 nm, and this value was used to record fluorescence spectra of the PC s. The PC s and metal ions were mixed in a 1:1 ratio and the fluorescence spectra were measured. After recording the spectra of the mixtures, the same volume of metal ion solution was added and the fluorescence spectra recorded. Thus, increased spectrum intensity was recorded. The addition of the metal ion solution was repeated 10 times.
The PC compound
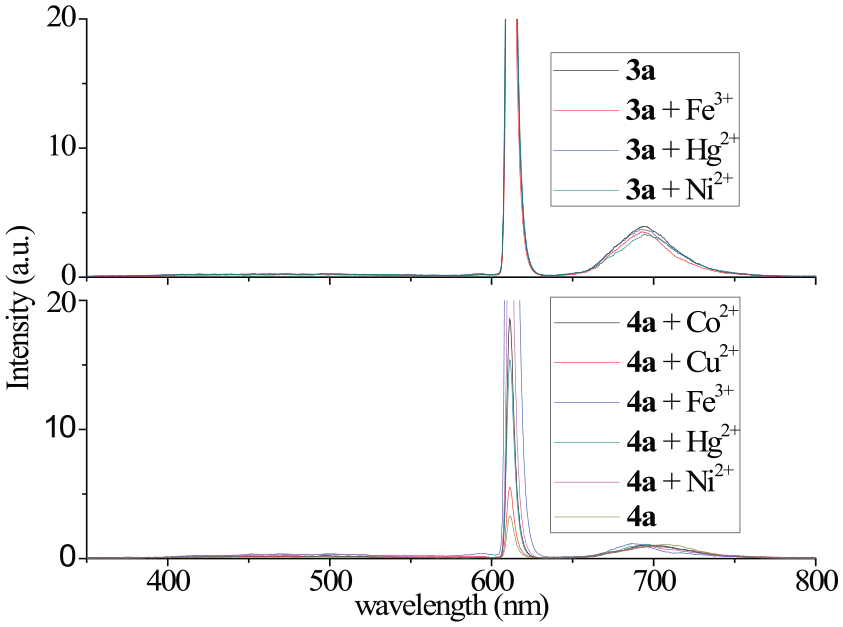
The metal ions that do not affect the fluorometric intensities of phthalocyanines

The metal ions that affect the fluorometric intensities of phthalocyanines
According to results of the fluroescence metal ion titrations of PC s
Conclusion
In conclusion, we have synthesized and characterized novel Zn(II), Mg(II), and Co(II) PC s which are substituted with chalcones bearing bromo or fluoro atoms. The structures of the synthesized compounds have been fully characterized by FTIR, 1H NMR, 13C NMR, MALDI-TOF MS, LC-HRMS, UV-Vis, and elemental analysis.
The electronic absorption spectra showed that all the MPcs were non-aggreging and were also well soluble in common organic solvents and compatible with the Beer–Lambert law. The electrochemical behavior was also investigated and the cyclic voltammograms gave one oxidation reaction for all the bromo-derived MPcs and one reduction reaction for all the fluoro-derived MPcs. Fe3+, Cd2+, Hg2+, Cu2+, Ni2+, and Co2+ ions were titrated fluorometrically with the PC compounds
Experimental
Materials and equipment
The used materials, equipment, and spectra (IR, NMR, and mass) are supplied as supplementary information that is available online.
Synthesis
General procedure for the synthesis of phthalonitrile derivatives 3 and 4
The chalcone-bound phthalonitriles were synthesized in dry DMF at 60°C by adding K2CO3. Equivalent amounts (1:1) of (4-nitrophthalonitrile and chalcone compounds
4-{3-((2E)-3-(3-bromophenyl)prop-2-enoyl)phenoxy}phthalonitrile (3)
Yield: (0.59 g) 60%, m.p. 76–79°C. IR (ATR),
4-{3-((2E)-3-(3-fluorophenyl)prop-2-enoyl)phenoxy}phthalonitrile (4)
Yield: (0.46 g) 60%, m.p. 98–101°C. IR (ATR),
General procedure for the synthesis of MPCs 3a, 4a; 3b, 4b; and 3c, 4c
The chalcone-substituted phthalonitriles,
The spectral data of MPCs 3a, 4a; 3b, 4b; and 3c, 4c
Supplemental Material
Supplementary_material – Supplemental material for New chalcone-substituted metallophthalocyanines: Synthesis, characterization, and investigation of their properties
Supplemental material, Supplementary_material for New chalcone-substituted metallophthalocyanines: Synthesis, characterization, and investigation of their properties by Ayse Aktas Kamiloglu, Hüseyin Karaca, Gonca Celik, Irfan Acar and Halit Kantekin in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Fund of Artvin Çoruh University, Project no: 2018.F91.02.01 (Artvin-Turkey). Huseyin Karaca is grateful to the research fund of Sakarya University (BAPK: 2018-3-12-165).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
